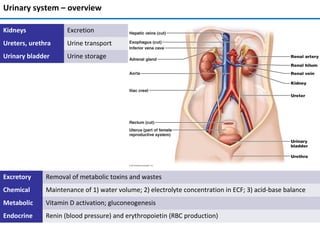
Renal system
- 1. Urinary system – overview Excretory Removal of metabolic toxins and wastes Chemical Maintenance of 1) water volume; 2) electrolyte concentration in ECF; 3) acid-base balance Metabolic Vitamin D activation; gluconeogenesis Endocrine Renin (blood pressure) and erythropoietin (RBC production) Kidneys Excretion Ureters, urethra Urine transport Urinary bladder Urine storage
- 2. Kidney anatomy • Retroperitoneal, superior lumbar region; ~ T12 to L5 • Right is lower; adrenal gland atop; bean-shape; medial side – renal hilum • Ureters, renal blood vessels, lymphatics, and nerves enter and exit at hilum • Surrounded by (from superficial to deep): renal fascia, perirenal fat capsule, fibrous capsule • Layers: cortex and medulla • Renal pyramids (medulla) produce urine; separated by renal columns (cortex) • Urine from papilla goes to minor calyx → major calyx → renal pelvis → ureter • Pyramid and surrounding columns = lobe Renal anatomy
- 3. Blood and nerve supply • Kidneys cleanse blood rich blood supply • Renal arteries – ~ ¼ (1200 ml) of cardiac output • Similar paths for arterial and venous flow • Nerve supply – sympathetic fibers from renal plexus Renal anatomy
- 4. Nephron • Structural and functional units that form urine; > 1 million per kidney • Filtration, reabsorption, and secretion (see bottom left) • Renal corpuscle (RC): glomerulus and glomerular capsule • Glomerulus (G): fenestrated capillaries filtrate • Like plasma but without proteins and cells; goes into glomerular capsule • Renal tubule: proximal convoluted tubule (PCT), nephron loop, distal convoluted tubule (DCT); reabsorption and secretion • DCT enters collecting duct (not the part of a nephron) Blood/ECF Filtrate Filtration (RC) Reabsorption (RT) Secretion (RT) Renal anatomy
- 5. Urine formation • 180 L fluid processed daily (60x entire plasma volume) → only 1.5 L urine • 20-25% oxygen consumption Three processes: • Glomerular filtration • Cell- and protein-free filtrate of blood plasma minus proteins • Tubular reabsorption • Selectively returns 99% of substances from filtrate to blood in renal tubules and collecting ducts • Tubular secretion • Selectively moves substances from blood to filtrate in renal tubules and collecting ducts • Eventually urine is produced → <1% of filtrate • Contains metabolic wastes and unneeded substances Renal physiology
- 6. Nephron epithelia Part Type Function and features Glomerular capsule (parietal) Simple squamous Structural Glomerular capsule (visceral) Branching cuboidal Filtration via filtration slits; located around capillaries PCT Cuboidal with microvilli ↑ surface area; Reabsorption and secretion; only in cortex NL (thick descending limb Cuboidal with microvilli ↑ surface area; reabsorption NL (thin descending limb) Simple squamous Reabsorption NL (thick ascending limb) Cuboidal to columnar Reabsorption DCT Cuboidal; few microvilli Secretion > reabsorption; only cortex Collecting duct Principal and intercalated cells – cuboidal Principal – sparse, short microvilli; H2O and Na+ balance Intercalated (A and B) – many microvilli; acid-base balance
- 7. Types of nephrons • Cortical – 85% of nephrons; in cortex • Glomerular capillaries: filtration • Peritubular capillaries: reabsorption and secretion • Juxtamedullary: long nephron loops; in the medulla • Glomerulus: filtration; capillaries are the same as in cortical N. • Vasa recta and nephron loop form countercurrent system; together with the collecting duct produce concentrated urine
- 8. Nephron capillary beds Capillary bed Blood flow Blood pressure Location Structure Function Glomerular afferent a. → glomerulus → efferent a. High (Ø of afferent a. > Ø of efferent a.) Glomerulus Fenestrated, surrounded by podocytes Filtration Peritubular efferent a. → capillaries → venules Low Around renal tubule Fenestrated, thin-walled Reabsorption, secretion Vasa recta efferent a. → capillaries → venules Low Around nephron loop Fenestrated, thin-walled Formation of concentrated urine Renal physiology
