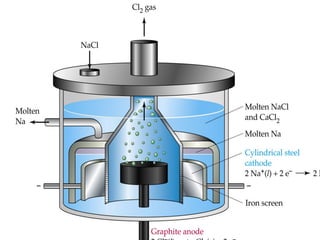
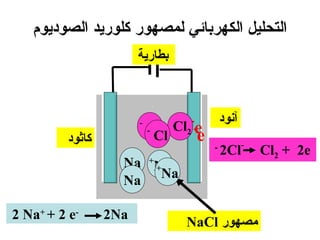
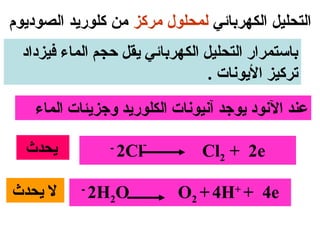
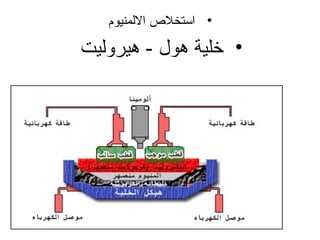
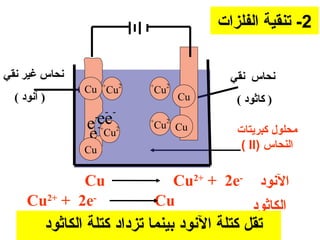
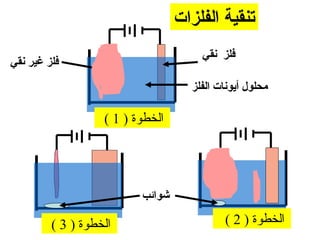
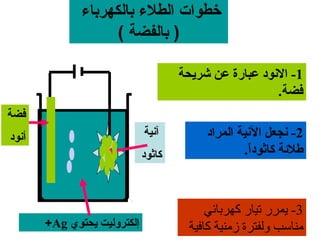
يتناول الوثيقة عمليات التحليل الكهربائي واستخدامات الخلايا الجلفانية لتنقية المعادن مثل النحاس والفضة. يتم شرح عملية التحليل الكهربائي لمحلول الكلوريد والصوديوم وكيفية تفاعل الأيونات في الكاثود والانوند. الوثيقة توضح أيضاً كيفية زيادة كتلة الكاثود وتقليل كتلة الأنوكود خلال عمليات التنقية.