Presentation1.pptx, radiological imaging of spinal dysraphism.
- 1. Radiological Imaging in Spinal Dysraphism. Dr/ ABD ALLAH NAZEER. MD.
- 2. Spinal dysraphism is a broad term given to a group of anomalies where there are malformations in the dorsum of the embryo. Neural tube defects come under this group as well. Pathology There is often abnormal fusion of the midline embryonic neural, vertebral and mesenchymal structures. Sub types Spinal dysraphism can be broadly divided to into two different pathological entities: open spinal dysraphism meningocoele myelomeningocoele closed spinal dysraphism dorsal dermal sinus lipomyelomeningcoele diastematomyelia neurenteric cyst thickened filum terminale
- 3. Prevalence The estimated incidence of spinal dysraphism is about 1–3/1000 live births. The prevalence of spinal dysraphism has been in decline the world over in the last few decades due to the better nutrition for women, folic acid supplementation, improved antenatal care and high-resolution ultrasound for prenatal screening and biochemical markers. Embryology Embryogenesis in the first 2 months of gestation can be divided into 23 stages. Neural plate is formed in stage 8 around the 18th day, followed by neural folds and their fusion. The expansion of the neural tube and subsequent closure is completed by day 28. Open defects occur when the caudal neuropore fails to close. The secondary neurulation sets the spinal cord formation. Defects at this stage result in occult dysraphism connecting the epidermis and the mesenchymal tissues, leading to variety of anomalies and tethered cord. Failure of primary neurulation leads to open dysraphism posing the risks of CSF leakage and exposure of neural placode. Extent and severity of neurological deficit depends on the degree of malformation of the neural placode and also the level of the defect. The higher the level, usually worse is the prognosis. A spectrum of neurological abnormalities like hydrocephalus, Chiari malformation, syrinx, gyral malformations, skeletal malformations and uro-vesical defects can be associated. In occult dysraphism, the overlying skin is intact but the spinal cord is anchored to various tissues starting from skin, subcutaneous tissue, adipose tissue or cartilage.
- 4. Etiology Spina bifida has a multifactorial causation, involving both genetic and environmental. Recent information stressed on the importance of maternal nutrition and folic acid supplementation which has contributed to its reduction. In India, certain regional factors and consanguinity seem to play a role [hospital-based epidemiological study from NIMHANS (unpublished). Symptomatology Open dysraphism presents with a swelling over the back which is noticed at birth. Symptoms are primarily referable to CSF leak or the exposed spinal cord. Since the skin over the swelling is poorly developed, it usually gives way during labor, resulting in CSF leak, contamination and meningitis. Defects predominantly involve thoracolumbar, lumbosacral, lumbar, thoracic, sacral and cervical in the order of frequency of occurrence. Incidence of high cervical lesions is about 3.9%. Neurological deficits include motor, sensory and sphincter dysfunction, depending upon the severity and level. In severe cases, hypotonic areflexic limbs, sphincter atonia with rectal prolapse may be associated. Chiari malformation presents with lower brainstem and lower cranial nerve dysfunction. The presence of large head usually indicates hydrocephalus. The associated skeletal abnormalities are kyphosis, scoliosis, and deformities of the long bones and feet, hemivertebrae, defective ribs.
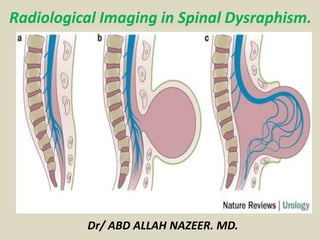
- 5. Classification of spinal dysraphism.
- 7. Radiography With spinal dysraphism, radiographs may show structural vertebral anomalies such as hemivertebra, butterfly vertebra, or incomplete fusion of posterior elements; it does not allow imaging of the spinal cord. Radiographs of the vertebrae provide information for early evaluation of infants born with myelomeningocele. Congenital spinal deformities need to be tracked closely. Paralytic spinal deformities require imaging based on clinical examination findings; these deformities should be followed up frequently during times of rapid growth. Plain radiographs of patients with myelomeningocele demonstrate incomplete fusion of posterior elements and increased interpedicular distance.
- 8. Posteroanterior (PA) chest radiograph shows defects of the laminae of the lower cervical spine. Plain abdominal radiograph in the same patient as in the previous image shows spina bifida occulta of S1.
- 9. Myelograms in a 5-year-old patient show the dorsal region of the spine and an anterior thoracic meningocele. Note the gross dorsal kyphosis.
- 10. Myelograms in a 4-year-old patient show the lumbosacral region; a long, tethered cord; and diastematomyelia. Right, plain radiograph of the lumbar spine shows diastematomyelia. Left, myelogram in the same patient shows a filling defect at the level of diastematomyelia.
