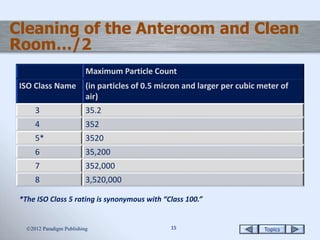
This document discusses the sterile compounding environment, including the setup and characteristics of anterooms and clean rooms. It describes the ISO classification levels for particulate matter in these areas and different types of hoods used for sterile compounding. USP Chapter <797> guidelines for hoods and the four risk levels for sterile compounding are also summarized.