- 9. Filtration pressures • Passive process; no energy required; from blood to capsule via filtration membrane; no reabsorption • Capillary hydrostatic pressure pushes fluid into the filtrate; equals BP • 55 mm Hg; due to the larger afferent a. diameter • Capsular hydrostatic pressure pushes fluid back into the blood; 15 mm Hg • Capillary osmotic pressure pulls fluid back into the blood; due to the plasma proteins; 30 mm Hg • Sum of pressures → net filtration pressure (NFP) = 10 mm Hg (55 one way – 45 other way) • Determines glomerular filtration rate • Why there is no capsular osmotic pressure? Glomerular filtration
- 10. Filtration mechanism • Filtration membrane: fenestrated capillary endothelium; basement membrane; foot processes of podocytes with filtration slits • Molecules smaller than 3 nm pass (water, glucose, amino acids, nitrogenous wastes) – everything but proteins and large biological macromolecules • Macromolecules "stuck" in filtration membrane engulfed by glomerular mesangial cells • Plasma proteins remain in blood maintain osmotic pressure prevents loss of all water to filtrate • Proteins in filtrate indicate membrane problem Glomerular filtration
- 11. Glomerular filtration rate (GFR) • Volume of filtrate formed per minute by both kidneys (normal = 120–125 ml/min) • GFR NFP, filtration membrane surface≡ (controlled by mesangial cells) and permeability (very high) • Constant GFR = normal filtration → ECF homeostasis • GFR affects BP: ↑BP → ↑GFR ↑urine output ↓BP, and vice versa • Goal of intrinsic controls – maintain GFR in kidney, not BP • Goal of extrinsic controls – maintain systemic BP • Intrinsic controls: local within kidney; maintain GFR; dominant at normal BP • Extrinsic controls: nervous, endocrine; maintain BP; can ↓ kidney function • Preside over intrinsic if systemic BP < 80 or > 180 mm Hg • Controlled via glomerular hydrostatic pressure • If rises NFP rises GFR rises; 18% ↓ of systemic BP → GFR = 0 NFP Membrane permeability Surface area GFR BP increases GFR increases Urine output increases Blood volume decreases BP decreases Glomerular filtration GFR ↑ Glomerular blood flow ↓ GFR ↓ INTRINSIC EXTRINSIC
- 12. Juxtaglomerular complex (JGC) • One per nephron; portions of: 1) ascending limb of nephron loop; afferent and efferent a. • Regulates GFR and BP • Macula densa cells (MDC): in ascending limb; sense NaCl in the filtrate • Granular/juxtaglomerular cells (JC): arteriolar SMC; dilate/constrict afferent a. • Respond to ↓ stretch of afferent a. releasing renin (BP regulation) • Extraglomerular mesangial cells (EMC): connected by gap junctions • Signaling between MDC and JC Glomerular filtration
- 13. Intrinsic controls of GFR • Myogenic mechanism • Smooth muscle contracts in response to stretch • Both help maintain normal GFR despite normal fluctuations in blood pressure • Protects glomeruli from ↑ BP • Tubuloglomerular feedback mechanism • Directed by macula densa cells; respond to filtrate NaCl concentration • Opposite for ↓ GFR BP GFR ↑ Stretch↑ GFR ↓ Stretch ↓ Afferent a. constrict Efferent a. dilate Afferent a. dilate Efferent a. constrict Blood flow ↓ Blood flow ↑ GFR ↓ GFR ↑ ↑ ↓ GFR ↑ Filtrate flow ↑ Reabsorption time ↓ Filtrate NaCl ↑ Afferent a. constrict GFR ↓ Reabsorption time ↑ Filtrate NaCl ↓ Glomerular filtration
- 14. Extrinsic controls: BP regulation BP ↓ Sympathetic response Adrenal medulla Juxtaglomerular cells Macula densa cells Kidneys Angiotensin Aldosterone Na/water retention BP ↑ Afferent a. constriction ↓ urine Reduced stretch BV ↑ Norepinephrine Vasoconstriction Epinephrine Filtrate NaCl ↓ Adenosine Prostaglandin E2 Renin 1 1,2 2 • Under normal conditions blood vessels dilated, intrinsic controls prevail • Three major mechanisms of BP regulation: 1) direct sympathetic stimulation; 2) renin-angiotensin- aldosterone cascade; 3) kidney paracrine/endocrine signaling Glomerular filtration
