This document discusses different concepts of acids and bases including:
1. Arrhenius concept - acids donate H+ ions in water, bases donate OH- ions.
2. Bronsted-Lowry concept - acids donate protons, bases accept protons in both aqueous and non-aqueous solutions.
3. Lewis concept - acids accept electron pairs, bases donate electron pairs.
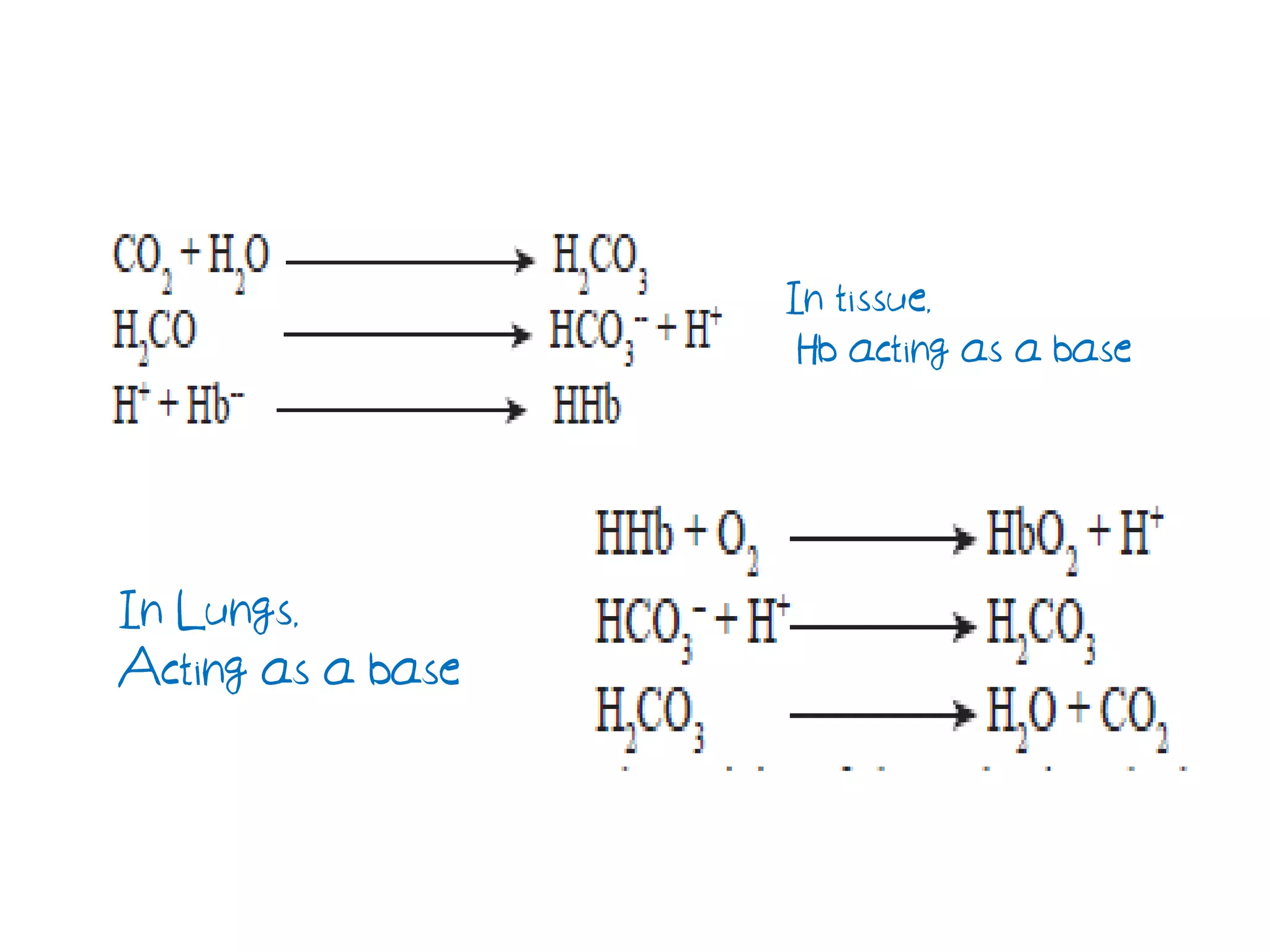
It also covers neutralization reactions between acids and bases to form salts and water. Buffers resist pH changes when acids or bases are added through conjugate acid-base pairs like acetate and bicarbonate buffers. Hemoglobin acts as a buffer through proton acceptance and donation by its histidine side chains.