A tandem-column method using chiralpak ad-h and chiralcel od-h columns was developed for baseline separation of four stereoisomers via supercritical fluid chromatography (SFC) with a specific mobile phase. Individual analysis with each column alone did not yield a complete separation, highlighting the advantages of using coupled columns. The study emphasizes the importance of selecting appropriate mobile phases and conditions to achieve effective separation in pharmaceutical applications.
![Barnhart, Gahm, Tomas, Notari, Semin, Cheetham 619
Wesley W. Barnhart Supercritical fluid chromatography tandem-column
Kyung H. Gahm
Sam Thomas method development in pharmaceutical sciences
Steve Notari for a mixture of four stereoisomers
David Semin
Janet Cheetham
A tandem-column method using Chiralpak AD-H and Chiralcel OD-H columns was
Discovery Analytical Sciences, achieved for baseline separation of a mixture of chiral pharmaceutical compounds
Molecular Structure, Amgen Inc., (i. e., four stereoisomers) via supercritical fluid chromatography (SFC) with a mobile
Thousand Oaks, CA 91320, USA phase consisting of 90% liquid carbon dioxide and 10% ethanol:isopropanol (50 : 50
v/v). On the contrary, this mixture (mixture A) could not be baseline separated by
SFC conditions explored with individual Chiralpak AD-H and Chiralcel OD-H columns.
The effects of various mobile phases on elution order, capacity factor, selectivity, and
resolution were determined with mixture A on the individual aforementioned columns
to develop the tandem-column method.
Key Words: Supercritical fluid chromatography; Tandem-column; Diastereomer;
Received: January 4, 2005; revised: February 15, 2005; accepted: February 24, 2005
DOI 10.1002/jssc.200500005
1 Introduction can be overcome is to utilize tandem columns to obtain a
desired single-method separation. In this paper, an analy-
Supercritical fluid chromatography (SFC) is an important tical SFC method was developed by employing the cou-
analytical and preparative tool used in the pharmaceutical pling of Chiralpak AD-H and Chiralcel OD-H columns to
industry. Separation of chiral pharmaceutical compounds separate a mixture of pharmaceutical compounds (i.e.,
is an ever-increasing chromatographic field [1], and chiral- four stereoisomers); this separation was not achieved
ity is an important factor in drug development [2 – 5] due to using a single-column method. By understanding the elu-
the chiral specificity of biological processes [6]. Separa- tion order and effects of various mobile phases on the
tion of chiral compounds is vital because the activity of separation of the compounds by Chiralpak AD-H and
each enantiomer must be explored; it is possible that one Chiralcel OD-H columns separately, the development of
enantiomer may be inactive, an antagonist, or toxic [5 – 7]. the tandem-column method was made possible. A single-
A classic example is thalidomide. Originally marketed as a method separation is more desirable than multiple meth-
sedative, thalidomide was a racemic mixture; one enantio- ods, since it is less time consuming and cumbersome,
Original Paper
mer was a sedative, while the other was teratogenic [8, 9]. requiring no change of column or mobile phase conditions
SFC was the new and exciting chiral chromatographic between the analyses of different compounds.
topic in the early to mid 1980s [10, 11] when it was demon-
The use of coupled columns to separate compounds chro-
strated to be a technique capable of separating enantio-
matographically is not a novel concept [19, 22 – 27]; the
mers [12]. The use of SFC for the separation of enantio-
difficulty, however, is the determination of the correct
mers has been one of its most successful applica-
combination of columns given the large number of station-
tions [13]. Though the technique is over 20 years old, its
ary phases available on the market. According to San-
potential is continuing to be explored. SFC is a popular
dra [23], the selectivity and efficiency of a single column
chiral separation technique due to the inherent advan-
often does not provide adequate separation of a given
tages of using liquid CO2 in the mobile phase [1, 3, 11,
mixture of compounds. Recently, Phinney et al. [24]
14 – 21].
coupled achiral and chiral columns to separate a mixture
The development of a successful and facile single-column of achiral and chiral compounds.
