science 9 2nd quarter TOS.docx
•Download as DOCX, PDF•
0 likes•7 views
exam
Report
Share
Report
Share
Recommended
— Heat exchangers included in air conditioning systems for aircraft are produced by brazing stamped thin alloys sheets made of nickel-based alloys, Alloy 600 and Ni 201, or stainless steel, AISI 444. Separation metal sheets and locking bars of Alloy 625 are used to complete the system. The brazing filler metal, mainly composed of nickel, manganese, silicon and copper, is referred as BNi-8. In order to control brazing process, a good knowledge of both the brazing filler metal metallurgical behavior and of the interaction with the base metal is essential. The study of the brazing filler metal melting behavior in itself reveals that the melting point is highly dependent on the chemical composition and especially on silicon content. Microstructures analysis showed the presence of several phases with significant differences in terms of mechanical properties at a small scale which could induce local embrittlement. Interactions between the brazing filler metal and the different alloys constitutive of the assembly induce chemical composition evolutions related to the local configuration of the assembly. Dissolution and interdiffusion processes as well as chemical exchanges with the furnace environment occur. Finally, due to this set of phenomena, significant brazing defects can affect the mechanical integrity of the component.Metallurgical and mechanical behavior of brazed thin alloys sheets assemblies

Metallurgical and mechanical behavior of brazed thin alloys sheets assembliesInternational Multispeciality Journal of Health
Recommended
— Heat exchangers included in air conditioning systems for aircraft are produced by brazing stamped thin alloys sheets made of nickel-based alloys, Alloy 600 and Ni 201, or stainless steel, AISI 444. Separation metal sheets and locking bars of Alloy 625 are used to complete the system. The brazing filler metal, mainly composed of nickel, manganese, silicon and copper, is referred as BNi-8. In order to control brazing process, a good knowledge of both the brazing filler metal metallurgical behavior and of the interaction with the base metal is essential. The study of the brazing filler metal melting behavior in itself reveals that the melting point is highly dependent on the chemical composition and especially on silicon content. Microstructures analysis showed the presence of several phases with significant differences in terms of mechanical properties at a small scale which could induce local embrittlement. Interactions between the brazing filler metal and the different alloys constitutive of the assembly induce chemical composition evolutions related to the local configuration of the assembly. Dissolution and interdiffusion processes as well as chemical exchanges with the furnace environment occur. Finally, due to this set of phenomena, significant brazing defects can affect the mechanical integrity of the component.Metallurgical and mechanical behavior of brazed thin alloys sheets assemblies

Metallurgical and mechanical behavior of brazed thin alloys sheets assembliesInternational Multispeciality Journal of Health
In electrical discharge machining (EDM), Copper
and Graphite are commonly used as electrode (tool) materials.
EDM process is based on thermoelectric energy between an
electrode and workpiece. In this paper, the effect of electrode wear
rate (EWR) in 202 stainless steel is observed. Copper Electrode is
used as tool material and SS-202 is used as workpiece. Copper
electrode possess high structural integrity, so it can produce very
fine surface finishes, even without special polishing circuits.
EFFECT OF ELECTRODE WEAR RATE ON MACHINING OF STAINLESS STEEL – 202 USING COP...

EFFECT OF ELECTRODE WEAR RATE ON MACHINING OF STAINLESS STEEL – 202 USING COP...International Journal of Technical Research & Application
This presentation was provided by William Mattingly of the Smithsonian Institution, during the fifth segment of the NISO training series "AI & Prompt Design." Session Five: Named Entity Recognition with LLMs, was held on May 2, 2024.Mattingly "AI & Prompt Design: Named Entity Recognition"

Mattingly "AI & Prompt Design: Named Entity Recognition"National Information Standards Organization (NISO)
More Related Content
Similar to science 9 2nd quarter TOS.docx
In electrical discharge machining (EDM), Copper
and Graphite are commonly used as electrode (tool) materials.
EDM process is based on thermoelectric energy between an
electrode and workpiece. In this paper, the effect of electrode wear
rate (EWR) in 202 stainless steel is observed. Copper Electrode is
used as tool material and SS-202 is used as workpiece. Copper
electrode possess high structural integrity, so it can produce very
fine surface finishes, even without special polishing circuits.
EFFECT OF ELECTRODE WEAR RATE ON MACHINING OF STAINLESS STEEL – 202 USING COP...

