Presentation1, radiological imaging of leigh disease.
- 1. Radiological imaging of Leigh disease. Dr/ ABD ALLAH NAZEER. MD.
- 2. Leigh disease, also known as subacute necrotizing encephalomyelopathy (SNEM), is a progressive neurodegenerative disorder and invariably leads to death in childhood. Clinical presentation Typically, symptoms become evident before the age of 2, with the presentation in later childhood (juvenile form) or adulthood (adult form) being uncommon. Symptoms include: psychomotor delay/regression superimposed signs of basal ganglia and brainstem dysfunction ataxia opthalmoplegia dystonia respiratory rhythm disturbance cranial nerve palsies
- 3. Pathology Leigh disease is one of many mitochondrial disorders, due to a broad range of genetic mutations in mitochondrial DNA (mt DNA). As such it is only inherited from the mother, as is the case with other mitochondrial disorders. Some mutations (e.g. SURF 1) are particularly devastating. Chronic energy deprivation leads to histological features such as: spongiform degeneration capillary proliferation demyelination neuronal loss gliosis These findings are similar to those seen in infarction 4. Genetics The inheritance pattern may be either autosomal recessive or X-linked. Markers CSF lactate may be elevated.
- 4. Radiographic features CT: CT demonstrates regions of low-density matching areas of the abnormal T2 signal on MRI. Occasionally some of these areas can show contrast enhancement. MRI: The distribution tends to be symmetrical. T2: characterized by high signal typically in: brainstem periaqueductal gray matter medulla midbrain putamen: characteristic but not always present other sites of T2 signal change include: the remainder of the corpus striatum [globus pallidus and caudate nucleus (heads) subthalamic nuclei substantia nigra thalami involvement of cerebral or cerebellar white matter is unusual T1: usually demonstrates reduced signal in T2 abnormal areas, although some areas of hyperintensity can be seen, as can some enhancement DWI: in the acute setting some restricted diffusion may be evident MR spectroscopy: elevated choline occasionally elevated lactate reduced NAA
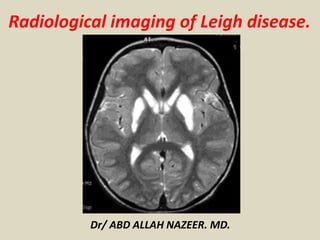
- 5. Symmetric low attenuation in lentiform nuclei in keeping with Leigh Syndrome.
- 6. Symmetric low attenuation in lentiform nuclei in keeping with Leigh Syndrome.
- 7. Computed tomography (CT) and magnetic resonance imaging (MRI) of patients IV7 and IV11. respectively. (A) CT scan of patients IV7 brain at the age of 5 years. The CT scan shows a hypodensity and slight atrophy of the caudate nuclei and the putamen (arrows) and a widening of the frontal horns of the cerebral ventricles. (B-1) Cranial MRI of patient IV11 at the age of 23 years. Prolongation of T2 weighted signals in the residual part of the nucleus caudatus and putamen and (B-2) at the level of the midbrain of the substantia nigra (arrow).
- 8. Axial T1 weighted MR image showing symmetrical hypointense lesions in the putamina.
- 9. A–C, Axial T2-weighted images (3000/120/1 [TR/TE/NEX]) show hyperintense lesions involving the region of the inferior olivary nuclei (arrow in A) and the dorsolateral medulla at the base of the restiform bodies (arrowhead in A), punctuate lesions in the pontine tegmentum (arrowhead in B) and more extensive abnormalities in the cerebellar white matter, and lesions in the periaqueductal area and subthalamic nuclei (arrowhead in C). D and E, Coronal T2-weighted sections (3000/120/1) confirm the presence of lesions in the subthalamic nuclei (arrowhead in D) and show extensive white matter involvement of the cerebellum centered on the dentate nuclei (in E).
- 10. A and B, Axial T2-weighted MR images (3000/120/1) obtained at first examination show hyperintense lesions in the substantia nigra (arrow in A) and medial thalamic nuclei (arrowhead in B). The globi pallidi and white matter, still unmyelinated, are slightly hyperintense, with a tiny, focal hyperintensity in the left pallidum. C, Follow-up MR image (2028/120/2), obtained 2 years and 2 months later, shows lesions in the basal ganglia that involve both the putamina and the head of the left caudate nucleus. A minimal residual right thalamic lesions is visible (arrowhead). Moderate diffuse brain atrophy is present.
- 11. 4 months old child with encephalopathy (Leigh disease): Axial T2-weighted images reveal symmetrical hyperintense lesions involving the substantia nigra(yellow arrow), central tegmental tracts in the pons (red arrow) as well as the cervical cord (Dotted arrow).
