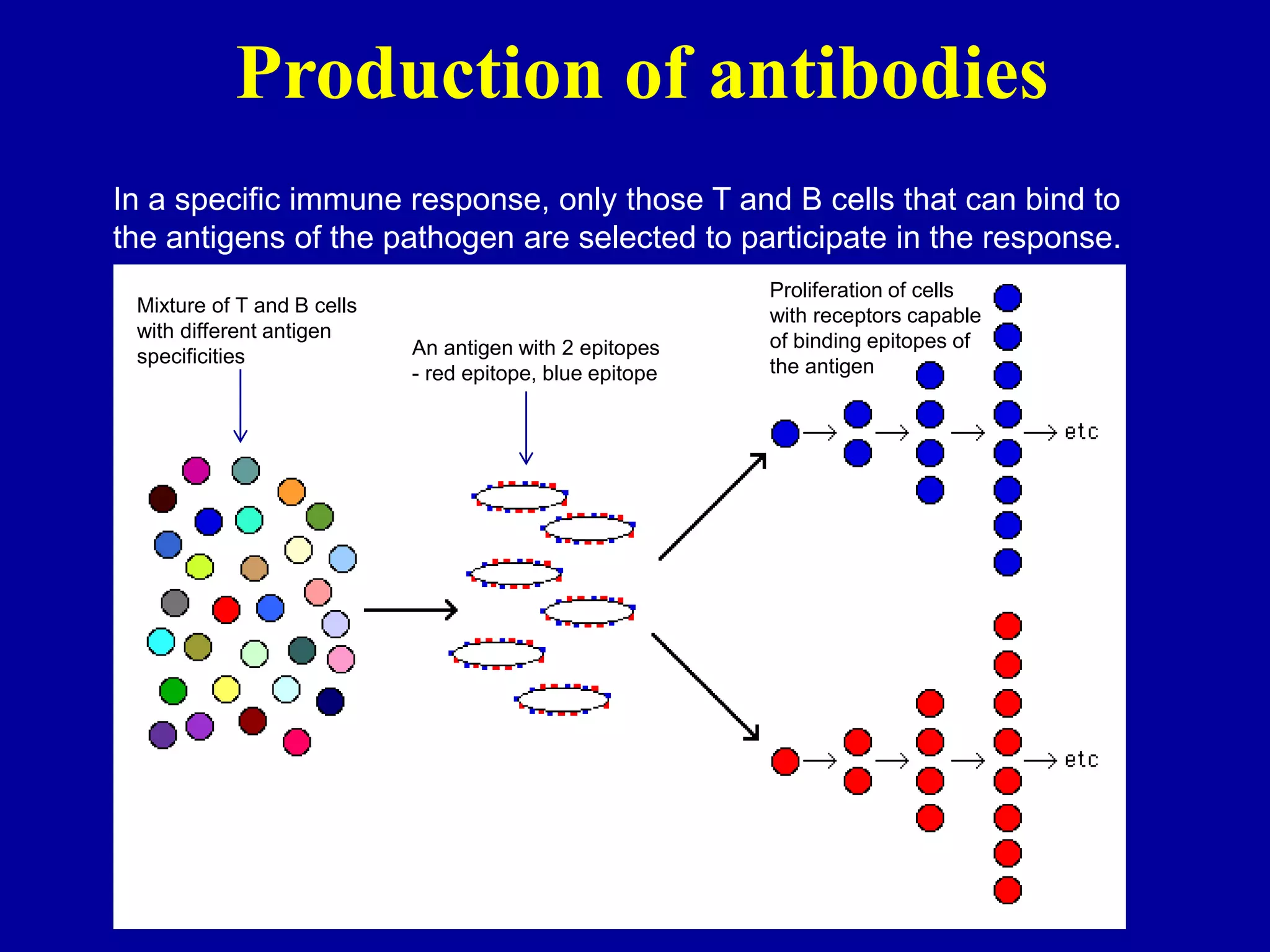
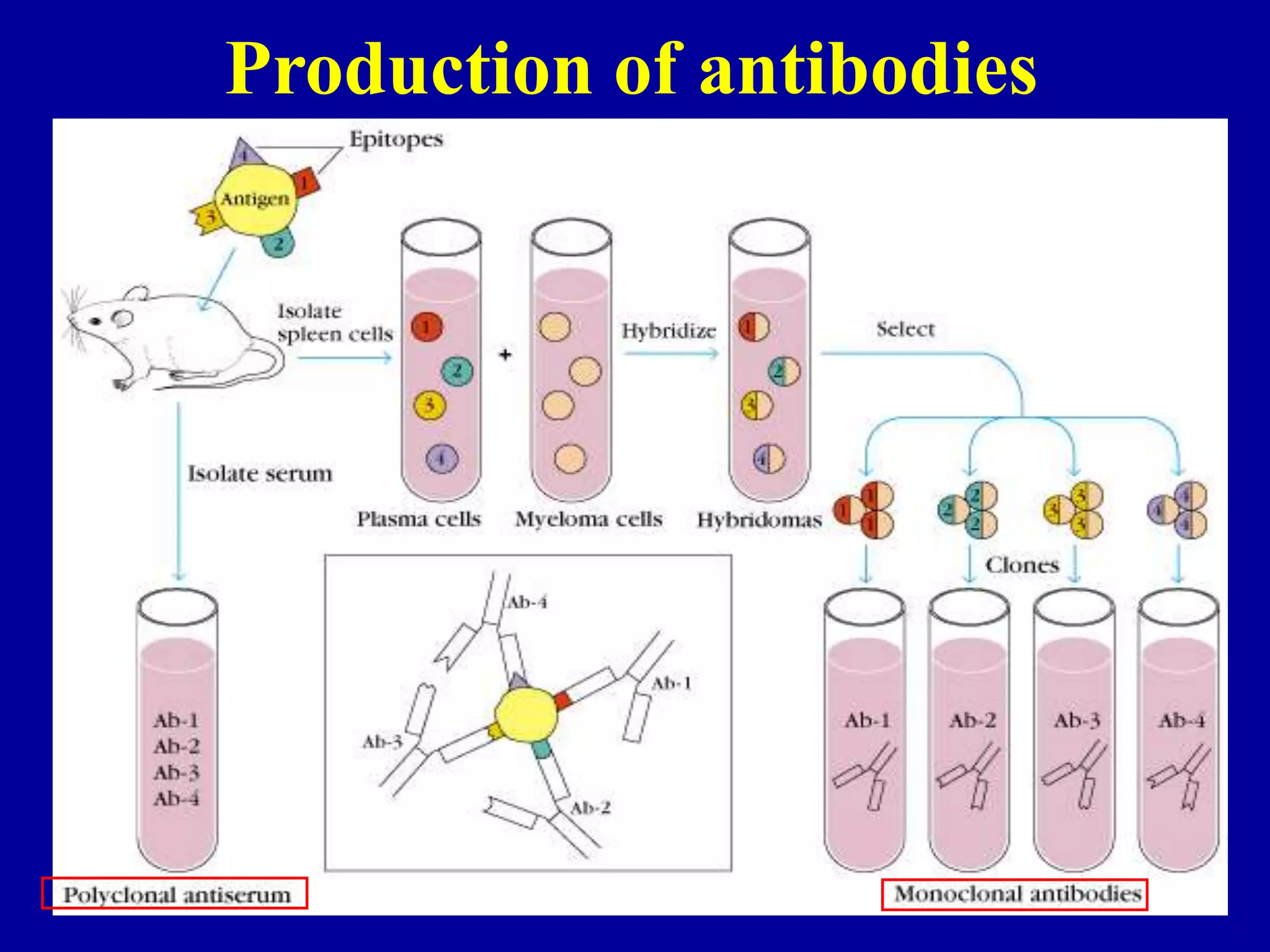
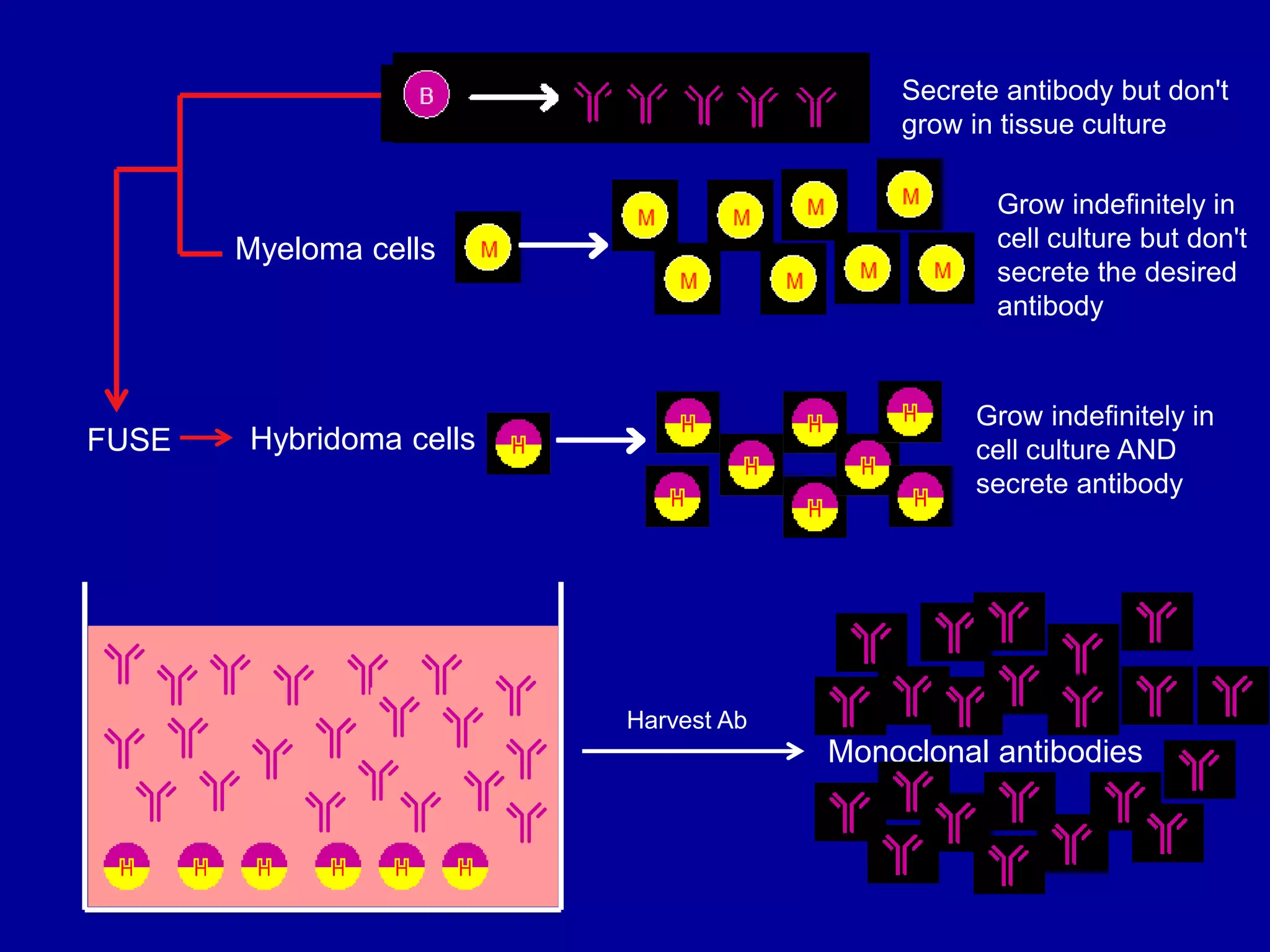