- 15. Transendothelial process • Most but not all of chemicals in filtrate are reabsorbed (selective process) • Organic nutrients (sugars, aminoacids) reabsorbed ≈completely; ions/water hormonally regulated • Active and passive transport via transcellular or paracellular routes • Transcellular: apical membrane → cytoplasm → basolateral membrane → ECF → peritubular capillaries • Paracellular: between cells, controlled by tight junctions Tubular reabsorption
- 16. Roles for 1° and 2° active transport • Requires ATP; 1° transport of Na+ at basal membrane → ↓ intracellular Na+ , (-) intracellular potential → Na+ diffusion via brush border • Na+ diffusion via brush border → coupled with 2° active transport → reabsorption of glucose (sodium-glucose co- transporter, SGLT), AA, H+ (sodium-hydrogen exchanger, NHE) etc. • Na+ reabsorption → H2O reabsorption by osmosis → ↓ chemical concentrations in filtrate → concentration gradient → passive reabsorption of Cl- , urea, lipid-soluble substances Tubular reabsorption
- 17. PCT Chemical Apical membrane Basolateral membrane Na+ Facilitated diffusion 1° AT (Na+ /K+ pump) Water Osmosis (obligate water reabsorption via aquaporins) K+ , Cl- , Ca2+ , Mg2+ , HCO3 – , PO4 3- 2° AT (Na+ symport), facilitated diffusion (Mg2+ ) Facilitated diffusion, paracellular diffusion (K+ , Ca2+ , Mg2+ ) AA, glucose, fructose, galactose, lactate, succinate, citrate 2° AT (Na+ symport) Facilitated diffusion • All nutrients, e.g., glucose and amino acids • 65% of Na+ and H2O; other ions • ~ all uric acid; ½ urea (later secreted back into filtrate)
- 18. Nephron loop Thin descending limb Water Osmosis (obligate reabsorption via aquaporins) Thick ascending limb Chemical Apical membrane Basolateral membrane Na+ Facilitated diffusion 1° AT (Na+ /K+ pump), paracellular diffusion K+ , Cl- , Ca2+ , Mg2+ 2° AT (Na+ cotransporter, except Mg2+ ) Paracellular diffusion ( K+ , Ca2+ , Mg2+ ) • Descending thin limb: H2O (main function), also Na+ and urea • Ascending thick limb: ions (Na+, Ca2+ , Mg2+ , HCO3 - etc) • Ascending thick limb is impermeable to water • Diluted tubular fluid at the end of the ascending limb • Allows kidneys to regulate urine concentration • Loop diuretics inhibit (1-Na+ , 2-Cl- , 1-K+ ) cotransporter Tubular reabsorption
- 19. DCT Chemical Apical membrane Basolateral membrane Na+ 2° AT (Na+ /Cl- cotransporter) 1° AT (Na+ /K+ pump), paracellular diffusion Cl- 2° AT (Na+ /Cl- cotransporter) Facilitated diffusion HCO3 - Forms intracellularly HCO3 - /Cl- cotransporter • Early DCT: ions (Na+ , K+ , Cl- etc.); no water or urea • Thiazide diuretics inhibit Na+ /Cl- symport • Late DCT: Na+ reabsorption, K+ excretion (aldosterone) • K+ -sparing diuretics: • Aldosterone antagonists XXX A. receptor • Na+ channel blockers indirectly XXX Na+ /K+ pump • DCT is important for acid- base balance Tubular reabsorption
- 20. Collecting duct. Summary Ion Apical membrane Basolateral membrane Na+ 2° AT (Na+ /Cl- cotransporter) 1° AT (Na+ /K+ pump), paracellular diffusion Cl- 2° AT (Na+ /Cl- cotransporter) Facilitated diffusion HCO3 - Forms intracellularly HCO3 - /Cl- cotransporter • Permeable to H2O via aquaporins (number is regulated by ADH) • Permeable to urea (diffuses into medullary interstitium); helps to regulate urine concentration • H+ secretion → acid-base balance regulation Summary of tubular reabsorption (<1 – net reabsorption, >1 – net secretion; H2O – 1) Tubular reabsorption
- 21. Transport maximum • Transport maximum (Tm) for ~ many reabsorbed substances (see below) • Reflects number of carriers/pumps in the membranes of tubular cells • When carriers saturated, excess excreted in urine • E.g., hyperglycemia filtered load is higher than reabsorption → exceed Tm glucose in urine • Some – gradient-time transport (Vd = D*(C1-C2); Vd – diffusion rate; D = diffusion coefficient) • Example: Na+ in the PCT Tubular reabsorption
