This document provides information about carbon compounds. It begins by defining carbon compounds as compounds that contain carbon as a constituent element. It then discusses why carbon is unique in its ability to form four covalent bonds and long chains of carbon atoms. The document separates carbon compounds into organic and inorganic compounds. It focuses on organic compounds, especially hydrocarbons. It discusses saturated and unsaturated hydrocarbons, including alkanes, alkenes, and alkynes. The document explains how to name compounds in each homologous series and describes some of their physical and chemical properties.




































![1.b) Combustion (Incomplete)
What happen if there is insufficient Oxygen?
The possible balanced chemical equation for the
incomplete combustion of methane gas is:
CH4 + O2 C + 2H2O [carbon only]
2CH4 + 3O2 2CO + 4H2O [carbon monoxide only]
4CH4 + 5O2 2CO2 + 2C + 8H2O [mixture of both]](https://image.slidesharecdn.com/topic3carboncompound-230314075557-843fb35f/85/TOPIC-3-CARBON-COMPOUND-pptx-37-320.jpg)









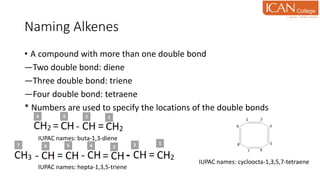
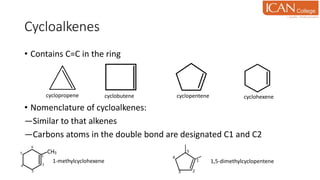








![Addition of hydroxyl group
• Alkenes react with acidified potassium
manganate(VII), KMnO4 to produce diol
compound
C2H4 + H2O + [O] → C2H4(OH)2
or
C2H4 C2H4(OH)2
KMnO4
Used to test for the presence of a carbon-carbon double bond](https://image.slidesharecdn.com/topic3carboncompound-230314075557-843fb35f/85/TOPIC-3-CARBON-COMPOUND-pptx-57-320.jpg)