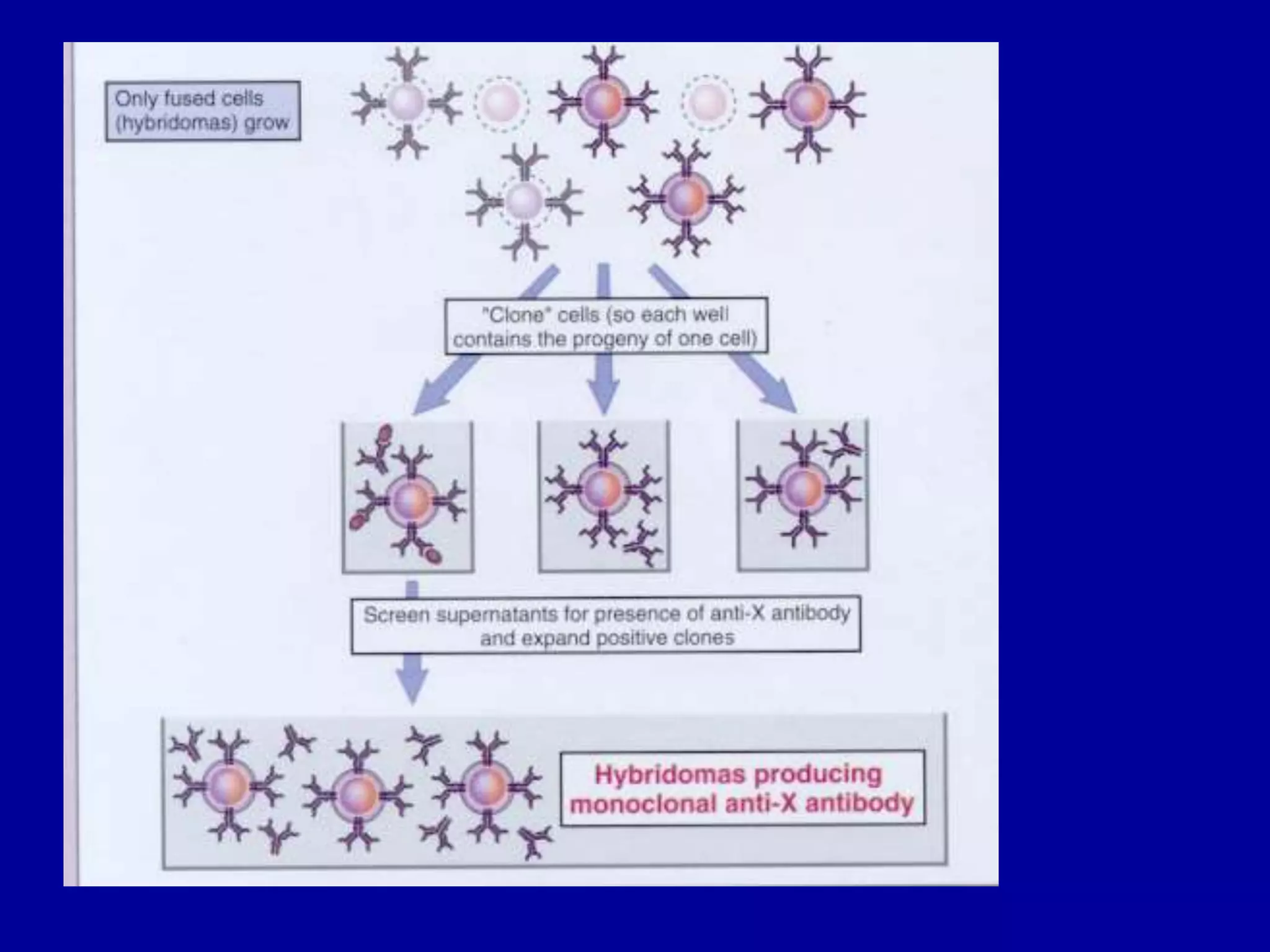
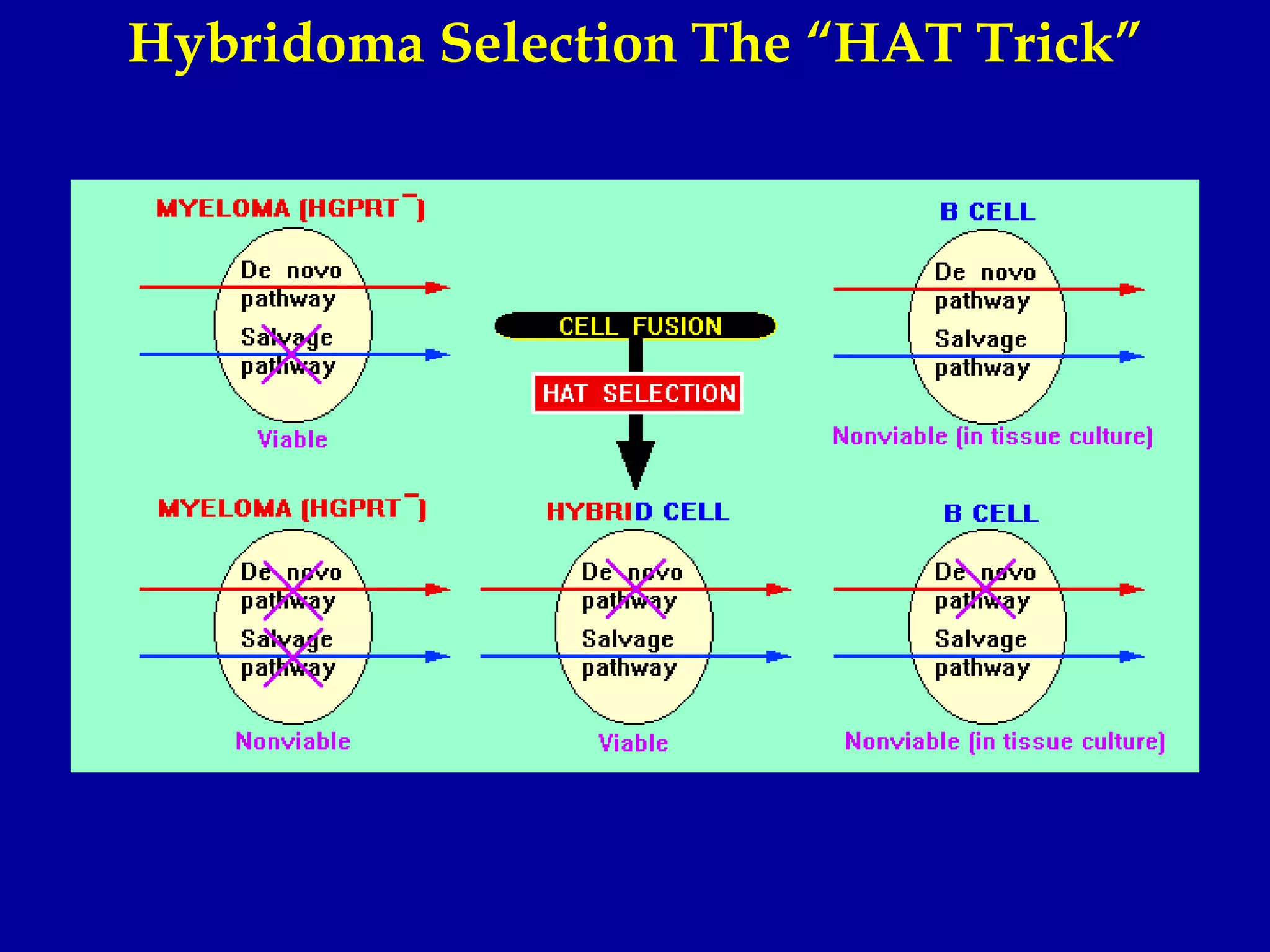
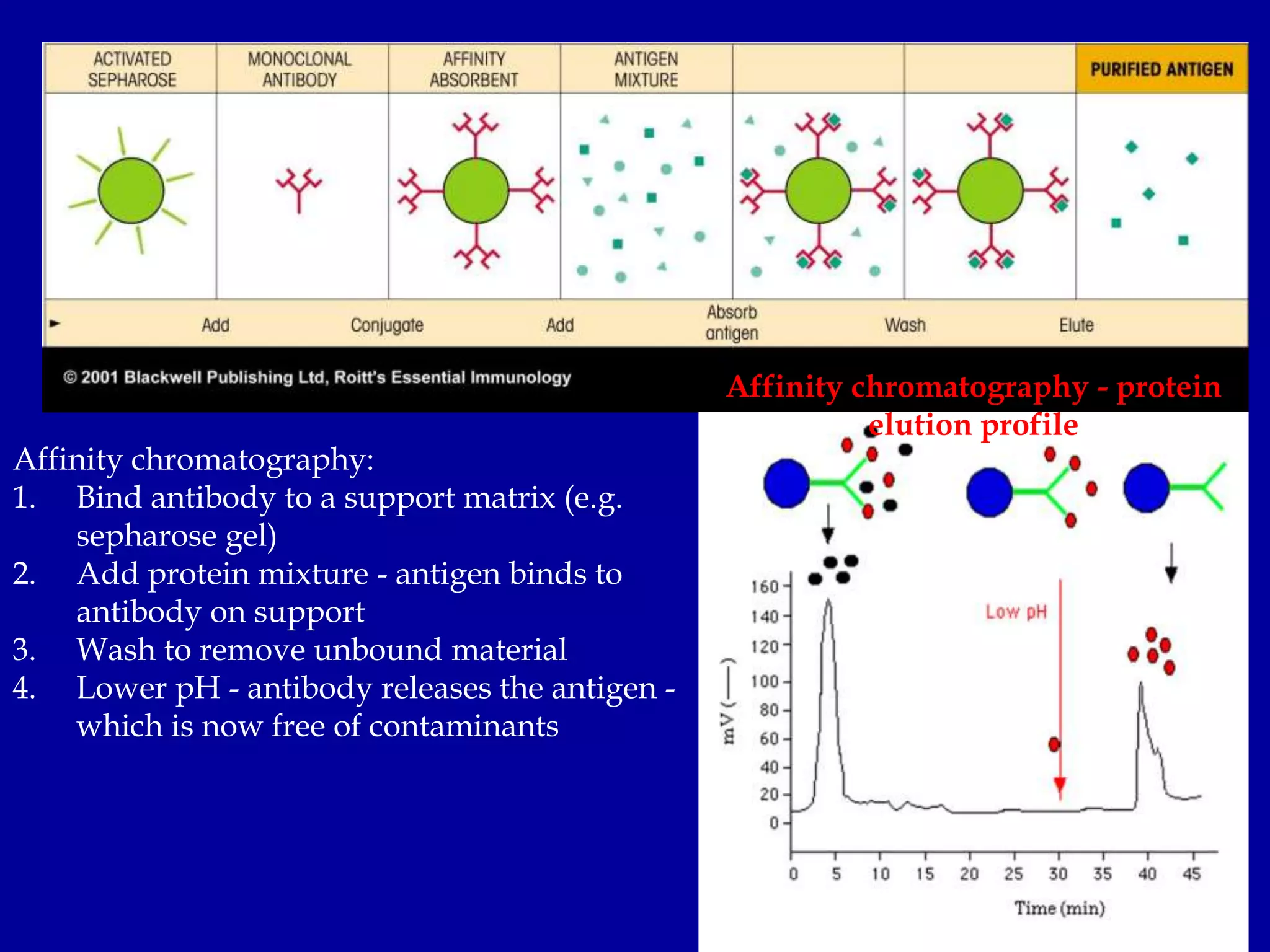
