Aminoglycoside SAR one page.doc
•
0 likes•195 views
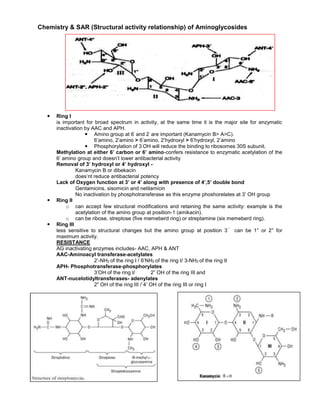
This document summarizes key structural activity relationships of aminoglycoside antibiotics. It notes that ring I is important for broad spectrum activity but is also the major site of inactivation by aminoglycoside modifying enzymes. The amino groups at positions 6' and 2' on ring I are particularly important for activity. Phosphorylation of the 3'OH on ring I reduces binding to the 30S ribosomal subunit. Methylation of the 6' carbon or amino group confers resistance to enzymatic acetylation of the 6' amino group without reducing antibacterial potency. Removal of the 3' or 4' hydroxyl, as seen in kanamycin B or dibekacin, does not reduce potency
Report
Share
Report
Share
Recommended
Sulphonamides (Sulfonamides) and Sulfones || B.Pharm VI Semester || Medicinal...

Sulfonamides, Sulfones, Historical development, chemistry, classification and SAR of Sulfonamides:
Sulphamethizole, Sulfisoxazole, Sulphamethizine, Sulfacetamide*, Sulphapyridine, Sulfamethoxaole*,
Sulphadiazine, Mefenide acetate, Sulfasalazine.
Folate reductase inhibitors, Trimethoprim, Cotrimoxazole,
Sulfones, Dapsone
ANTI-MALARIAL DRUGS AND ANALOGUES

THIS PRESENTATION ABOUT ANTIMALARIAL DRUGS DETAILING THE COMPLETE INFORMATION ABOUT THE DRUGS USED WITH ITS MECHANISM OF ACTION, STRUCTURAL ACTIVITY AND DOSES.
ANTIBIOTICS[PENICILLINS] MEDICINAL CHEMISTRY BY RAVISANKAR![ANTIBIOTICS[PENICILLINS] MEDICINAL CHEMISTRY BY RAVISANKAR](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![ANTIBIOTICS[PENICILLINS] MEDICINAL CHEMISTRY BY RAVISANKAR](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
ANTIBIOTICS[PENICILLINS} MEDICIANL CHEMISTRY BY P.RAVISANKAR
VIGNAN PHARMACY COLLEGE
VADLAMUDI
GUNTUR- A.P
INDIA
Quinolines- Antimalarial drugs.pptx

General introduction about quinoline anti-malarial drugs, QSAR, examples, Applications and side effects.
Macroloid antibiotics

Definition
Source
Chemistry
Structure activity relationship
Physical & chemical properties
Mechanism of action
Types of macrolide
Pharmacokinetics
Macrolide resistance
Spectrum of activity
Indication
Contraindication
Side effect
Market preparation
Recommended
Sulphonamides (Sulfonamides) and Sulfones || B.Pharm VI Semester || Medicinal...

Sulfonamides, Sulfones, Historical development, chemistry, classification and SAR of Sulfonamides:
Sulphamethizole, Sulfisoxazole, Sulphamethizine, Sulfacetamide*, Sulphapyridine, Sulfamethoxaole*,
Sulphadiazine, Mefenide acetate, Sulfasalazine.
Folate reductase inhibitors, Trimethoprim, Cotrimoxazole,
Sulfones, Dapsone
ANTI-MALARIAL DRUGS AND ANALOGUES