- 22. Summary and regulation (3) (1) (2) (4) (5) 1 2 3 4 5 Adrenal gland Liver Posterior pituitary gland Heart Parathyroid gland Some compounds Regulation Tubular reabsorption
- 23. Functions and features • From ECF to tubular fluid; almost all in PCT • K+ , H+, NH4 + , creatinine, organic acids and bases, drugs, urea, uric acid • Some are from tubular cells, e.g. HCO3 - • Disposes of substances (e.g., drugs) bound to plasma proteins • Disposing of undesirable passively reabsorbed substances (e.g., urea and uric acid) • Disposing of excess K+ (aldosterone) • Acid-base regulation by secreting H+ or HCO3 – in urine Location Compounds secreted PCT Bile salts, oxalate, urate, catecholamines, drugs (e.g., penicillin), salicylates, H+, NH 4 + Nephron loop Urea (thin ascending limb), H+ (thick ascending limb) DCT K+ (aldosterone-regulated), H+ Collecting duct H+ Tubular secretion
- 24. Urine concentration and volume • Osmolality: number of solute particles in 1 kg of H2O; in milliosmols (mOsm); ability to cause osmosis • Osmolality of plasma – ~300 mOsm via regulating urine concentration and volume • Countercurrent mechanism: fluid flows in opposite directions in two adjacent segments of same tube with hair pin turn • Countercurrent multiplier – filtrate flow in the nephron loops of juxtamedullary nephrons • Countercurrent exchanger - blood flow in vasa recta • Establish and maintain osmotic gradient (300 mOsm to 1200 mOsm) from renal cortex through medulla → allows to vary urine concentration Osmolality. Countercurrent mechanism
- 25. Countercurrent multiplier: nephron loop • Descending limb: freely permeable to H2O; medullary IF is hyperosmotic → H2O from filtrate to medullary IF → filtrate osmolality ↑ to ~1200 mOsm • Ascending limb: impermeable to H2O, selectively permeable to solutes → Na+ , Cl– reabsorbed → filtrate osmolality ↓ to 100 mOsm • Constant 200 mOsm difference between two limbs of nephron loop and between ascending limb and IF • Difference "multiplied" along length of loop to ~ 900 mOsm Urine concentration and volume
- 26. Countercurrent exchanger: vasa recta (VR) • Ascending vasa recta remove reabsorbed H2O from 1) descending VR; 2) nephron loop (descending limb); 3) collecting duct; → volume of blood at end of vasa recta higher than at beginning • NaCl: diffuses from concentrated IF into descending VR → goes into ascending VR → diffuses from ascending VR into diluted IF → preservation of medullary gradient by recycling NaCl • Gradient allows H2O reabsorption from collecting duct if aquaporins are present (regulated by ADH) Urine concentration and volume
- 27. ADH regulates urine concentration Urine concentration and volume
- 28. Urea helps to maintain medullary osmotic gradient • Descending limb: water reabsorption → urea in filtrate ↑ • Ascending thin limb: passive diffusion of urea from the IF, water reabsorption → urea in filtrate ↑ • Ascending thick limb: no water or urea transport • Cortical collecting duct: water reabsorption reabsorbs water → urea in filtrate ↑ • Medullary collecting duct: high filtrate urea diffusion into medullary IF high osmolality in medulla Urine concentration and volume
- 29. Diuretics increase urinary output Acetazolamide (1) PCT XXX carbonic anhydrase; ↓ blood pH, excretion of HCO3 - , Na+ ; H2O follows Osmotic d. (2) PCT, DL of NL E.g., mannitol; not reabsorbed, ↑ filtrate osmolality; prevent H2O reabsorption Loop d. (3) AL of NL Inhibit 1-Na+ , 2-Cl- , 1-K+ cotransporter → ↑ Na+ excretion → H2O follows Thiazid d. (4) DCT Inhibit Na+ /Cl- symport → ↑ Na+ excretion → H2O follows K+ -sparing d. (5) DCT, CD Aldosterone inhibitors → ↑ Na+ excretion → H2O follows, K+ stays (sparing) Na+ channel inhibitors DCT, CD Indirectly inhibit Na+ /K+ basolateral pump → ↑ Na+ excretion → H2O follows ADH inhibitors CD Decrease water reabsorption → ↑ H2Oexcretion Urine concentration and volume