analytical chiral separation method is not always achieved For our study, however, two chiral columns were coupled
due to limited in-house column selections or time restric- for the purpose of efficiently separating chiral compounds
tions imposed by the project cycle times of early discovery (a mixture of four stereoisomers). The initial choice to
chemistry projects. One way in which these limitations screen the Chiralpak AD-H and Chiralcel OD-H columns
was based on past successful experiences with these col-
Correspondence: Wesley W. Barnhart, Discovery Analytical
Sciences, Molecular Structure, Amgen Inc., Thousand Oaks, CA umns; Chiralpak AD and Chiralcel OD columns have been
91320, USA. Phone: +1 805 447 2055. Fax: +1 805 480 3015. shown to be very successful and widely used in chiral
E-mail: wesleyb@amgen.com. chromatography [1, 28, 29]. In addition to separating four
J. Sep. Sci. 2005, 28, 619 – 626 www.jss-journal.de i 2005 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim](https://image.slidesharecdn.com/sfctandemcolumn-13250222261964-phpapp02-111227154523-phpapp02/75/Amgen2-1-2048.jpg)


![SFC tandem-column method development 623
3.4 Analytical scale AD-H-OD-H tandem column
analyses of mixture A
Individual AD-H and OD-H columns did not provide an
adequate method for the baseline separation of all four
stereoisomers. The individual column results previously
discussed, however, indicated the possibility of separat-
ing all four stereoiosomers if the AD-H and OD-H columns
were coupled. Organic modifiers were chosen based on
the results of the individual AD-H and OD-H column
screening. When considering tandem-column LC, the
retention time noted for a particular analyte is the sum of
the retention times of the analyte for the individual col-
umns; this is independent of the column order [25]. It was
by adding the peak retention times for the individual AD-H
and OD-H columns (with EtOH and IsOH as the organic
modifiers) that there appeared the possibility of resolving
all four stereoisomers through the coupling of the two col-
umns.
The tandem-column methods explored utilized mobile
phases that consisted of 90% liquid CO2 and 10% EtOH,
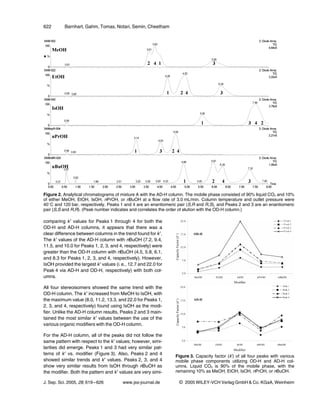
IsOH, or EtOH : IsOH (50 : 50 v/v) (Figure 5). Looking at
Figure 4. Resolution of Peaks 1 & 4 (D) and 2 & 3 (F) utilizing
different mobile phase components (MeOH, EtOH, IsOH, the tandem-column separation with EtOH and IsOH indivi-
nPrOH, and nBuOH) with the OD-H and AD-H columns. dually, it was predicted that a combination of the two
Liquid CO2 is 90% of the mobile phase, with the modifier would provide the desired separation. A combination of
(MeOH, EtOH, IsOH, nPrOH, or nBuOH) as the remaining EtOH and IsOH (EtOH : IsOH (50 : 50 v/v)) was then used
10%.
to achieve the separation of the four stereoisomers with
an analysis time of less than 15 min. Since baseline sep-
lar, indicating the AD-H is a column that had very similar
aration was already achieved with the 50 : 50 mixture,
affinities for all three peaks with IsOH, nPrOH, and
further exploration of EtOH : IsOH combinations for
nBuOH as the modifiers.
method optimization was not conducted. Also, reversing
the order of the tandem-columns did not result in a notable
difference in the separation of the stereoisomers.
3.3 Resolution (R ) of enantiomeric pairs of By utilizing the separation characteristics of individual
mixture A for OD-H and AD-H columns Chiralpak AD-H and Chiralcel OD-H columns, it was pos-
sible to obtain baseline separation of all four of the stereo-
The resolution (R) for the enantiomeric pairs was calcu- isomers through the coupling of the two columns; this was
lated and plotted (Figure 4). The resolution plots in Fig- not achieved with a single column. In addition, the base-
ure 4 show similar patterns found for the selectivities (not line separation was only accomplished by combining two
shown), as expected. For the OD-H column, the resolution mobile phase components (EtOH and IsOH) for the
patterns of both peak pairs (Peaks 1 & 4 and 2 & 3) were coupled column method.
very similar; however, Peaks 1 & 4 demonstrated much
greater R values than those found with Peaks 2 & 3, thus
indicating that the OD-H column was much better suited 3.5 Application of the analytical tandem-column
for separating the enantiomers 1 & 4 than 2 & 3. Peaks 1 & method
4 were best separated on the OD-H column using MeOH The analytical tandem-column SFC method was success-
as the organic modifier. fully applied to quickly determine the results of a reaction
A closer look at the AD-H results showed that each of the designed to transform a precursor into two of the four
enantiomeric pairs had much different behavior. The stereoisomers found in mixture A. Figure 6 shows the
modifier that resulted in the greatest R value for enantio- chromatograms indicating the precursor, the two stereo-
meric pairs 2 & 3 was MeOH. The lowest R value for enan- isomers formed, and a chromatogram of the original mix-
tiomeric pairs 2 & 3 was obtained through the use of IsOH ture A. The precursor could be easily separated from the
as the organic modifier. For 1 & 4, the greatest R value stereoisomers in mixture A, which would provide a quick
was observed with nPrOH as the modifier, and the lowest and facile procedure to monitor both the presence of the
R value resulted from the use of MeOH as the modifier. precursor and the product of the reaction utilizing a single
J. Sep. Sci. 2005, 28, 619 – 626 www.jss-journal.de i 2005 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim](https://image.slidesharecdn.com/sfctandemcolumn-13250222261964-phpapp02-111227154523-phpapp02/85/Amgen2-5-320.jpg)
![624 Barnhart, Gahm, Tomas, Notari, Semin, Cheetham
Figure 5. Chromatograms of the AD-H and OD-H column coupling for the separation of the four stereoisomers. The mobile
phase consisted of 90% liquid CO2 and 10% EtOH, IsOH, or EtOH : IsOH (50 : 50 v/v). The temperature and outlet pressure were
408C and 120 bar, respectively. Total flow rate was 3.0 mL/min. (Peak number indicates and correlates the order of elution with
the OD-H column.)