THIS PRESENTATION ABOUT ANTIMALARIAL DRUGS DETAILING THE COMPLETE INFORMATION ABOUT THE DRUGS USED WITH ITS MECHANISM OF ACTION, STRUCTURAL ACTIVITY AND DOSES.
ANTIBIOTICS[PENICILLINS] MEDICINAL CHEMISTRY BY RAVISANKAR![ANTIBIOTICS[PENICILLINS] MEDICINAL CHEMISTRY BY RAVISANKAR](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![ANTIBIOTICS[PENICILLINS] MEDICINAL CHEMISTRY BY RAVISANKAR](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
ANTIBIOTICS[PENICILLINS} MEDICIANL CHEMISTRY BY P.RAVISANKAR
VIGNAN PHARMACY COLLEGE
VADLAMUDI
GUNTUR- A.P
INDIA
Quinolines- Antimalarial drugs.pptx

General introduction about quinoline anti-malarial drugs, QSAR, examples, Applications and side effects.
Macroloid antibiotics

Definition
Source
Chemistry
Structure activity relationship
Physical & chemical properties
Mechanism of action
Types of macrolide
Pharmacokinetics
Macrolide resistance
Spectrum of activity
Indication
Contraindication
Side effect
Market preparation
Anti Malarial Drugs of medicinal chemistry

This slide contain information about Anti Malarial Drugs and their description with the synthesis of Chloroquine and pamaquine
SAR of quinolines
Miscellaneous agents of anti Malarial
Cephalosporin- Beta lactam Antibiotic 

This presentation highlights on the Introduction, chemistry, classification, structures, SAR and Mechanism of Action (MOA) of cephalosporins
Tetracyclines Medicinal Chemistry

tetracyclines, structure activity relationship, Mechanism of action
Aminoglycosides (Antibiotics)

Here is the presentation of aminoglycosides- antibiotics as per PCI syllabus (Pharmacy)
Chemotherapy of malignancy

Malignancy is most familiar as a characterization of cancer.Chemotherapy is a category of cancer treatment that uses one or more anti-cancer drugs as part of a standardized chemotherapy regimen
Tetracyclines- Ravisankar- Medicinal chemistry, Definition,classification,S...

Tetracyclines- Medicinal chemistry, Definition,classification,SAR,Mechanism of action, Side effects, uses.
By-Ravisankar.P,Vignan Pharmacy College, Vadlamudi
medicinal chemistry of Anticancer agents

introduction,cell cycle,classification,chemical structure,synthesis of anticancer agents.
Sythesis of heterocyclic drugs ketoconazole and metronidazole

A Heterocyclic compounds are those which has atoms of at least two different elements as members of its ring.
Heterocyclic chemistry is a branch of organic chemistry dealing with the synthesis, properties, and applications of these heterocycles.
002. Cephalosporins for students 2023 Prof. P. Ravisankar.pdf

Cephalosporins, Why Cephalosporins? Advantages of cephalosporins over penicillin, Mechanism of action of cephalosporins, Classification of cephalosporins, Structures of some important cephalosporins and cephamycins, Oximinocephalosporins, SAR of cephalosporins,Hydrolytic reactions, degradation and stability of cephalosporins, Uses of cephalosporins, Comparison between 6-APA and 7-ACA and penam and cepham.
Basic concepts and application of prodrug design

Aprodrugisachemicallymodifiedinertdrugprecursor,whichuponbiotransformationliberatesthepharmacologicallyactiveparentcompound.
H1 and h2 receptor antagonist

H1 and h2 receptor antagonist , their distribution, uses, SAR , Ppis (proton pump inhibitors)
Antiprotozoal Drugs- Medicinal Chemistry-Pharmacy

Antiprotozoal Drugs- Medicinal Chemistry-Pharmacy- As per PCI Syllabus- Lecture Notes
Complete Chapter of Anti-malarial Drugs Part -1 

You can watch this lecture on youtube
https://www.youtube.com/watch?v=7_oFbKXamac
Complete Chapter of
Anti-malarial Drugs Part -1
Introduction
-Stages Of Malaria
-Chemical classification
-SAR of Anti-Malarial Drugs
Properties of Amino Acids.pptx

Properties of amino acids:
- Amino Acids have an Asymmetric Center
- D and L stereoisomerism of amino acids
- Acid-Base Properties of Amino Acids
- Titration of amino acids
- Absorption
- Solubility
- Chemical properties of amino acid
More Related Content
What's hot
Anti Malarial Drugs of medicinal chemistry

