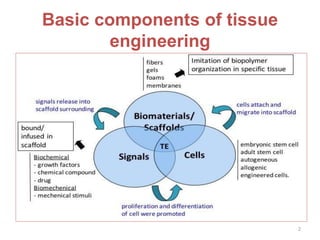
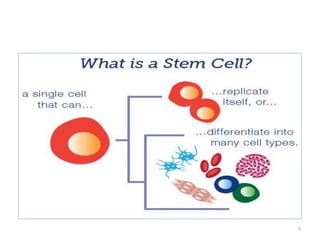
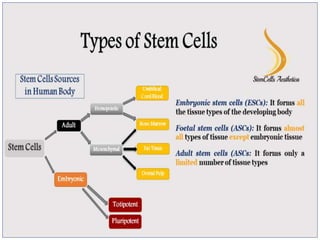
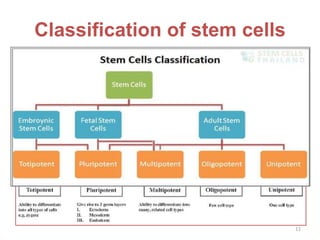
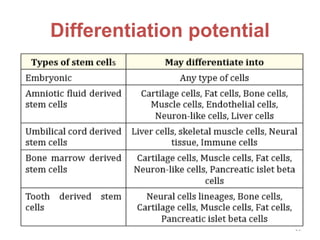
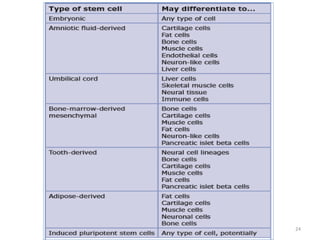
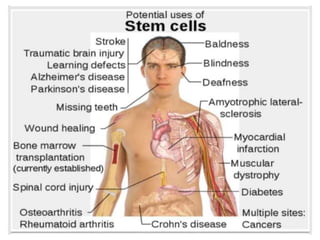
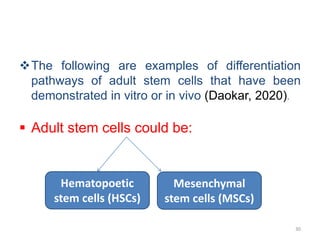
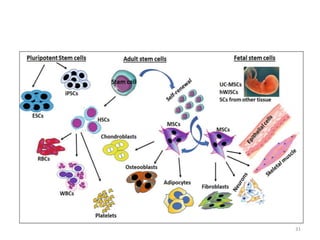
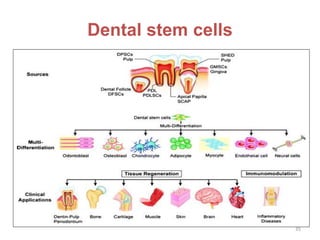
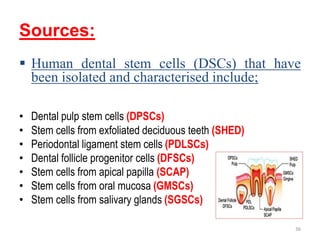
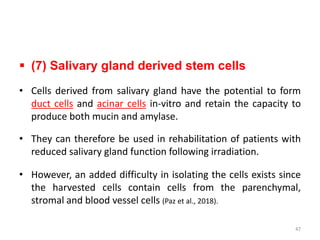
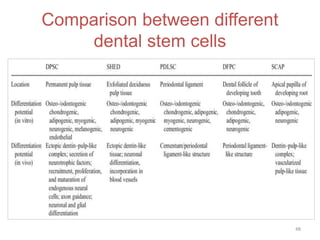
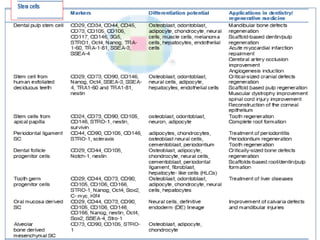
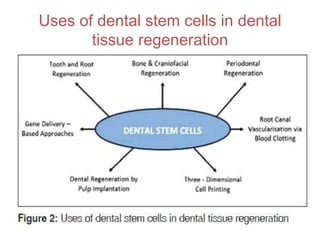
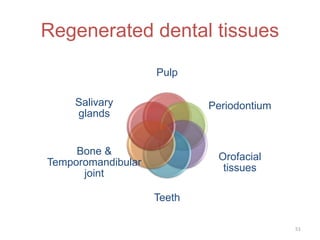
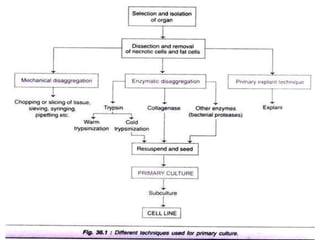
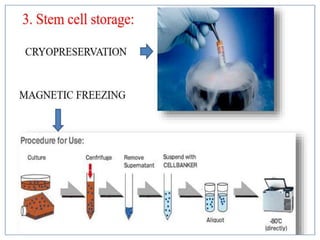
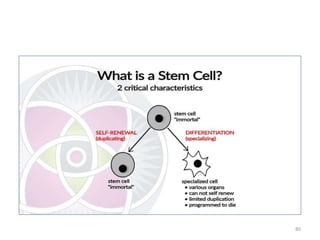
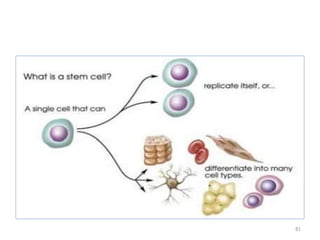
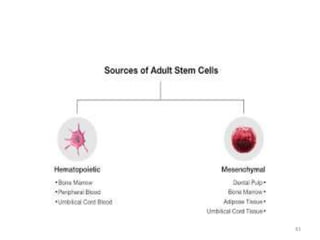
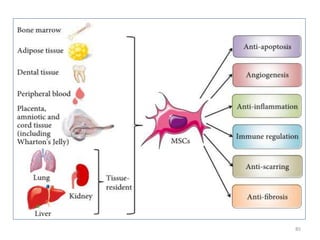
This document discusses stem cells and their applications in dentistry, focusing on adult stem cells. It defines stem cells as undifferentiated cells capable of becoming other cell types. Adult stem cells are found in adult tissues and can differentiate into multiple cell types. Sources of adult stem cells discussed include umbilical cord blood, amniotic fluid, bone marrow, adipose tissue, and dental tissues. Dental stem cells specifically can differentiate into bone, tooth, fat, nerve, and other tissues. Applications of adult stem cells in dentistry include cell-based therapy by direct injection or scaffold seeding for tissue engineering damaged tissues.