method. This was critical for a timely analysis of the single-column method. However, with the columns in tan-
results of the reaction. A single column could have been dem, a single method was successfully established. The
employed to resolve Peaks 1 and 2; however, both benefit of a single method was quickly realized when a
Peaks 3 and 4 would not have been identified (i. e., reaction (that converted a precursor into two of the stereo-
resolved) if they had also been formed during the reac- isomers of mixture A) was efficiently and effectively moni-
tion. tored; this provided critical data in a timely manner to
easily fit within the timelines provided for project support.
3.6 Preparative scale AD-H-OD-H tandem-column Overall, the order of elution of the four stereoisomers with
separation of mixture A the AD-H column was difficult to predict, while the order of
After establishing a tandem-column analytical method, elution remained constant with the OD-H column. This
the next step was to attempt a preparative separation. To indicates the separation mechanism on the AD-H column
demonstrate proof-of-concept, the same mobile phase for these compounds is more complicated compared to
composition (90% liquid CO2 and 10% EtOH : IsOH the OD-H column. Since both columns contain the same
(50 : 50 v/v)) was used for the preparative separation (Fig- derivative (tris-3,5-dimethylphenylcarbamate [30, 31]),
ure 7) as that used on the analytical scale. The overall the behavior of the stereoisomers with the AD-H column is
separation time was 24 min. This example illustrates how most likely due to the difference in the structure of amy-
a single method with coupled columns can be used to lose (AD-H) compared to cellulose (OD-H). The helical
baseline separate four stereoisomers and facilitate the nature of amylose allows for better inclusion when com-
purification process. pared to the more planar cellulose [3]. With the AD-H col-
umn, different alcohol modifiers in the mobile phase can
alter the chiral cavities and could modify the elution order
4 Conclusions of the peaks [32]. The AD-H column may therefore allow
Method development to separate a mixture of four stereo- more diverse interactions than the OD-H column resulting
isomers with AD-H or OD-H columns did not produce a in varied elution order with change of organic modifier.
J. Sep. Sci. 2005, 28, 619 – 626 www.jss-journal.de i 2005 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim](https://image.slidesharecdn.com/sfctandemcolumn-13250222261964-phpapp02-111227154523-phpapp02/85/Amgen2-6-320.jpg)
![SFC tandem-column method development 625
Figure 6. Application of the tandem-column method for the analysis of the end products of a reaction. Chromatograms of a pre-
cursor, peaks 1 & 2, and mixture A are shown. The mobile phase consisted of 90% liquid CO2 and 10% EtOH : IsOH (50 : 50 v/v).
The temperature and outlet pressure were 408C and 120 bar, respectively. Total flow rate was 3.0 mL/min. (Peak number indi-
cates and correlates the order of elution with the OD-H column.)
Figure 7. Preparative chroma-
togram of 1 mg of mixture A uti-
lizing the tandem-column
method. The mobile phase con-
sisted of 90% liquid CO2 and
10% EtOH : IsOH (50 : 50 v/v).
The column oven and nozzle
temperature were 408C, and
the outlet pressure was
120 bar. Total flow rate was
55 mL/min. (Peak number indi-
cates and correlates the order
of elution with the OD-H col-
umn.)
An advantage of analyzing the effects of organic modifiers References
on elution order of the stereoisomers is to enable an effi-
[1] L. Toribio, J.L. Bernal, M.J. del Nozal, J.J. Jimenez, E.M.
cient purification of future mixtures of one or more of the Nieto, J. Chromatogr. A 2001, 921, 305 – 313.
stereoisomers. Typically after the potency of the individual
[2] Y. Zhao, G. Woo, S. Thomas, D. Semin, P. Sandra, J. Chro-
stereoisomers is ascertained, further purification may only matogr. A 2003, 1003, 157 – 166.
involve one or two of the stereoisomers. With the ability to
[3] A. Medvedovici, P. Sandra, L. Toribio, F. David, J. Chroma-
change the selectivity of the peak with the AD-H column, it togr. A 1997, 785, 159 – 171.
is concluded that a scalable purification method could be [4] U. Selditz, S. Copinga, J.P. Franke, H. Wikstrom, R.A. de
derived based on the current data. Zeeuw, Chirality 1996, 8, 574 – 578.