This slide contain information about Anti Malarial Drugs and their description with the synthesis of Chloroquine and pamaquine
SAR of quinolines
Miscellaneous agents of anti Malarial
Cephalosporin- Beta lactam Antibiotic 

This presentation highlights on the Introduction, chemistry, classification, structures, SAR and Mechanism of Action (MOA) of cephalosporins
Tetracyclines Medicinal Chemistry

tetracyclines, structure activity relationship, Mechanism of action
Aminoglycosides (Antibiotics)

Here is the presentation of aminoglycosides- antibiotics as per PCI syllabus (Pharmacy)
Chemotherapy of malignancy

Malignancy is most familiar as a characterization of cancer.Chemotherapy is a category of cancer treatment that uses one or more anti-cancer drugs as part of a standardized chemotherapy regimen
Tetracyclines- Ravisankar- Medicinal chemistry, Definition,classification,S...

Tetracyclines- Medicinal chemistry, Definition,classification,SAR,Mechanism of action, Side effects, uses.
By-Ravisankar.P,Vignan Pharmacy College, Vadlamudi
medicinal chemistry of Anticancer agents

introduction,cell cycle,classification,chemical structure,synthesis of anticancer agents.
Sythesis of heterocyclic drugs ketoconazole and metronidazole

A Heterocyclic compounds are those which has atoms of at least two different elements as members of its ring.
Heterocyclic chemistry is a branch of organic chemistry dealing with the synthesis, properties, and applications of these heterocycles.
002. Cephalosporins for students 2023 Prof. P. Ravisankar.pdf

Cephalosporins, Why Cephalosporins? Advantages of cephalosporins over penicillin, Mechanism of action of cephalosporins, Classification of cephalosporins, Structures of some important cephalosporins and cephamycins, Oximinocephalosporins, SAR of cephalosporins,Hydrolytic reactions, degradation and stability of cephalosporins, Uses of cephalosporins, Comparison between 6-APA and 7-ACA and penam and cepham.
Basic concepts and application of prodrug design

Aprodrugisachemicallymodifiedinertdrugprecursor,whichuponbiotransformationliberatesthepharmacologicallyactiveparentcompound.
H1 and h2 receptor antagonist

H1 and h2 receptor antagonist , their distribution, uses, SAR , Ppis (proton pump inhibitors)
Antiprotozoal Drugs- Medicinal Chemistry-Pharmacy

Antiprotozoal Drugs- Medicinal Chemistry-Pharmacy- As per PCI Syllabus- Lecture Notes
Complete Chapter of Anti-malarial Drugs Part -1 

You can watch this lecture on youtube
https://www.youtube.com/watch?v=7_oFbKXamac
Complete Chapter of
Anti-malarial Drugs Part -1
Introduction
-Stages Of Malaria
-Chemical classification
-SAR of Anti-Malarial Drugs
What's hot (20)
Tetracyclines- Ravisankar- Medicinal chemistry, Definition,classification,S...

Tetracyclines- Ravisankar- Medicinal chemistry, Definition,classification,S...
Sythesis of heterocyclic drugs ketoconazole and metronidazole

Sythesis of heterocyclic drugs ketoconazole and metronidazole
002. Cephalosporins for students 2023 Prof. P. Ravisankar.pdf

002. Cephalosporins for students 2023 Prof. P. Ravisankar.pdf
Similar to Aminoglycoside SAR one page.doc
Properties of Amino Acids.pptx

Properties of amino acids:
- Amino Acids have an Asymmetric Center
- D and L stereoisomerism of amino acids
- Acid-Base Properties of Amino Acids
- Titration of amino acids
- Absorption
- Solubility
- Chemical properties of amino acid
Protection for carboxylic group & Protection for the Amino group