- 30. Evaluation of kidney function. Renal clearance • Kidney function is to filter out from/reabsorb chemicals in the blood • To determine, one has to know both plasma and urine concentrations • Renal clearance of a substance is the volume of plasma that kidneys clear of this substance in a given time • Used to determine GFR; helps to detect glomerular damage • C = UV/P • Measured using Inulin (plant polysaccharide) • Freely filtered; neither reabsorbed nor secreted by kidneys; its renal clearance = GFR = 125 ml/min C renal clearance rate (ml/min) U concentration (mg/ml) of substance in urine V flow rate of urine formation (ml/min) P concentration of same substance in plasma If C… Substance is… = 0 completely reabsorbed or not filtered = 125 ml/min no net reabsorption/secretion; freely filtered < 125 ml/min reabsorbed > 125 ml/min secreted Clinical evaluation
- 31. Chronic kidney disease • Several stages with varying degrees of ↓ GFR; chronic renal disease: < 60 ml/min for 3 months • May be caused by diabetes mellitus, hypertension, damage to kidneys after infection • Renal failure: GFR < 15 ml/min; leads to uremia – ↑ urea in blood, ionic and hormonal imbalances; metabolic abnormalities • Treated with hemodialysis or transplant Clinical evaluation
- 32. Control of water balance Electrolyte balance • In = out = ~ 2500 ml/day • Obligatory water losses: urine (60%), skin and lungs, perspiration, and feces (2.5 L/day) • Water intake: beverages, food, metabolic water (2.5 L/day) • Regulated vi ADH (hypothalamus); ↑ osmolality → ↑ thirst • Dehydration (diarrhea, burns, fever etc.) also increases ADH
- 33. Dehydration •Hemorrhage, severe burns, vomiting, diarrhea, sweating, water deprivation, diuretics, endocrine diseases •Signs and symptoms: "cottony" oral mucosa, thirst, dry flushed skin, oliguria •Weight loss, fever, mental confusion, hypovolemic shock, and loss of electrolytes Hypotonic hydration •Renal insufficiency or overhydration •ECF osmolality ↓ → hyponatremia → net osmosis into tissue cells → swelling of cells → nausea, vomiting, muscular cramping, cerebral edema → death •Treated with hypertonic saline Edema •↑ fluid out of blood: ↑ HPC (incompetent venous valves, localized blood vessel blockage, congestive heart failure, ↑ blood volume) and permeability (inflammatory response) •↓ fluid into blood: ↓OPC (hypoproteinemia), malnutrition, liver disease, glomerulonephritis Control of water balance Electrolyte balance • Blocked (or surgically removed) lymphatic vessels: proteins accumulate in IF, ↑ OPIF • Increases diffusion distance for nutrients and oxygen; ↓ BP, impaired circulation
- 34. Control of sodium ECF content Electrolyte balance • Main component of osmotic pressure; regulates ECF volume; water distribution; acid-base control • Concentration is stable (water moves between ECF and ICF due to osmosis) • Body content changes (changes in BP and BV) • No receptors; regulation through the blood volume • Na+ is reabsorbed in PCT, NL, and DCT; not secreted • Water follows Na+ ; ↑Na+ in filtrate → diluted urine • Renin/angiotensin/aldosterone system if ↑K+ or ↓Na+ → Na+ reabsorption, K+ secretion; BV ↑; slow
- 35. • ANP in response to ↑ BP → natriuresis, water secretion, ↓BV and BP; vasodilation • Inhibits renin, ADH, and aldosterone release • Estrogen: ↑ NaCl reabsorption → H2O retention (menstrual cycle, pregnancy) → edema • Progesterone: ↓ Na+ reabsorption (blocks aldosterone); promotes Na+ and H2O loss • Glucocorticoids: ↑ Na+ reabsorption → edema Anions • Cl– is major anion in ECF; 99% are reabsorbed under normal pH conditions • Acidosis ↓ Cl- reabsorption • Other anions have transport maximums and excesses are excreted in urine Control of sodium ECF content. Anions Electrolyte balance