- 11. Left, plain anteroposterior (AP) radiograph of the lumbar spine shows spina bifida occulta. Right, myelogram of the same patient shows a thick tethered cord Left, anteroposterior (AP) plain radiograph of the lumbar spine shows a defect within the laminae of S1 and S2. Right, myelograms in the same patient show a markedly thickened, low tethered cord.
- 12. Plain anteroposterior (AP) lumbar spinal radiograph in a 7-year-old patient shows a defect within the laminae of L4-5 and S1. Note the diastematomyelia. Myelograms in the same patient as in the previous image show a low, tethered cord. Note also the diastematomyelia.
- 13. Left, plain radiograph of the lumbar spine shows bony defects in the laminae of L2 to S1. Right, myelogram shows a split cord. Plain radiographs show posterior scalloping.
- 14. Ultrasonongraphy: Spinal sonography is usually not possible after the age of 6 months except in cases of a persistent posterior spinal defect; in such cases, sonography may be performed at any age In the neonate, the cord appears as a tubular hypoechoic structure with hyperechoic walls. The central canal is hyperechoic. The cord position is dependent. The subarachnoid space around the cord is echo poor. In the neonate, the conus medullaris is smooth and tapering and lies above the middle of L2; the range varies from D10 to L2-L3. The cauda equina is seen as echogenic lines surrounding a hyperechoic filum terminale with dependent positioning. The normal filum terminale is 1.0-1.5 mm in diameter. The vertebral bodies are seen as echogenic segmental structures lying anterior to the spinal cord. In the normal infant, the cord lies one third to one half the distance between the anterior and posterior walls of the spinal canal. There is normal pulsatile movement of the cord. When the cord is viewed axially, it appears round to oval and is surrounded by the fluid-filled subarachnoid space. The cord is fixed within the spinal canal by the dentate ligaments, which pass laterally from the cord. Below L2, the echogenic nerve roots are identified with a vertical or oblique orientation
- 15. The spinal cord may be depicted throughout its length, allowing visualization of the conus and free movement of the nerve roots. An absence of normal transmitted pulsations and a lack of free movement of nerve roots on sonograms suggest a tethered cord. In cases in which there is a low tethered cord, the conus is low and the spinal cord is displaced dorsally. There is lack of normal cord pulsatility, and the filum terminale is thickened to over 2 mm. The thickened filum terminale may be fibrous or lipomatous. An abnormal cord may lie in a dorsal position rather than being dependent. The clinical significance of a low cord without tethering is unknown. In a newborn term infant, the normal conus usually lies above the level of the mid L2. In cases in which there is a skin-covered back mass, the contents of the mass may be characterized. Axial spinal sonograms readily show the 2 hemicords and the echogenic spur in cases of diastematomyelia. In cases of diastematomyelia, sonography may show the spur. A dorsal dermal sinus may be depicted as an echogenic tract deep to a hole in the skin. It may be difficult to be confident in the sonographic findings if the tract communicates with the spinal canal. However, a low-lying cord suggests tethering by intraspinal extension of the sinus; sonograms may show abnormal echogenicity at the depth of the tract, suggesting lipoma or dermoid. Alternatively, images may show matted nerve roots, caused by arachnoiditis. No conus may be identified if the cord terminates in a lipoma.
- 16. Antenatal ultrasonogram shows a lumbar meningocele.
- 17. Antenatal ultra sonogram shows a lemon sign and a banana sign.
- 18. Computed Tomography CT myelography demonstrates splitting of the cord and, in some cases, splitting of the meningeal sheath. In addition, other bony anomalies, such as an intervertebral septum, and aberrant fibroneural bands may be depicted. CT myelography allows better definition of cord expansion or deformity than can be achieved by conventional myelography. In the case of intrinsic cord tumors, repeat CT after 24 hours reveals intramedullary contrast enhancement if associated syringomyelia is present. MRI is the imaging procedure of choice. Spinal lipomas, with their fatty tissue contents, are identifiable both on CT scans and on MRIs. On CT scans, fatty tissue has a strongly hypoattenuating appearance that may best be appreciated in comparison with CSF on soft tissue windows and in comparison with air on bone windows. On MRIs, fatty tissue is strongly hyperintense on images obtained with both short and long repetition times. Newer techniques of fat suppression, such as short-tau inversion recovery (STIR) imaging, may resolve any doubts. The 2 techniques are complementary: On the one hand, CT better shows osseous abnormalities associated with the lipomas; on the other, MRI is preferred because it allows better depiction of detail and contrast resolution of soft tissues.
- 19. Axial CT scans through the lumbar spine with bone window setting in the same patient as in the previous images show a bony bar due to diastematomyelia.