Protection for carboxylic group & Protection for the Amino groupCollege of Pharmacy,Sri Ramakrishna Institute of Paramedical Sciences,Coimbatore
PROTECTING GROUPS (PROTECTION FOR THE CARBOXYLIC GROUP & PROTECTION FOR THE AMINO GROUPSimilar to Aminoglycoside SAR one page.doc (7)
Protection for carboxylic group & Protection for the Amino group

Protection for carboxylic group & Protection for the Amino group
More from Mahavir Ghante
Neuroglia, nerve fiber, action potential, synapse, neurotransmitters.ppt

Neuroglia, nerve fiber, action potential, synapse, neurotransmitters
More from Mahavir Ghante (11)
Neuroglia, nerve fiber, action potential, synapse, neurotransmitters.ppt

Neuroglia, nerve fiber, action potential, synapse, neurotransmitters.ppt
Recently uploaded
Flu Vaccine Alert in Bangalore Karnataka

As flu season approaches, health officials in Bangalore, Karnataka, are urging residents to get their flu vaccinations. The seasonal flu, while common, can lead to severe health complications, particularly for vulnerable populations such as young children, the elderly, and those with underlying health conditions.
Dr. Vidisha Kumari, a leading epidemiologist in Bangalore, emphasizes the importance of getting vaccinated. "The flu vaccine is our best defense against the influenza virus. It not only protects individuals but also helps prevent the spread of the virus in our communities," he says.
This year, the flu season is expected to coincide with a potential increase in other respiratory illnesses. The Karnataka Health Department has launched an awareness campaign highlighting the significance of flu vaccinations. They have set up multiple vaccination centers across Bangalore, making it convenient for residents to receive their shots.
To encourage widespread vaccination, the government is also collaborating with local schools, workplaces, and community centers to facilitate vaccination drives. Special attention is being given to ensuring that the vaccine is accessible to all, including marginalized communities who may have limited access to healthcare.
Residents are reminded that the flu vaccine is safe and effective. Common side effects are mild and may include soreness at the injection site, mild fever, or muscle aches. These side effects are generally short-lived and far less severe than the flu itself.
Healthcare providers are also stressing the importance of continuing COVID-19 precautions. Wearing masks, practicing good hand hygiene, and maintaining social distancing are still crucial, especially in crowded places.
Protect yourself and your loved ones by getting vaccinated. Together, we can help keep Bangalore healthy and safe this flu season. For more information on vaccination centers and schedules, residents can visit the Karnataka Health Department’s official website or follow their social media pages.
Stay informed, stay safe, and get your flu shot today!
HOT NEW PRODUCT! BIG SALES FAST SHIPPING NOW FROM CHINA!! EU KU DB BK substit...

Contact us if you are interested:
Email / Skype : kefaya1771@gmail.com
Threema: PXHY5PDH
New BATCH Ku !!! MUCH IN DEMAND FAST SALE EVERY BATCH HAPPY GOOD EFFECT BIG BATCH !
Contact me on Threema or skype to start big business!!
Hot-sale products:
NEW HOT EUTYLONE WHITE CRYSTAL!!
5cl-adba precursor (semi finished )
5cl-adba raw materials
ADBB precursor (semi finished )
ADBB raw materials
APVP powder
5fadb/4f-adb
Jwh018 / Jwh210
Eutylone crystal
Protonitazene (hydrochloride) CAS: 119276-01-6
Flubrotizolam CAS: 57801-95-3
Metonitazene CAS: 14680-51-4
Payment terms: Western Union,MoneyGram,Bitcoin or USDT.
Deliver Time: Usually 7-15days
Shipping method: FedEx, TNT, DHL,UPS etc.Our deliveries are 100% safe, fast, reliable and discreet.
Samples will be sent for your evaluation!If you are interested in, please contact me, let's talk details.
We specializes in exporting high quality Research chemical, medical intermediate, Pharmaceutical chemicals and so on. Products are exported to USA, Canada, France, Korea, Japan,Russia, Southeast Asia and other countries.
Evaluation of antidepressant activity of clitoris ternatea in animals