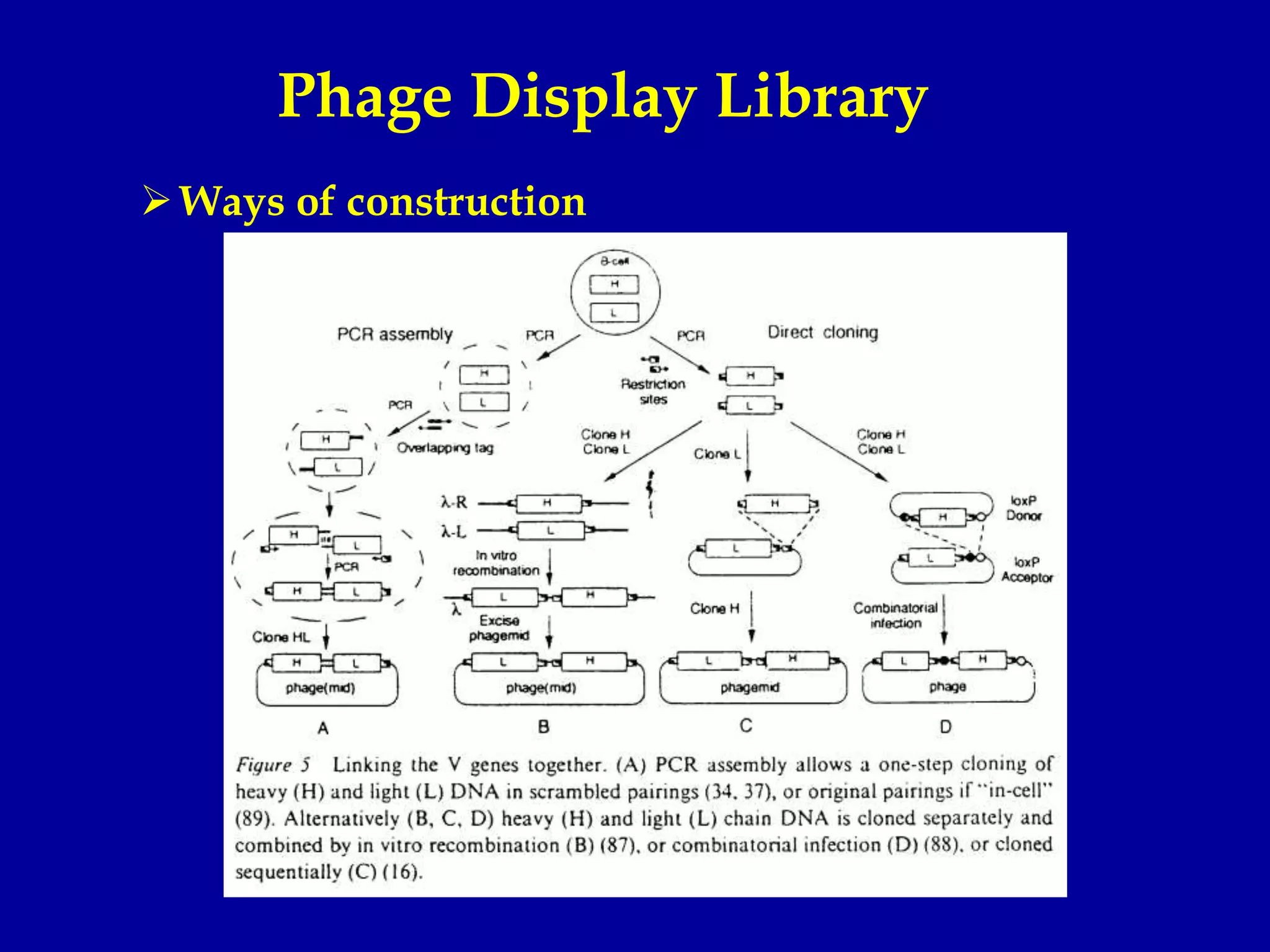
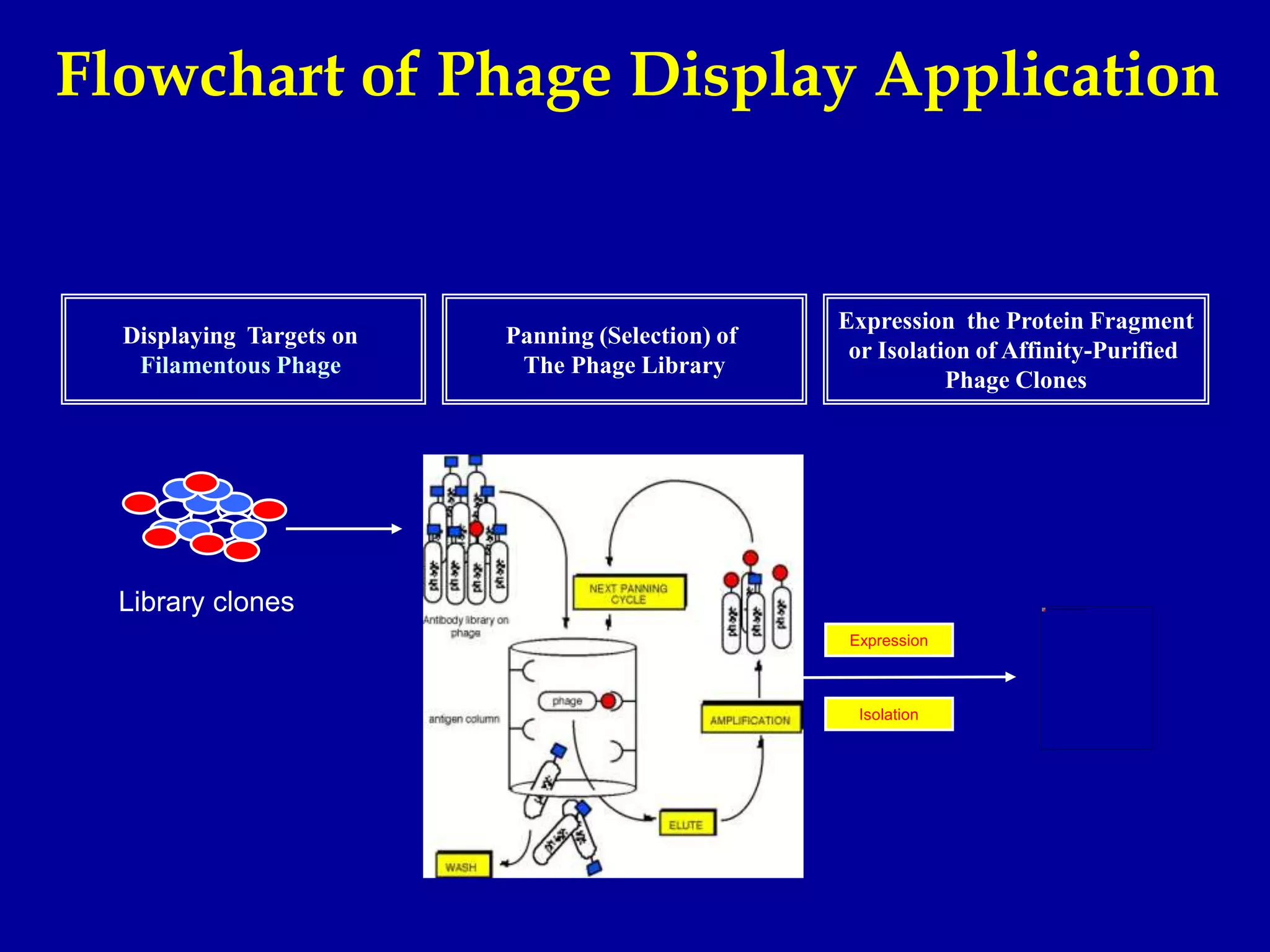
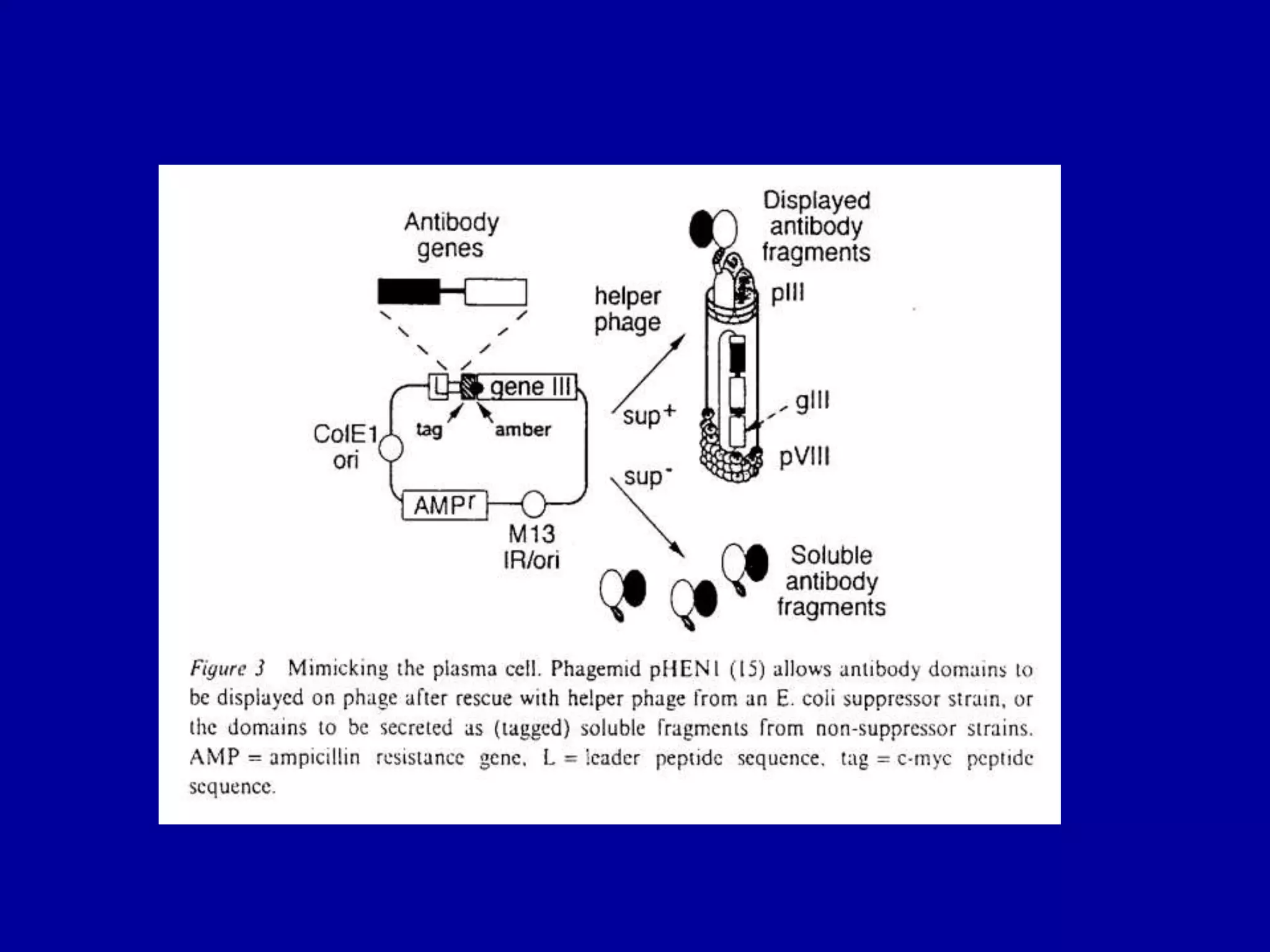
This document discusses techniques for producing antibodies, including both polyclonal and monoclonal antibodies. It describes the hybridoma technique developed by Kohler and Milstein, which involves fusing B cells that produce antibodies with myeloma cells to create immortal cell lines that produce monoclonal antibodies targeting a single epitope. The document outlines the characteristics of immunogens and antibodies, comparing polyclonal versus monoclonal antibodies. It also discusses applications of monoclonal antibodies in research, diagnostics, and therapeutics.