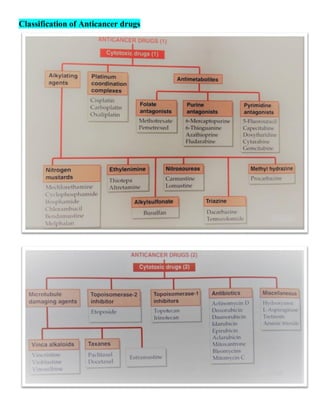
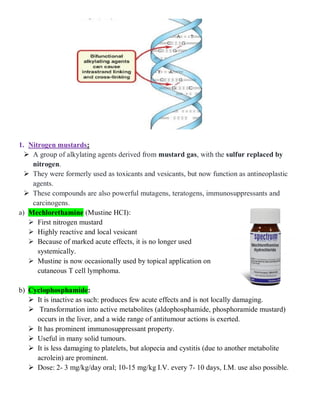
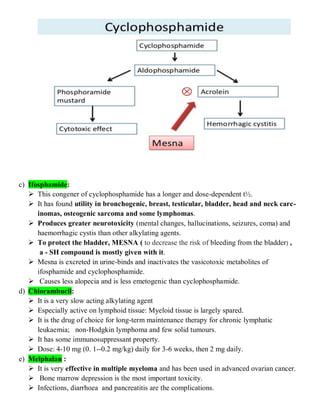
The document discusses anticancer drugs, emphasizing their classification, the nature of neoplasms, and general toxicity, particularly from alkylating agents. It outlines the characteristics of benign and malignant tumors, the mechanisms and types of anticancer therapy, and the specific toxicities associated with various drug classes. Additionally, the document covers the development of cancer therapy and the challenges faced due to toxic effects on bone marrow, lymphatic systems, and other organs.