1. The document discusses the structure and function of eukaryotic cells and their organelles. It describes the plasma membrane, mitochondria, nucleus, endoplasmic reticulum, Golgi apparatus, lysosomes, peroxisomes, and cytoskeleton.
2. Key organelles include the mitochondria, which produces ATP through oxidative phosphorylation, and the nucleus, which contains DNA and directs protein synthesis.
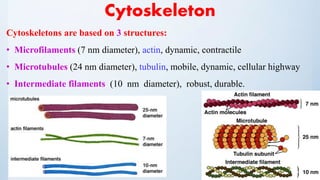
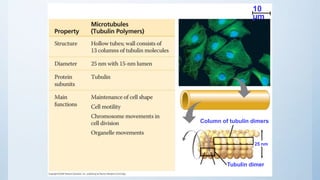
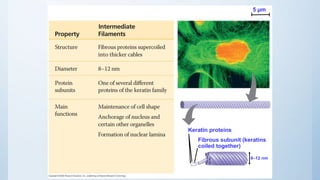
3. The cytoskeleton is composed of microfilaments, microtubules, and intermediate filaments which help maintain cell shape and enable cell movement.



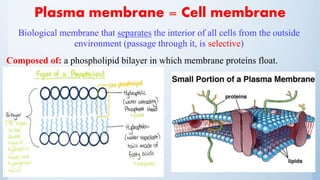
![Plasma membrane
– Transporters: (in the membrane) proteins which allow active transport.
– Receptors: (in the membrane) three-part structure, coupled to 2nd messenger
system]
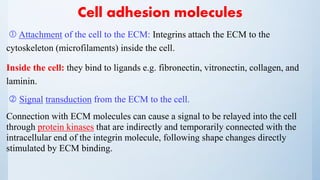
1. An extracellular component
2. A transmembrane component
3. An intra- cellular component
– The cell surface is irregularly pitted with invaginations and evaginations.
These include specialized structures for endocytosis and exocytosis.
– Other specializations (depending on the role of the cell) for example:
Rod photoreceptor: develops an evagination of the plasma membrane, which
folds upon itself many times to form stacked membranous discs.](https://image.slidesharecdn.com/biology-160326215521/85/Ocular-Biology-5-320.jpg)
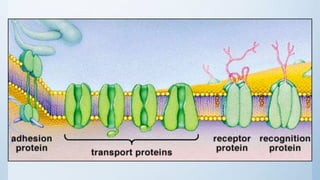














































![Cell signalling
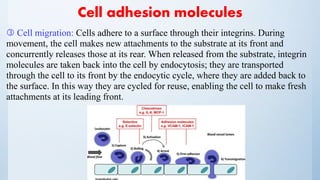
3. Membrane-bound receptors that couple to effectors through transducing
proteins [GTP-binding regulatory protein (G-protein)]
A range of signalling molecules use this type of receptor, e.g. muscarinic
acetylcholine receptors, adrenoceptors, dopamine receptors, 5-hydroxytryptamine
receptors, opioid receptors, peptide receptors, light receptors (rhodopsin), and
smell and taste receptors.
Receptor activation conformational change in the G-protein release of
GDP.
GTP takes its place and the α-GTP and βγ complex dissociate from the receptor
and go on to interact with effector molecules, which may be enzymes or ion
channels. The α-subunit has GTPase activity, which hydrolyses GTP and returns
the α-subunit to its inactive configuration and the α-GDPβγ heterotrimer reforms.](https://image.slidesharecdn.com/biology-160326215521/85/Ocular-Biology-61-320.jpg)















![Cells of C.T
1. Fibroblasts: synthesize and maintain the extracellular material.
Role: wound healing
[N.B.contractile fibroblasts (myofibroblasts) shrinkage of the scar tissue]
2. Adipocytes: store and maintain fat (a large droplet occupies most of
cytoplasm)
The main cell type in adipose tissue.
3. Chondroblasts and chondrocytes: produce and maintain cartilage.
4. Osteoblasts, osteocytes, and osteoclasts: are specialized cells that produce,
maintain, and break down bone, respectively.](https://image.slidesharecdn.com/biology-160326215521/85/Ocular-Biology-78-320.jpg)



![Extracellular material of C.T
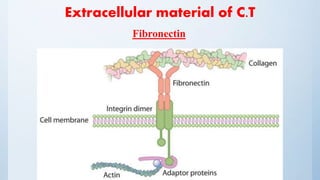
Fibronectin
Adhesive ECM glycoprotein.
Binds to integrins and ECM components such as collagen, fibrin, and heparan
sulphate proteoglycans (e.g. syndecans)
Structure: a dimer*, consisting of two nearly identical monomers linked by a
pair of disulphide bonds* [ high-molecular weight (~440 kDa) ]](https://image.slidesharecdn.com/biology-160326215521/85/Ocular-Biology-83-320.jpg)


![Extracellular material of C.T
Collagen
Fibrous protein with great tensile strength and resistance to stretching.
The main component of connective tissue, it is the most abundant protein in
mammals [It makes up 25% of the protein in mammals]](https://image.slidesharecdn.com/biology-160326215521/85/Ocular-Biology-86-320.jpg)




