Evaluation of antidepressant activity of clitoris ternatea in animals
Physiology of Chemical Sensation of smell.pdf

Title: Sense of Smell
Presenter: Dr. Faiza, Assistant Professor of Physiology
Qualifications:
MBBS (Best Graduate, AIMC Lahore)
FCPS Physiology
ICMT, CHPE, DHPE (STMU)
MPH (GC University, Faisalabad)
MBA (Virtual University of Pakistan)
Learning Objectives:
Describe the primary categories of smells and the concept of odor blindness.
Explain the structure and location of the olfactory membrane and mucosa, including the types and roles of cells involved in olfaction.
Describe the pathway and mechanisms of olfactory signal transmission from the olfactory receptors to the brain.
Illustrate the biochemical cascade triggered by odorant binding to olfactory receptors, including the role of G-proteins and second messengers in generating an action potential.
Identify different types of olfactory disorders such as anosmia, hyposmia, hyperosmia, and dysosmia, including their potential causes.
Key Topics:
Olfactory Genes:
3% of the human genome accounts for olfactory genes.
400 genes for odorant receptors.
Olfactory Membrane:
Located in the superior part of the nasal cavity.
Medially: Folds downward along the superior septum.
Laterally: Folds over the superior turbinate and upper surface of the middle turbinate.
Total surface area: 5-10 square centimeters.
Olfactory Mucosa:
Olfactory Cells: Bipolar nerve cells derived from the CNS (100 million), with 4-25 olfactory cilia per cell.
Sustentacular Cells: Produce mucus and maintain ionic and molecular environment.
Basal Cells: Replace worn-out olfactory cells with an average lifespan of 1-2 months.
Bowman’s Gland: Secretes mucus.
Stimulation of Olfactory Cells:
Odorant dissolves in mucus and attaches to receptors on olfactory cilia.
Involves a cascade effect through G-proteins and second messengers, leading to depolarization and action potential generation in the olfactory nerve.
Quality of a Good Odorant:
Small (3-20 Carbon atoms), volatile, water-soluble, and lipid-soluble.
Facilitated by odorant-binding proteins in mucus.
Membrane Potential and Action Potential:
Resting membrane potential: -55mV.
Action potential frequency in the olfactory nerve increases with odorant strength.
Adaptation Towards the Sense of Smell:
Rapid adaptation within the first second, with further slow adaptation.
Psychological adaptation greater than receptor adaptation, involving feedback inhibition from the central nervous system.
Primary Sensations of Smell:
Camphoraceous, Musky, Floral, Pepperminty, Ethereal, Pungent, Putrid.
Odor Detection Threshold:
Examples: Hydrogen sulfide (0.0005 ppm), Methyl-mercaptan (0.002 ppm).
Some toxic substances are odorless at lethal concentrations.
Characteristics of Smell:
Odor blindness for single substances due to lack of appropriate receptor protein.
Behavioral and emotional influences of smell.
Transmission of Olfactory Signals:
From olfactory cells to glomeruli in the olfactory bulb, involving lateral inhibition.
Primitive, less old, and new olfactory systems with different path
Charaka Samhita Sutra sthana Chapter 15 Upakalpaniyaadhyaya

Charaka Samhita Sutra sthana Chapter 15 Upakalpaniyaadhyaya
New Directions in Targeted Therapeutic Approaches for Older Adults With Mantl...

