Immunogenicity manipulation
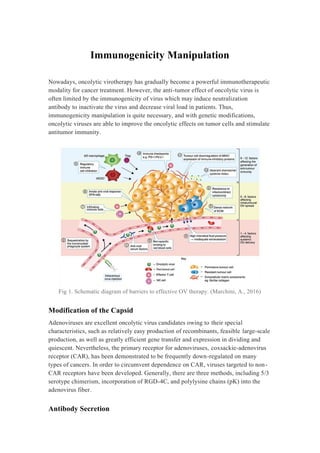
- 1. Immunogenicity Manipulation Nowadays, oncolytic virotherapy has gradually become a powerful immunotherapeutic modality for cancer treatment. However, the anti-tumor effect of oncolytic virus is often limited by the immunogenicity of virus which may induce neutralization antibody to inactivate the virus and decrease viral load in patients. Thus, immunogenicity manipulation is quite necessary, and with genetic modifications, oncolytic viruses are able to improve the oncolytic effects on tumor cells and stimulate antitumor immunity. Fig 1. Schematic diagram of barriers to effective OV therapy. (Marchini, A., 2016) Modification of the Capsid Adenoviruses are excellent oncolytic virus candidates owing to their special characteristics, such as relatively easy production of recombinants, feasible large-scale production, as well as greatly efficient gene transfer and expression in dividing and quiescent. Nevertheless, the primary receptor for adenoviruses, coxsackie-adenovirus receptor (CAR), has been demonstrated to be frequently down-regulated on many types of cancers. In order to circumvent dependence on CAR, viruses targeted to non- CAR receptors have been developed. Generally, there are three methods, including 5/3 serotype chimerism, incorporation of RGD-4C, and polylysine chains (pK) into the adenovirus fiber. Antibody Secretion
- 2. To solve immunogenicity problems, antibody secretion is a very useful method that enhance the anti-tumor immune response. For example, an oncolytic group B adenovirus EnAdenotucirev (EnAd) has been designed to express a bispecific single- chain antibody, secreted from infected tumour cells into the microenvironment. This strategy increases the cytotoxicity of EnAd, and it has proven that using primary pleural effusions and peritoneal malignant ascites that infection of cancer cells with the BiTE-expressing EnAd causes activation of endogenous T cells to kill endogenous tumour cells. In this way, an EnAd combining both direct oncolysis and T cell- mediated killing is performed. Cytokine/chemokine Expression Expressing cytokine/chemokine to attract immune cells migration to the tumor site is also a powerful approach. Researchers have constructed two oncolytic herpes simplex viruses (HSV) which enable to express the murine GM-CSF and murine IL-12 genes. The two cytokine-carrying variants are contrasted with the analogous non-cytokine- carrying control virus in the treatment of murine SCC VII squamous cell carcinoma. It has demonstrated that the GM-CSF and IL-12 expression HSV displays a much stronger antitumor efficacy than non-cytokine-carrying control virus. Load with Immune Checkpoint Inhibitors Immune system has an essential capacity to distinguish normal cells in the body and those it considers as “foreign.” To achieve this, “checkpoints” are used, which refers to molecules on certain immune cells needed to be activated (or inactivated) to start an immune response. However, sometimes cancer cells find ways to utilize these checkpoints to avoid being attacked. Thus, drugs that target these checkpoints hold great promises as cancer treatments. Fig 2. Combinatorial methods to overcome barriers to oncolytic virus‐induced oncolysis and immune stimulation. (Marchini, A., 2016)
- 3. Reference: 1. Marchini, A., (2016). “Overcoming barriers in oncolytic virotherapy with HDAC inhibitors and immune checkpoint blockade.” Viruses, 8(1), 9. 2. Wong, R. J., (2001). “Cytokine gene transfer enhances herpes oncolytic therapy in murine squamous cell carcinoma.” Human gene therapy, 12(3), 253-265.