- 20. Axial CT scans through the upper lumbar spine show a split cord.
- 21. Axial CT scans through the lumbosacral junction shows absence of the posterior spinal elements at L5-S1. Note sclerosis of the laminae and the wide spinal canal.
- 22. Spine Bifida
- 23. Dermal sinus.
- 24. T1- and T2-weighted sagittal MRIs of the cervical and dorsal spine in the same patient as in the previous 3 images show evidence of previous surgery for cervical meningocele. Note the associated congenital fusion of C5 and C6. Axial T2-weighted MRIs of the cervical spine in the same patient as in the previous 4 images show a large spinal canal, evidence of previous surgery, and a split cord.
- 26. Sagittal MRIs of the lumbar spine show diastematomyelia. Note the congenital fusion of L1 and L2. Axial MRIs in the same patient as in the previous image shows a hypo intense bar, which is in an anteroposterior location because of diastematomyelia that splits the cord.
- 27. T1- and T2-weighted sagittal MRIs of the lumbar spine show an extradural spinal lipoma communicating with the subcutaneous fat.
- 28. T1- and T2-weighted sagittal MRIs of the lumbar spine show an intradural sacral lipoma. Note the scalloping of the posterior sacral vertebral bodies and the syrinx.
- 29. Terminal lipoma
- 30. Meningocele.
- 31. Tethered cord
- 32. Tethered cord.
- 35. MRI sagittal T2 weighted image showing myelocystocele with hydromyelia in mid dorsal area with SCM Type1 at D7-8, D10, L2-3.
- 37. Axial T1-weighted (A) and T2-weighted (B) MRI scans show a similar depiction as sonography. Arrows point to the deformed and displaced spinal cord. M = meningocele; S = syringocele; Vb = vertebral body.
- 38. Congenital spinal dysraphism with meningomyelocele and lipomas.
- 39. (A-C) and corresponding occult spinal dysraphism detected by sagittal, T1-weighted magnetic resonance imaging (MRI) studies of the spinal cord (D-G). A, Sacral lipoma and deviated gluteal furrow (DGF); B, lumbar port-wine stain, lipoma, dermal sinus, and DGF; and C, dorsal and lumbar unclassified hamartomas. D, Lipoma of the conus (arrow); E, dermal sinus (arrow); F, top of the lipoma of the filum terminale (upper arrow) and fistula (lower arrow); G, multiple lipomas of the thoracic cord (upper arrow) and posterior conus (lower arrow).
- 40. T2-weighted sagittal (a) and coronal (b) magnetic resonance imaging of dorso-lumbar spine shows a hemivertebrae from D10 to D12 level along with a bony spur at D11. Tethering of cord and meningomyelocele is also visible. Continuation of left hemicord is appreciable in coronal view (b)
- 41. T2 weighted axial image shows a bony spur dividing the cord into two halves (a and b). Herniation of right hemicord, meninges and cerebrospinal fluid is noted through the defect in posterior element, forming a large hemi-meningomyelocele (c). Two hemicords are visible below the level of herniation (d) which reunites at D12 level to form a single cord(e).
- 42. Sagittal MRI sequences (a) T1-weighted, (b) T2-weighted, (c) MR myelography, and (d) 3D-CISS sequence; arrow points to the fluid containing hydromyelic cord herniating into the meningocele sac
- 43. Sagittal T2-weighted image with fat saturation (A), T1-weighted image (B), and contrast-enhanced T1- weighted image (C) of L-spine MRI show hypoplastic S2 vertebra with agenesis of lower sacrum and coccyx. The spinal cord terminates above L1 with club-shaped (chisel-shaped), blunted and angulated caudal end. Separation of the anterior and posterior spinal roots of the cauda equina is also noted. Distended urinary bladder with coarse trabeculation pattern is noted due to neurogenic bladder. Coronal T2-weighted image (D) of L-spine MRI shows both iliac bones articulated with S1. MRI = magnetic resonance imaging.
- 44. MRI Sagittal T2w images of lumbo sacral spine shows: Hypoplastic S1 and S2 with failure of formation of S3 and onwards. Cord ending at a higher level at D12-L1 with 'wedge' shaped termination. No cord tethering. No terminal cord cyst or syrinx. Imaging diagnosis : Caudal Regression, Group I.
- 45. Axial T2-weighted MR images demonstrating spina bifida occulta (left) with failure of fusion of the posterior elements at the midline, and the lipoma- placode interface (right, arrow). (left) and T2-weighted (right) MR images demonstrating a lipomyelomeningocele. Note the lipomatous component extending in the intradural and epidural spaces (arrows) as well as the subcutaneous space.
- 50. Thank You.
