Periodic table
- 1. THE PERIODIC TABLE BROUGHT TO YOU BY: MARLEE EVANS
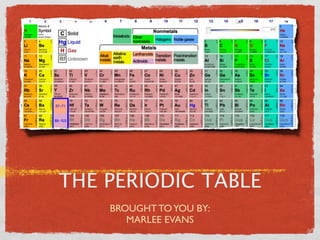
- 2. DEVELOPMENT OF THE PERIODIC TABLE In 1869, Dmitri Mendeleev created the Periodic Table. He put the known elements of his time in order of atomic weight, then grouped them into rows and columns based on their chemical and physical properties.
- 3. ELEMENT ARRANGEMENT ? They are arranged by proton Eight vertical groups share number. similar properties and have Metals are found on the left side the same number of valence of the periodic table and non- electrons. metals are on the right. The elements are also Some elements found between arranged horizontally in metals and non-metals are Periods. The Period an metalloids, which have element is in indicates the properties similar to both number of electron shells metals and non-metals. the atom has.
- 4. ELEMENT RELATIONSHIP? Elements are grouped together by their physical and chemical characteristics.
- 5. ELEMENT RELATIONSHIP BASED Atomic mass increases towards the right. Vertical groups have similar properties. Non-Metals on the left. Metals on the right. Horizontal periods have the same number of atomic orbitals.
- 6. IRIDIUM Atomic number: 77 Element symbol: Ir Atomic Mass: 192.217 Additional Info: Transitional Metal, one of the least abundant elements in the Earth's crust, and is used for hardening platinum.
- 7. ASTATINE Atomic number: 85 Element symbol: At Atomic Mass: 210 Additional Info: Elemental astatine has never been viewed, because a mass large enough to be seen by the naked eye, would be immediately vaporized by the heat generated by its own radioactivity.
- 8. BARIUM Atomic number: 56 Element symbol: Ba Atomic Mass: 137.327 Additional Info: Barium is and Alkaline Earth Metal and is added to steel and cast iron to reduce the size of carbon grains within the microstructure of the metal.
- 9. SOURCES CITED Dayah, Michael. Dynamic Periodic Table. 1 Oct. 1997. Web. 27 Oct 2012 <http:// www.ptable.com>. Helmenstine, Anne M. "Iridium Facts." About.com Chemistry. N.p., n.d. Web. 27 Oct. 2012. <http://chemistry.about.com/od/elementfacts/a/iridium.htm>. http://images.google.com
Editor's Notes
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
- \n