i3 Health is pleased to make the speaker slides from this activity available for use as a non-accredited self-study or teaching resource.
This slide deck presented by Dr. Kami Maddocks, Professor-Clinical in the Division of Hematology and
Associate Division Director for Ambulatory Operations
The Ohio State University Comprehensive Cancer Center, will provide insight into new directions in targeted therapeutic approaches for older adults with mantle cell lymphoma.
STATEMENT OF NEED
Mantle cell lymphoma (MCL) is a rare, aggressive B-cell non-Hodgkin lymphoma (NHL) accounting for 5% to 7% of all lymphomas. Its prognosis ranges from indolent disease that does not require treatment for years to very aggressive disease, which is associated with poor survival (Silkenstedt et al, 2021). Typically, MCL is diagnosed at advanced stage and in older patients who cannot tolerate intensive therapy (NCCN, 2022). Although recent advances have slightly increased remission rates, recurrence and relapse remain very common, leading to a median overall survival between 3 and 6 years (LLS, 2021). Though there are several effective options, progress is still needed towards establishing an accepted frontline approach for MCL (Castellino et al, 2022). Treatment selection and management of MCL are complicated by the heterogeneity of prognosis, advanced age and comorbidities of patients, and lack of an established standard approach for treatment, making it vital that clinicians be familiar with the latest research and advances in this area. In this activity chaired by Michael Wang, MD, Professor in the Department of Lymphoma & Myeloma at MD Anderson Cancer Center, expert faculty will discuss prognostic factors informing treatment, the promising results of recent trials in new therapeutic approaches, and the implications of treatment resistance in therapeutic selection for MCL.
Target Audience
Hematology/oncology fellows, attending faculty, and other health care professionals involved in the treatment of patients with mantle cell lymphoma (MCL).
Learning Objectives
1.) Identify clinical and biological prognostic factors that can guide treatment decision making for older adults with MCL
2.) Evaluate emerging data on targeted therapeutic approaches for treatment-naive and relapsed/refractory MCL and their applicability to older adults
3.) Assess mechanisms of resistance to targeted therapies for MCL and their implications for treatment selection
NVBDCP.pptx Nation vector borne disease control program

NVBDCP was launched in 2003-2004 . Vector-Borne Disease: Disease that results from an infection transmitted to humans and other animals by blood-feeding arthropods, such as mosquitoes, ticks, and fleas. Examples of vector-borne diseases include Dengue fever, West Nile Virus, Lyme disease, and malaria.
263778731218 Abortion Clinic /Pills In Harare ,

263778731218 Abortion Clinic /Pills In Harare ,ABORTION WOMEN’S CLINIC +27730423979 IN women clinic we believe that every woman should be able to make choices in her pregnancy. Our job is to provide compassionate care, safety,affordable and confidential services. That’s why we have won the trust from all generations of women all over the world. we use non surgical method(Abortion pills) to terminate…Dr.LISA +27730423979women Clinic is committed to providing the highest quality of obstetrical and gynecological care to women of all ages. Our dedicated staff aim to treat each patient and her health concerns with compassion and respect.Our dedicated group ABORTION WOMEN’S CLINIC +27730423979 IN women clinic we believe that every woman should be able to make choices in her pregnancy. Our job is to provide compassionate care, safety,affordable and confidential services. That’s why we have won the trust from all generations of women all over the world. we use non surgical method(Abortion pills) to terminate…Dr.LISA +27730423979women Clinic is committed to providing the highest quality of obstetrical and gynecological care to women of all ages. Our dedicated staff aim to treat each patient and her health concerns with compassion and respect.Our dedicated group of receptionists, nurses, and physicians have worked together as a teamof receptionists, nurses, and physicians have worked together as a team wwww.lisywomensclinic.co.za/
KDIGO 2024 guidelines for diabetologists

KDIGO guidelines 2024 for evaluation and management of CKD, related to diabetes and management of diabetic kidney disease
ACUTE SCROTUM.....pdf. ACUTE SCROTAL CONDITIOND

Acute scrotum is a general term referring to an emergency condition affecting the contents or the wall of the scrotum.
There are a number of conditions that present acutely, predominantly with pain and/or swelling
A careful and detailed history and examination, and in some cases, investigations allow differentiation between these diagnoses. A prompt diagnosis is essential as the patient may require urgent surgical intervention
Testicular torsion refers to twisting of the spermatic cord, causing ischaemia of the testicle.
Testicular torsion results from inadequate fixation of the testis to the tunica vaginalis producing ischemia from reduced arterial inflow and venous outflow obstruction.
The prevalence of testicular torsion in adult patients hospitalized with acute scrotal pain is approximately 25 to 50 percent
ARTHROLOGY PPT NCISM SYLLABUS AYURVEDA STUDENTS