- 12. 6 months old child with altered sensorium: A) Coronal T2-weighted image reveals symmetrical hyperintense lesions involving the thalamus(dotted arrow), subthalamic nuclei(black long arrows) and substantia nigra(arrow).B and C) Diffusion-weighted images reveal restricted diffusion in substantia nigra(black small arrow), tegmentum and periaqueductal location(red arrow), basal ganglia (Dotted yellow arrow) and thalamus (Dotted white arrow).
- 13. 6 months old with altered sensorium: A) Axial T2-weighted image reveals symmetrical hyperintense lesions involving the substantia nigra. B-C) Axial T2 and Diffusion-weighted images reveal swollen basal ganglia with hyperintense signal and restricted diffusion.
- 14. Leigh’s Syndrome: MRI Brain Diffusion and Axial T2- weighted images: Bilateral symmetric T2 hyper intensity with faint high signal on diffusion involving putamen and caudate nuclei.
- 15. Leigh's disease: Areas of high signal intensity in putamen bilaterally as well as in the head of the caudate nucleus on T2-weighted images, with sparing of globus pallidus.
- 16. 2 year old with developmental delay shows bilateral, symmetric regions of signal abnormality (yellow arrow) and diffusion restriction (blue) in the basal ganglia concerning for a metabolic disorder such as Leigh Syndrome (LS).
- 17. Leigh Syndrome.
- 18. Leigh disease. Axial T1 W1 images show homogenous hypointensity in bilateral caudate nuclei and anteromedial thalami. Axial T2 WI images show homogenous hyperintensity in bilateral caudate nuclei, lentiform nuclei, antero medial thalami, cerebral peduncles and periaqueductal gray matter. All the above mentioned areas show restricted diffusion.
- 19. Leigh's Disease. Symmetric T2 hyperintensities involving the basal ganglia and brain stem.
- 21. Axial T2-weighted MRI showing progression of the striatal lesions at the ages of nine (A), ten (B), fourteen (C), seventeen (D1&D2) and eighteen (E) years.
- 22. Bilateral symmetric T2 hyper intensity with faint high signal on diffusion involving putamen and caudate nuclei. An upright Doublet of lactate at 1.3ppm on short TE of 35 ms with inversion at long TE of 144 ms on MRS.
- 24. Bilateral symmetrical T2 hyper intensity with restricted diffusion involving Putamen, Paramedian Thalami, Substantia nigra of mid brain, Dentate nuclei of cerebellum and periventricular white matter. An upright Doublets of lactate at 1.3 ppm on short TE of 35 ms and inversion at long TE of 144 ms on MR Spectroscopy.
- 25. Pedigree chart of the family with the Leigh syndrome (LS) and neuroimaging of the proband. (A) Pedigree chart. I and II denote generation number, and 1–2 individual number; the black circle denotes the proband. (B–E) T2-weighted images (T2WI) from magnetic resonance imaging (MRI) of the proband show prolonged signals (arrows) in the (B) basal ganglia and thalamus, (C) midbrain, (D) pons, and (E) medulla oblongata. (F and G) As shown in the magnetic resonance spectra, there is a markedly increased lactate doublet (arrows) in the prolonged signal region in the bilateral basal ganglia. Following one-year treatment, the lesions disappeared in the (H) thalamus, (I) bilateral cerebral peduncle, (J) pons and (K) medulla oblongata, and partially reduced in the (H) basal ganglia and (I) the dorsal midbrain, as shown by T2WI.
- 26. Axial MRI scan (A) and MRS spectrum (B) of a 9-year old boy with Leigh syndrome due to a mutation in the mtDNA
- 27. Layout of the ROIs and the brain MRI (T2WI) in patient 1. ROIs were placed manually in the pons (1), mid brain (2), and bilateral cerebellar hemisphere (3) (at the slice level with the maximum cerebellar hemisphere), thalami (4), basal ganglia (5), and lower temporal (6), lower frontal (7), mid temporal (8), mid frontal (9), occipital (10), upper frontal (11), and parietal cortices (12). The MRI showed nodular high intensity lesions in the bilateral caudate, putamina and thalami on T2WI.
- 28. Results of FDG-PET study in patients with Leigh syndrome. The images of FDG-PET (a–b, e–f, i–j, m–n) and MRI (T2WI) (c–d, g– h, k–l) of patient 1 (a–d), patient 4 (e–h) and patient 2 (i–l) and disease control (m–n) were shown. The glucose up take was reduced in the cerebellum in patients 1 and 4, bilateral basal ganglia in patient 1, 2 and 4, and temporal lobes in patients 1 and 4. The cerebellar hypometabolism was observed even in a patient whose MRI showed no abnormalities (a–d).
- 29. FDG-PET (left), axial T2 MRI (middle) and superimposed PET-MRI images of the brain showing frontal hypermetabolism (A), no detectable uptake in the putamina and reduced uptake in the left caudate (B) and hypometabolism in the cerebellum (C).
- 30. Thank You.