J. Sep. Sci. 2005, 28, 619 – 626 www.jss-journal.de i 2005 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim](https://image.slidesharecdn.com/sfctandemcolumn-13250222261964-phpapp02-111227154523-phpapp02/85/Amgen2-7-320.jpg)
![626 Barnhart, Gahm, Tomas, Notari, Semin, Cheetham
[5] G. Cancelliere, I. D'Acquarica, F. Gasparrini, D. Misiti, C. Vil- [20] Y.K. Ye, K.G. Lynam, R.W. Stringham, J. Chromatogr. A
lani, PSTT 1999, 2, 484 – 492. 2004, 1041, 211 – 217.
[6] J. Szymura-Oleksiak, J. Bojarski, H.Y. Aboul-Enein, Chiral- [21] Y. Liu, A. Berthod, C.R. Mitchell, T.L. Xiao, B. Zhang, D.W.
ity 2002, 14, 417 – 435. Armstrong, J. Chromatogr. A 2002, 978, 185 – 204.
[7] M.S. Villeneuve, R.J. Anderegg, J. Chromatogr. A 1998, [22] T.A. Berger, W.H. Wilson, Anal. Chem. 1993, 65, 1451 –
826, 217 – 225. 1455.
[8] J.C. Reepmeyer, Chirality 1996, 8, 11 – 17. [23] K. Anton, C. Berger, Supercritical Fluid Chromatography
[9] S.N. Osipov, P. Tsouker, L. Hennig, K. Burger, Tetrahedron with Packed Columns: Techniques and Applications, Marcel
2004, 60, 271 – 274. Dekker, Inc., New York 1998.
[24] K.W. Phinney, L.C. Sander, S.A. Wise, Anal. Chem. 1998,
[10] R.M. Smith, J. Chromatogr. A 1999, 856, 83 – 115.
70, 2331 – 2335.
[11] G. Terfloth, J. Chromatogr. A 2001, 906, 301 – 307.
[25] W.H. Pirkle, C.J. Welch, J. Chromatogr. A 1996, 731, 322 –
[12] O. Gyllenhaal, J. Chromatogr. A 2004, 1042, 173 – 180. 326.
[13] K.L. Williams, L.C. Sander, J. Chromatogr. A 1997, 785, [26] T.A. van Beek, M.S. Wintermans, J. Chromatogr. A 2001,
149 – 158. 930, 109 – 117.
[14] W.C. Ripka, G. Barker, J. Krakover, DDT 2001, 6, 471 – 477. [27] Q.B. Cass, R.F. Gomes, S.A. Calafatti, J. Pedrazolli Jr., J.
[15] K.G. Lynam, E.C. Nicolas, J. Pharm. Biomed. Anal. 1993, Chromatogr. A 2003, 987, 235 – 241.
11, 1197 – 1206. [28] J. Whatley, J. Chromatogr. A 1995, 697, 251 – 255.
[16] M. Johannsen, J. Chromatogr. A 2001, 937, 135 – 138. [29] F. Wang, T. Dowling, D. Ellison, J. Wyvratt, J. Chromatogr.
[17] K.L. Williams, L.C. Sander, S.A. Wise, Chirality 1996, 8, A 2004, 1034, 117 – 123.
325 – 331. [30] E. Lipka-Belloli, C. Len, G. Mackenzie, G. Ronco, J.-P.
[18] C.J. Welch, W.R. Leonard Jr., J.O. DaSilva, M. Biba, J. Alba- Bonte, C. Vaccher, J. Chromatogr. A 2001, 943, 91 – 100.
neze-Walker, D.W. Henderson, B. Laing, D.J. Mathre, [31] L. Chen, R.A. Thompson, B.D. Johnson, J.M. Wyvratt, Chir-
LCGC North Am. 2005, 23(1), 16 – 29. ality 2002, 14, 393 – 399.
[19] E. Lesellier, C. West, A. Tchapla, J. Chromatogr. A 2003, [32] R.M. Wenslow Jr., T. Wang, Anal. Chem. 2001, 73, 4190 –
1018, 225 – 232. 4195.
J. Sep. Sci. 2005, 28, 619 – 626 www.jss-journal.de i 2005 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim](https://image.slidesharecdn.com/sfctandemcolumn-13250222261964-phpapp02-111227154523-phpapp02/85/Amgen2-8-320.jpg)