- 36. Control of potassium ECF concentration. Anions Electrolyte balance • K+ concentration in plasma/ECF affects resting membrane potential • ↑K+ – ↓ excitability; ↓K+ – hyperpolarization, nonresponsiveness • Control is complicated – 98% K+ is in cells • Cellular regulation: shift between ECF and ICF • Renal regulation: reabsorption in PCT, secretion in NL and collecting duct • ↑ K+ in ECF (diet intake) → ↑ secretion (without aldosterone) • ↑ K+ in ECF → ↑ aldosterone → ↑ secretion (Na+ retained) • ↑ Na+ intake → ↑ tubular flow rate → ↑ secretion • If K+ intake is low, secretion ↓ - passive mechanism
- 37. Control of calcium ECF concentration Electrolyte balance • Mostly in the bones; regulates blood clotting, membrane permeability, secretion, neuromuscular excitability • ↑ Ca2+ → ↑ excitability and muscle tetany • ↓ Ca2+ → ↓ neurons and muscle cells, heart arrhythmias • Controlled by PTH; ↑ Ca2+ renal reabsorption (98%) • ↓ phosphate ion reabsorption (reabsorbed mainly in PCT), ↑ excretion (passively) • Via Ca2+ channels (apical membrane), Ca2+ pumps and antiporters (basal membrane), transcellular route • Insulin ↑ and glucagon ↓ Ca2+ reabsorption
- 38. Acid-base balance. Buffer systems • Sources of H+ : protein breakdown (phosphoric acid), lactic acid, fatty acids, ketone bodies • CO2 + H2O → H2CO3 → HCO3 - + H+ • H+ regulated by: buffer systems (rapid), respiratory centers (rapid), renal mechanisms (slow, most potent) • Strong bases quickly bind H+ ; strong acids quickly bind OH- Bicarbonate buffer • Mixture of H2CO3 (weak acid) and salts of HCO3 – (e.g., NaHCO3, a medium base) • Buffers ICF and ECF; only important ECF buffer Arterial blood Venous blood, IF ICF Alkalosis Acidosis pH 7.4 pH 7.35 pH 7.0 pH > 7.45 pH < 7.35 Phosphate buffer system • NaH2PO4 , a weak acid; Na2HPO4, a weak base • Buffers urine and ICF (PO4 3- concentrations are high) Protein buffer system • Buffer both plasma and ICF • Protein molecules are amphoteric (both weak acid and week base) • pH ↑ → COOH groups release H+ , become COO- • pH ↓ → -NH2 groups bind H+ , become NH3+ Acid-base balance
- 39. Renal and respiratory compensatory systems • Respiratory and renal systems are major (slow) regulators of pH; higher capacity than chemical buffers • Chemical buffers cannot eliminate excess acids or bases from body • Lungs: eliminate carbonic acid by eliminating CO2 • Kidneys: eliminate nonvolatile acids (phosphoric, uric, lactic acids; ketones); prevent metabolic acidosis • Kidneys: regulate blood levels of bases; renew chemical buffers • CO2 unloading → reaction shifts to left (and H+ incorporated into H2O) • CO2 loading → reaction shifts to right (and H+ buffered by proteins) CO2 ↑ H+ ↑ medullary receptors chemoreceptors respiratory rate and depth ↑H+ ↓ H+ ↓ respiratory center ↓ H+ ↑ • Hypercapnia → medullary chemoreceptors → ↑ respiratory rate and depth • ↑ plasma H+ → peripheral chemoreceptors → ↑ respiratory rate and depth • CO2 is removed from blood; H+ ↓ • Alkalosis (hypocapnea) → ↓ respiratory rate and depth decrease; H+ ↑ • Hypoventilation → respiratory acidosis • Hyperventilation → respiratory alkalosis Respiratory regulation Acid-base balance
- 40. • Renal mechanisms are most important; retain/secrete H+ • Cannot reabsorb HCO3 – , have to go around • H+ secretion in PCT and collecting duct • H+ comes from H2CO3 produced inside cells • As H+ secreted, Na+ reabsorbed Acid-base regulation in kidneys (1) Acid-base balance
- 41. • Rate of H+ secretion changes with ECF CO2 levels; ↑ CO2 in peritubular capillary blood ↑ H+ + secretion • To maintain alkaline reserve kidneys must replenish bicarbonate; cannot reabsorb • PCT cells: CO2 + H2O → H2CO3 → H+ (secreted) + HCO3 – (into capillary blood) – steps 1-3 • Filtrate: H+ + HCO3 – → H2CO3 → CO2 (into cells, starts over) + H2O – steps 4-6 Acid-base regulation in kidneys (2) Acid-base balance