PPT RELATED TO ARTHROLOGY ACCORDING TO NCISM AYURVEDA
The Normal Electrocardiogram - Part I of II

These lecture slides, by Dr Sidra Arshad, offer a quick overview of physiological basis of a normal electrocardiogram.
Learning objectives:
1. Define an electrocardiogram (ECG) and electrocardiography
2. Describe how dipoles generated by the heart produce the waveforms of the ECG
3. Describe the components of a normal electrocardiogram of a typical bipolar leads (limb II)
4. Differentiate between intervals and segments
5. Enlist some common indications for obtaining an ECG
Study Resources:
1. Chapter 11, Guyton and Hall Textbook of Medical Physiology, 14th edition
2. Chapter 9, Human Physiology - From Cells to Systems, Lauralee Sherwood, 9th edition
3. Chapter 29, Ganong’s Review of Medical Physiology, 26th edition
4. Electrocardiogram, StatPearls - https://www.ncbi.nlm.nih.gov/books/NBK549803/
5. ECG in Medical Practice by ABM Abdullah, 4th edition
6. ECG Basics, http://www.nataliescasebook.com/tag/e-c-g-basics
Lung Cancer: Artificial Intelligence, Synergetics, Complex System Analysis, S...

RESULTS: Overall life span (LS) was 2252.1±1742.5 days and cumulative 5-year survival (5YS) reached 73.2%, 10 years – 64.8%, 20 years – 42.5%. 513 LCP lived more than 5 years (LS=3124.6±1525.6 days), 148 LCP – more than 10 years (LS=5054.4±1504.1 days).199 LCP died because of LC (LS=562.7±374.5 days). 5YS of LCP after bi/lobectomies was significantly superior in comparison with LCP after pneumonectomies (78.1% vs.63.7%, P=0.00001 by log-rank test). AT significantly improved 5YS (66.3% vs. 34.8%) (P=0.00000 by log-rank test) only for LCP with N1-2. Cox modeling displayed that 5YS of LCP significantly depended on: phase transition (PT) early-invasive LC in terms of synergetics, PT N0—N12, cell ratio factors (ratio between cancer cells- CC and blood cells subpopulations), G1-3, histology, glucose, AT, blood cell circuit, prothrombin index, heparin tolerance, recalcification time (P=0.000-0.038). Neural networks, genetic algorithm selection and bootstrap simulation revealed relationships between 5YS and PT early-invasive LC (rank=1), PT N0—N12 (rank=2), thrombocytes/CC (3), erythrocytes/CC (4), eosinophils/CC (5), healthy cells/CC (6), lymphocytes/CC (7), segmented neutrophils/CC (8), stick neutrophils/CC (9), monocytes/CC (10); leucocytes/CC (11). Correct prediction of 5YS was 100% by neural networks computing (area under ROC curve=1.0; error=0.0).
CONCLUSIONS: 5YS of LCP after radical procedures significantly depended on: 1) PT early-invasive cancer; 2) PT N0--N12; 3) cell ratio factors; 4) blood cell circuit; 5) biochemical factors; 6) hemostasis system; 7) AT; 8) LC characteristics; 9) LC cell dynamics; 10) surgery type: lobectomy/pneumonectomy; 11) anthropometric data. Optimal diagnosis and treatment strategies for LC are: 1) screening and early detection of LC; 2) availability of experienced thoracic surgeons because of complexity of radical procedures; 3) aggressive en block surgery and adequate lymph node dissection for completeness; 4) precise prediction; 5) adjuvant chemoimmunoradiotherapy for LCP with unfavorable prognosis.
Couples presenting to the infertility clinic- Do they really have infertility...

