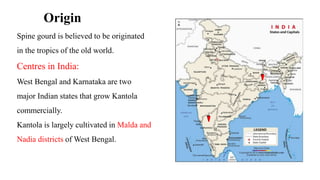
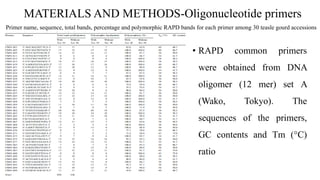
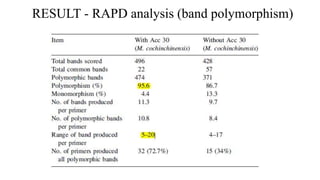
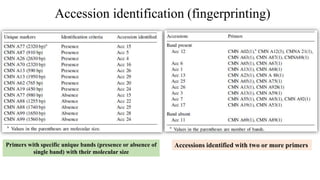
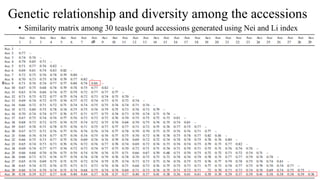
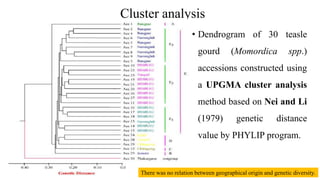
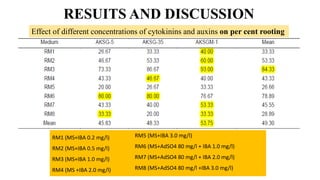
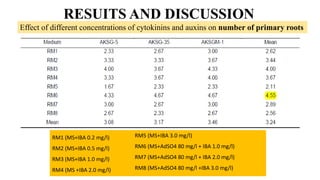
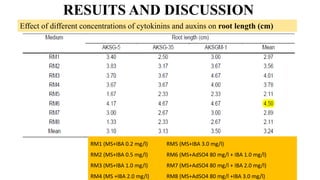
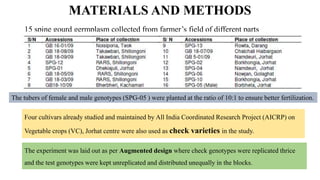
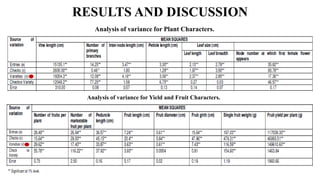
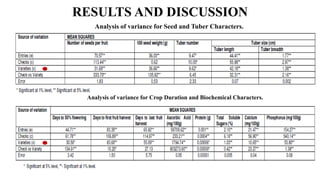
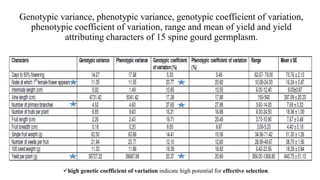
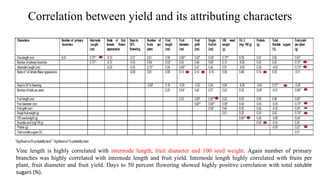
The document summarizes information about the spine gourd (Momordica dioica) plant. It discusses the origin, nutritional value, medicinal uses, varieties, cultivation practices, propagation methods, harvesting, and yield of spine gourd. It also describes some related Momordica species and includes references to research articles on genetic diversity analysis and in vitro propagation of spine gourd.