- 42. • New HCO3 – has to be produced and added to alkaline reserve (balancing dietary H+ ) • Via renal excretion of acid (via secretion and excretion of H+ or NH4 + ) • Excreted H+ is buffered by phosphate in the urine Replenishing alkaline reserve Acid-base balance
- 43. Ammonium excretion • NH4 + – important mechanism for excreting acid • Glutamine in PCT cells → 2 NH4 + , 2 "new" HCO3 – • HCO3 – → blood; NH4 + → in urine • Replenishes alkaline reserve of blood • In alkalosis, type B intercalated cells secrete HCO3 – and reclaim H+ • Mechanism is opposite of HCO3 – ion reabsorption type A intercalated cells • Even in alkalosis, more HCO3 – is conserved than excreted Acid-base balance
- 44. Type Acidosis Alkalosis Respiratory Most important indicator is blood PCO2 PCO2 > 45 mm Hg Decreased in ventilation or gas exchange CO2 accumulates in blood PCO2 <35 mm Hg Common result of hyperventilation Often due to stress or pain CO2 eliminated faster than produced Metabolic Abnormally ↓ HCO3 - Ingestion of too much alcohol (→ acetic acid) Excessive loss of HCO3 – (e.g., persistent diarrhea) Accumulation of lactic acid (exercise or shock) Less common than metabolic acidosis ↑ blood pH and HCO3 – Vomiting of acid contents of stomach Intake of excess base (e.g., antacids) Symptoms Diabetic ketosis, starvation, kidney failure Blood pH below 6.8 → depression of CNS → coma → death Blood pH above 7.8 → excitation of nervous system → muscle tetany, extreme nervousness, convulsions, death often from respiratory arrest • Failure of respiratory system to regulate pH – respiratory acidosis/alkalosis • Other abnormalities other than caused by abnormal PCO2 – metabolic acidosis/alkalosis • Respiratory system tries to compensate metabolic acid-base imbalances; renal system – respiratory ones • Respiratory system cannot compensate for respiratory acidosis or alkalosis • Renal system cannot compensate for acid-base imbalances caused by renal problems Acid-base balance Imbalance Respiratory Metabolic Acidosis Alkalosis Acidosis Alkalosis Responses Excretion of H+ Retention of H+ ↑ H+ → ↑ rate and depth of breathing ↓ H+ → ↓ rate and depth of breathing Results H+ excretion → ↑ HCO3 - ↑ PCO2 (acidosis) H+ retention → ↓ HCO3 - ↓ PCO2 (alkalosis) pH < 7.35 (acidosis) HCO3 - ↓, PCO2 ↓ pH > 7.45 (alkalosis) HCO3 - ↑, PCO2 ↑ (> 45 mm Hg) Respiratory and renal compensation mechanisms
- 45. Physical and chemical characteristics Property Normal Abnormal Color • Pale to deep yellow (urochrome from hemoglobin breakdown) • More concentrated urine deeper color Pink, brown, smoky (food ingestion, bile pigments, blood, drugs) Transparency Clear Cloudy (may indicate UTI) Odor • Slightly aromatic when fresh • Ammonia odor upon standing (bacteria metabolize solutes) May be altered by some drugs and vegetables pH Slightly acidic (~pH 6, within 4.5 to 8.0) • Acidic diet (protein, whole wheat) ↓ pH • Alkaline diet (vegetarian), prolonged vomiting, or urinary tract infections ↑pH Specific gravity 1.001 – 1.035 Out of the range Composition Water – 95% Solutes – 5% Nitrogenous wastes Urea (from amino acid breakdown) Uric acid (from nucleic acid metabolism) Creatinine (from creatine phosphate) Other normal solutes: Na+ , K+, PO4 3– , and SO4 2– , Ca2+ , Mg2+ and HCO3 – Abnormally ↑ or ↓ concentrations of any constituent – pathology (e.g., glycosuria in diabetes) Abnormal components - pathology (e.g., blood proteins, WBCs, bile pigments) Urine transport, storage, and elimination
- 46. Ureters • Convey urine from kidneys to bladder; begin at L2 from renal pelvis; retroperitoneal • Enter base of bladder through posterior wall; bladder pressure ↑ → distal ends of ureters close → no urine backflow Urine transport, storage, and elimination Layer Structure Function Mucosa Transitional epithelium Secretion, protection Muscularis Smooth muscle Contraction in response to stretch; propels urine Adventitia Fibrous connective tissue Support
