This document outlines the structure and key elements that should be included in a systematic review report. It recommends including an abstract, introduction, methods, results, discussion and conclusion sections. The methods section should describe the literature search strategy, eligibility criteria, data extraction and quality assessment processes. Results should be presented according to the review questions. The discussion should synthesize the findings, discuss limitations and draw conclusions. Guidelines like PRISMA can help improve reporting quality.











![Present the complete electronic search strategy — including any limits used — in the Appendix of the report. Brief example: We used the following search terms to search all trials registers and databases: immunoglobulin; IVIG [intravenous immunoglobulin]; sepsis; septic shock; septicaemia; and septicemia. The purpose of including the entire search strategy is to ensure transparency and to permit replication of the review. Methods: Literature Search Strategy Alejandria MM, et al. Cochrane Database Syst Rev 2002;(1):CD001090.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-13-320.jpg)

![State the processes used to select studies for review: Screening Eligibility assessment Inclusion/exclusion criteria applied for the systematic review and, if applicable, the meta-analysis Example: We included trials if the randomization scheme included groups that assigned patients to treatment guided by the PAC [pulmonary artery catheter] or treatment without the PAC. We only included trials if they reported death and number of days hospitalized or the number of days in the ICU as outcome measures. Studies were excluded if the randomization scheme did not specify groups as PAC or no PAC, if patients were not randomized to a conventional PAC, if investigators combined randomized and nonrandomized groups when reporting outcomes, or if there were no outcome data on death or hospitalizations. Methods: Eligibility Criteria (l) Shah MR, et al. JAMA 2005;294:1664-70.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-15-320.jpg)
![Type of studies: Example: Randomised clinical trials studying the administration of hepatitis B vaccine to CRF [chronic renal failure] patients, with or without dialysis. No language, publication date, or publication status restrictions were imposed. Types of participants: Example: Participants of any age with CRF or receiving dialysis (haemodialysis or peritoneal dialysis) were considered.… Renal transplant patients were excluded from this review. Methods: Eligibility Criteria (II) Schroth RJ, et al. Cochrane Database Syst Rev 2004;(3):CD003775.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-16-320.jpg)
![Types of interventions: Example: Trials comparing the beneficial and harmful effects of hepatitis B vaccines with adjuvant or cytokine co-interventions [and] trials comparing the beneficial and harmful effects of immunoglobulin prophylaxis.… Hepatitis B vaccines (plasma or recombinant [yeast] derived) of all types, dose, and regimens versus placebo, control vaccine, or no vaccine. Types of outcomes: Example: Primary outcome measures: Seroconversion , [that is], proportion of patients with adequate anti-HBs response (10 IU/L or Sample Ratio Units).…Secondary outcome measures: Adverse events of hepatitis B vaccinations…[and] mortality . Methods: Eligibility Criteria (III) Schroth RJ, et al. Cochrane Database Syst Rev 2004;(3):CD003775.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-17-320.jpg)
![Describe the method of data extraction. Example: We developed a data extraction sheet[,]…pilot-tested it on ten randomly-selected included studies, and refined it accordingly. One review author extracted the…data…and the second author checked the extracted data. …Disagreements were resolved by discussion between the two review authors. Describe any processes used to obtain and confirm data from other investigators. Methods: Data Extraction Mistiaen P, Poot E. Cochrane Database Syst Rev 2006;(4):CD004510.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-18-320.jpg)
![List and define all variables for which data were sought, using the PICOTS framework as a guide. List any assumptions and simplifications that were made in defining the variables. Example: Information was extracted from each included trial on: (1) characteristics of trial participants…and the trial’s inclusion and exclusion criteria; (2) type of intervention…(versus placebo or versus the type, dose, duration and frequency of another NSAID [nonsteroidal antiinflammatory drug]; or versus another pain management drug; or versus no treatment); (3) type of outcome measure. Methods: Data Items Allen C, et al. Cochrane Database Syst Rev 2005;(4):CD004753. PICOT(S) = population, intervention, comparator, outcome, timing, and study design or setting](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-19-320.jpg)

![For each meta-analysis: Describe the methods used to handle the data and to combine the results of studies. Describe measures of consistency (e.g., I-squared). Examples from two separate studies: In very few instances, estimates of baseline mean or mean QOL [quality of life] responses were obtained without corresponding estimates of variance (standard deviation [SD] or standard error). In these instances, an SD was imputed from the mean of the known SDs. In a number of cases, the response data available were the mean and variance in a prestudy condition and after therapy.… We tested for heterogeneity with the Breslow-Day test, and used the method proposed by Higgins et al. to measure inconsistency. Methods: Synthesis of Results Jones M, et al. Cancer 2004;101:1720-32; Briel M, et al. Am J Med 2004;117:596-606.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-21-320.jpg)



