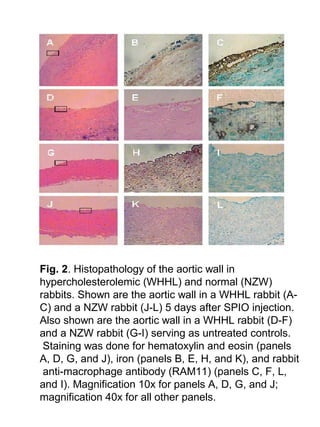
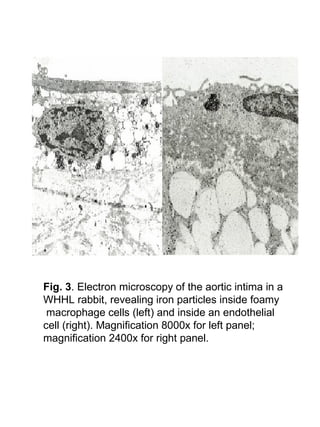
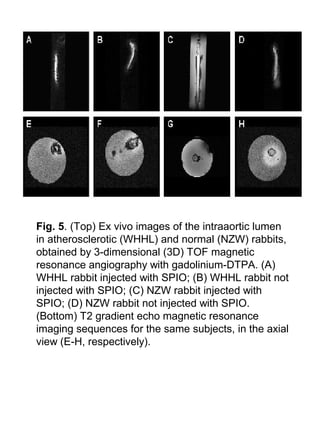
This study aimed to detect vulnerable atherosclerotic plaques using superparamagnetic iron oxide (SPIO) nanoparticles and MRI. The researchers found that SPIO was taken up more by inflamed plaques in atherosclerotic rabbits compared to normal arterial walls, as SPIO is taken up more by macrophages. MRI revealed decreased aortic wall signal intensity in atherosclerotic rabbits after SPIO injection. This preliminary study suggests SPIO-enhanced MRI may help detect vulnerable plaques by targeting plaque inflammation.






![In Vivo:
Using a 1.5T MRI system (Signa,
General Electric) equipped with a
conventional extremity coil, baseline MRI
of the aorta was done in 4 WHHL and 2
NZW rabbits (T2 gradient echo: TR =
1200 msec, TE = 6 msec, FOV = 16 x 12
cm, matrix size = 256 x 192 pixels; 3-
dimensional magnetic resonance [3D
MR] angiography with gadolinium-DTPA:
TE = 1.3 msec, TR = 5.6 msec). The
rabbits were injected with SPIO (2 mmol
Fe/kg) IV via an ear vein. Post-contrast
MRI was performed on day 5 using the
same MRI sequences. MRI was done
with respiratory and cardiac gating. The
rabbits were anesthetized with isoflurane
for the duration of their studies.](https://image.slidesharecdn.com/lctbqau4tvkag3peyygb-signature-185c9c8f74195a69485b096cbf5bfb73f6b3ed8b7754452e736ee4598185e9e3-poli-160919191610/85/Plaque-inflammation-july-2-7-320.jpg)