- 47. Urinary bladder • Muscular sac; temporary stores urine; retroperitoneal, on pelvic floor • Posterior to pubic symphysis, superior to prostate (males), anterior to vagina and uterus (females) • Ureters in, urethra out; trigone – between openings of ureters and urethra; most frequently infected • Collapses when empty; expands and rises during filling; pressure remains stable • ~ Full bladder 12 cm long; holds ~ 500 ml; can hold ~ twice that if necessary, but can burst Urine transport, storage, and elimination Layer Structure Function Mucosa Transitional epithelium Secretion, protection Muscularis Smooth detrusor muscle (3 layers) Contraction; propels urine Adventitia Fibrous connective tissue Support
- 48. Urethra Epithelium Where Transitional Near bladder Stratified squamous External urethral orifice Sphincters Muscles Where Control Internal urethral Smooth Bladder-urethra junction Involuntary (ANS) External urethral Skeletal At pelvic floor Voluntary • Drains urinary bladder • Female: 3–4 cm; bound to anterior vaginal wall; orifice anterior to vaginal opening; posterior to clitoris • Male: carries semen and urine; prostatic (2.5 cm), membranous (2 cm), and spongy urethra (15 cm) Urine transport, storage, and elimination
- 49. • Reflexive urination (urination in infants): pontine control (storage and micturition) centers develop only between 2 and 3 yo (right part of the picture is absent in kids) Micturition (urination) Urine transport, storage, and elimination
- 50. • Incontinence: usually weak pelvic muscles • Stress incontinence: increased intra-abdominal pressure forces urine through external sphincter • Overflow incontinence: urine dribbles when bladder overfills • Urinary retention: bladder unable to expel urine; common after general anesthesia; hypertrophy of prostate; treatment is catheterization Pathologies of micturition • Frequent micturition in infants: small bladders and less-concentrated urine • Incontinence in infants: normal; control of voluntary urethral sphincter develops with nervous system • Urinary tract infections: E. coli (≈80%), Klebsiella, Proteus mirabilis, Enterobacter, Streptococcus saprophyticus, Staphylococcus aureus, Enterococcus faecalis • Untreated infections may cause long-term renal damage → hypertension (?) • Sexually transmitted diseases: gonorrhea, chlamydiasis etc. may lead to urinary tract inflammation and kidney problems • Elderly people: most have abnormal kidneys histologically • Kidneys shrink; nephrons decrease in size and number; tubule cells less efficient • GFR ½ that of young adult by age 80; atherosclerosis of renal arteries (?) • Bladder shrinks; loss of bladder tone nocturia and incontinence Urine transport, storage, and elimination
- 51. Common congenital abnormalities • Horseshoe kidney (left): two kidneys fuse across midline single U-shaped kidney; usually asymptomatic • Hypospadias (right): urethral orifice on ventral surface of penis; corrected surgically at ~ 12 months • Polycystic kidney disease (left): many fluid-filled cysts interfere with function • Autosomal dominant form – less severe, more common; autosomal recessive – more severe • Cause unknown but involves defect in signaling proteins Urinary diseases
- 52. Kidney infections. Renal calculi • Urinary tract infections can reach the kidneys; cause inflammation of the pelvis, calyces, or entire organ • Pyelitis – pelvis and calyces are affected (left top); pyelonephritis – whole organ is affected (left bottom) • Treatment – antibiotics, sometimes surgical removal of affected kidney • Renal calculi – kidney stones in renal pelvis; formed when calcium, magnesium, or uric acid salts crystalize • Large stones block ureter pressure & pain • Causes: chronic bacterial infection, urine retention, ↑Ca2+ in blood, ↑pH of urine • Treatment - shock wave lithotripsy – noninvasive; shock waves shatter calculi Urinary diseases