Dr Sujoy Dasgupta presented the study on "Couples presenting to the infertility clinic- Do they really have infertility? – The unexplored stories of non-consummation" in the 13th Congress of the Asia Pacific Initiative on Reproduction (ASPIRE 2024) at Manila on 24 May, 2024.
Hemodialysis: Chapter 3, Dialysis Water Unit - Dr.Gawad

- Video recording of this lecture in English language: https://youtu.be/lK81BzxMqdo
- Video recording of this lecture in Arabic language: https://youtu.be/Ve4P0COk9OI
- Link to download the book free: https://nephrotube.blogspot.com/p/nephrotube-nephrology-books.html
- Link to NephroTube website: www.NephroTube.com
- Link to NephroTube social media accounts: https://nephrotube.blogspot.com/p/join-nephrotube-on-social-media.html
Recently uploaded (20)
HOT NEW PRODUCT! BIG SALES FAST SHIPPING NOW FROM CHINA!! EU KU DB BK substit...

HOT NEW PRODUCT! BIG SALES FAST SHIPPING NOW FROM CHINA!! EU KU DB BK substit...
Evaluation of antidepressant activity of clitoris ternatea in animals

Evaluation of antidepressant activity of clitoris ternatea in animals
Charaka Samhita Sutra sthana Chapter 15 Upakalpaniyaadhyaya

Charaka Samhita Sutra sthana Chapter 15 Upakalpaniyaadhyaya
New Directions in Targeted Therapeutic Approaches for Older Adults With Mantl...

New Directions in Targeted Therapeutic Approaches for Older Adults With Mantl...
NVBDCP.pptx Nation vector borne disease control program

NVBDCP.pptx Nation vector borne disease control program
Maxilla, Mandible & Hyoid Bone & Clinical Correlations by Dr. RIG.pptx

Maxilla, Mandible & Hyoid Bone & Clinical Correlations by Dr. RIG.pptx
Lung Cancer: Artificial Intelligence, Synergetics, Complex System Analysis, S...

Lung Cancer: Artificial Intelligence, Synergetics, Complex System Analysis, S...
Couples presenting to the infertility clinic- Do they really have infertility...

Couples presenting to the infertility clinic- Do they really have infertility...
Triangles of Neck and Clinical Correlation by Dr. RIG.pptx

Triangles of Neck and Clinical Correlation by Dr. RIG.pptx
Hemodialysis: Chapter 3, Dialysis Water Unit - Dr.Gawad

Hemodialysis: Chapter 3, Dialysis Water Unit - Dr.Gawad
Aminoglycoside SAR one page.doc
- 1. Chemistry & SAR (Structural activity relationship) of Aminoglycosides Ring I is important for broad spectrum in activity, at the same time it is the major site for enzymatic inactivation by AAC and APH. Amino group at 6΄ and 2΄ are important (Kanamycin B> A>C). 6’amino, 2’amino > 6’amino, 2’hydroxyl > 6’hydroxyl, 2’amino Phosphorylation of 3΄OH will reduce the binding to ribosomes 30S subunit. Methylation at either 6’ carbon or 6’ amino-confers resistance to enzymatic acetylation of the 6’ amino group and doesn’t lower antibacterial activity Removal of 3’ hydroxyl or 4’ hydroxyl - Kanamycin B or dibekacin does’nt reduce antibacterial potency Lack of Oxygen function at 3’ or 4’ along with presence of 4’,5’ double bond Gentamicins, sisomicin and netilamicin No inactivation by phosphotransferase as this enzyme phoshorelates at 3’ OH group Ring II o can accept few structural modifications and retaining the same activity: example is the acetylation of the amino group at position-1 (amikacin). o can be ribose, streptose (five memeberd ring) or streptamine (six memeberd ring). Ring III less sensitive to structural changes but the amino group at position 3΄΄ can be 1° or 2° for maximum activity. RESISTANCE AG inactivating enzymes includes- AAC, APH & ANT AAC-Aminoacyl transferase-acetylates 2’-NH2 of the ring I / 6’NH2 of the ring I/ 3-NH2 of the ring II APH- Phosphotransferase-phosphorylates 3’OH of the ring I/ 2” OH of the ring III and ANT-nucelotidyltransferases- adenylates 2” OH of the ring III /`4’ OH of the ring III or ring I