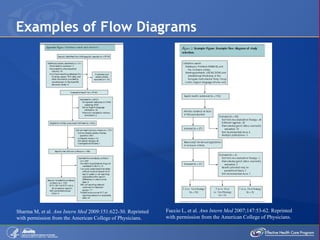
![Place at the beginning of the Results section, not in the Methods section. Give numbers of studies screened, assessed for eligibility, and included in the review. Give the reasons for exclusions at each stage of the assessment, ideally illustrated with a flow diagram. Example: The search of Medline, PsycInfo and Cinahl [sic] databases provided a total of 584 citations. After adjusting for duplicates 509 remained. Of these, 479 studies were discarded because…. Three additional studies…were discarded because…. The full text of the remaining 27 citations was examined in more detail. It appeared that 22 studies did not meet the inclusion criteria as described. Five studies…met the inclusion criteria and were included in the systematic review. Results: Study Selection Uitterhoeve RJ, et al. Br J Cancer. 2004;91:1050-62.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-25-320.jpg)




![Include risk of bias, directness, consistency, and precision in reporting how evidence was graded. Example: There is a low level of evidence…that RFA [radiofrequency catheter ablation] improves quality of life more than medical treatment. Three RCTs [randomized controlled trials] and one observational study reported more improvement in the general or physical functioning…in patients who underwent RFA.…However, these studies assessed the results at nonuniform time points and therefore the findings may be difficult to interpret. Results: Grading Strength of Evidence Ip S, et al. AHRQ Comparative Effectiveness Review No. 15. Available at: http://www.effectivehealthcare.ahrq.gov/ehc/products/51/114/2009_0623RadiofrequencyFinal.pdf.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-31-320.jpg)
![Give the results of additional analyses, such as sensitivity or subgroup analyses and meta-regressions. Include the results of additional analyses to facilitate a better understanding of heterogeneity. Example 1: [B]enefits of chondroitin were smaller in trials with adequate concealment of allocation compared with trials with unclear concealment (P for interaction = 0.050), in trials with an intention-to-treat analysis compared with those that had excluded patients from the analysis (P for interaction = 0.017), and in large compared with small trials (P for interaction = 0.022). Results: Additional Sensitivity Analyses Reichenbach S, et al. Ann Intern Med 2007;146:580-90.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-32-320.jpg)
![Example 2: Subgroup analyses according to antibody status, antiviral medications, organ transplanted, treatment duration, use of antilymphocyte therapy, time to outcome assessment, study quality and other aspects of study design did not demonstrate any differences in treatment effects. Multivariate meta-regression showed no significant difference in CMV [cytomegalovirus] disease after allowing for potential confounding or effect-modification by prophylactic drug used, organ transplanted or recipient serostatus in CMV positive recipients and CMV negative recipients of CMV positive donors. Results: Additional Subgroup Analyses Hodson EM, et al. Cochrane Database Syst Rev 2008;(2):CD003774.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-33-320.jpg)

![Example: Compared with men who used watchful waiting , men with clinically localized prostate cancer detected by methods other than PSA [prostate-specific antigen] testing and treated with radical prostatectomy experienced fewer deaths from prostate cancer, marginally fewer deaths from any cause, and fewer distant metastases. The greater benefit of RP on cancer-specific and overall mortality appears to be limited to men under 65 years of age but is not dependent on baseline PSA level or histologic grade. Discussion: Summary of Evidence Wilt TJ, et al. AHRQ Comparative Effectiveness Review No. 13. Available at: . http://www.effectivehealthcare.ahrq.gov/ehc/products/9/80/2008_0204ProstateCancerFinal.pdf.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-35-320.jpg)

![Provide a general interpretation of the results in the context of other evidence and the implications for future research. Example: [T]he available clinical trial evidence supporting the use of combination therapies over higher dose statin therapy is insufficient to guide clinical decisions. The long term clinical benefits and risks of combination therapies have yet to be demonstrated. There are some instances, such as failure to reach targets in spite of maximal statin therapy, and populations with elevated triglycerides who need to achieve secondary goals, in which clinicians may choose combinations pending definitive evidence. Discussion: Conclusions Sharma M, et al. AHRQ Comparative Effectiveness Review No. 16. Available at: http://www.effectivehealthcare.ahrq.gov/ehc/products/11/171/reptbodyfin-typofixed4-12-2010.pdf.](https://image.slidesharecdn.com/reportingthereview-100719142119-phpapp01/85/Reporting-the-Review-37-320.jpg)








