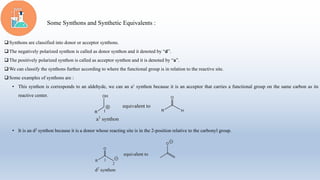
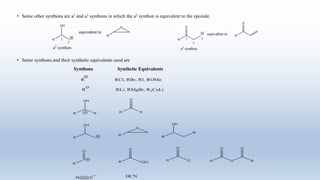
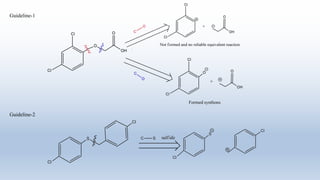
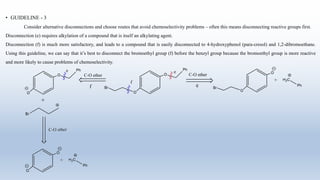
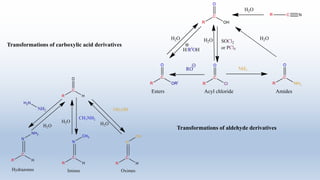
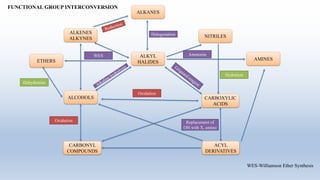
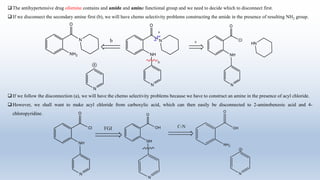
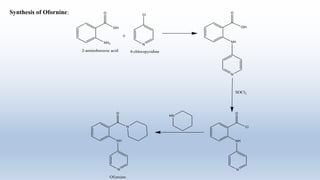
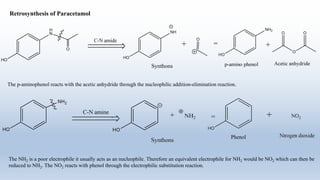
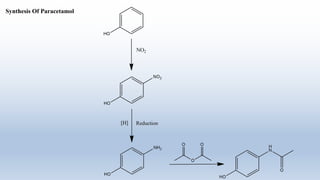
The document provides an introduction to retrosynthesis, outlining its definition as the process of working backward from a target molecule to determine suitable synthetic pathways. It discusses key terminologies related to retrosynthesis, such as target molecule, disconnection, and functional group interconversion, and highlights the importance of various guidelines for effective retrosynthetic analysis. Additionally, it features practical applications of retrosynthesis, including the synthesis of compounds like paracetamol.