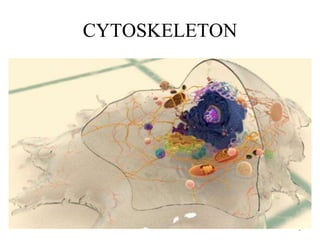
The document discusses the cytoskeleton's role in cell movement, including its importance in wound healing, immune response, and cancer cell metastasis. It explores the functions of different cytoskeletal components—actin filaments, microtubules, and intermediate filaments—in maintaining cell shape, enabling division, and facilitating intracellular transport. Additionally, the document covers the mechanisms of actin polymerization, cell-matrix interactions, and the dynamics of microtubules relevant to various cellular processes.