EFFECT OF ELECTRODE WEAR RATE ON MACHINING OF STAINLESS STEEL – 202 USING COP...International Journal of Technical Research & Application
Similar to science 9 2nd quarter TOS.docx (20)
Iaetsd structural and electronic properties of doped

Iaetsd structural and electronic properties of doped
Pseudo potential DFT numerical approximation methods

Pseudo potential DFT numerical approximation methods
First principles study on structural, electronic, elastic and thermal propert...

First principles study on structural, electronic, elastic and thermal propert...
Preparation and properties of polycrystalline YBa2Cu3o7-x and Fe mixtures

Preparation and properties of polycrystalline YBa2Cu3o7-x and Fe mixtures
Effect of Temperature and Nickel Concentration on the Electrical and Dielectr...

Effect of Temperature and Nickel Concentration on the Electrical and Dielectr...
EFFECT OF ELECTRODE WEAR RATE ON MACHINING OF STAINLESS STEEL – 202 USING COP...

EFFECT OF ELECTRODE WEAR RATE ON MACHINING OF STAINLESS STEEL – 202 USING COP...
Recently uploaded
This presentation was provided by William Mattingly of the Smithsonian Institution, during the fifth segment of the NISO training series "AI & Prompt Design." Session Five: Named Entity Recognition with LLMs, was held on May 2, 2024.Mattingly "AI & Prompt Design: Named Entity Recognition"

Mattingly "AI & Prompt Design: Named Entity Recognition"National Information Standards Organization (NISO)
https://app.box.com/s/m9ehjx5owsaox9ykvb9qg3csa8a0jnoxTỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...Nguyen Thanh Tu Collection
https://app.box.com/s/71kthbth9ww0fyjrppmh1p2gasinqj5zĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH FORM 50 CÂU TRẮC NGHI...

ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH FORM 50 CÂU TRẮC NGHI...Nguyen Thanh Tu Collection
Recently uploaded (20)
Sternal Fractures & Dislocations - EMGuidewire Radiology Reading Room

Sternal Fractures & Dislocations - EMGuidewire Radiology Reading Room
Mattingly "AI & Prompt Design: Named Entity Recognition"

Mattingly "AI & Prompt Design: Named Entity Recognition"
Transparency, Recognition and the role of eSealing - Ildiko Mazar and Koen No...

Transparency, Recognition and the role of eSealing - Ildiko Mazar and Koen No...
When Quality Assurance Meets Innovation in Higher Education - Report launch w...

When Quality Assurance Meets Innovation in Higher Education - Report launch w...
UChicago CMSC 23320 - The Best Commit Messages of 2024

UChicago CMSC 23320 - The Best Commit Messages of 2024
TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...

TỔNG HỢP HƠN 100 ĐỀ THI THỬ TỐT NGHIỆP THPT TOÁN 2024 - TỪ CÁC TRƯỜNG, TRƯỜNG...
ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH FORM 50 CÂU TRẮC NGHI...

ĐỀ THAM KHẢO KÌ THI TUYỂN SINH VÀO LỚP 10 MÔN TIẾNG ANH FORM 50 CÂU TRẮC NGHI...
Including Mental Health Support in Project Delivery, 14 May.pdf

Including Mental Health Support in Project Delivery, 14 May.pdf
The Liver & Gallbladder (Anatomy & Physiology).pptx

The Liver & Gallbladder (Anatomy & Physiology).pptx
PSYPACT- Practicing Over State Lines May 2024.pptx

PSYPACT- Practicing Over State Lines May 2024.pptx
science 9 2nd quarter TOS.docx
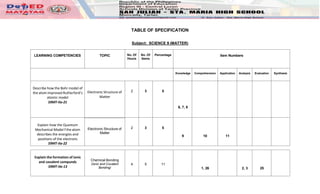
- 1. TABLE OF SPECIFICATION Subject: SCIENCE 9 (MATTER) LEARNING COMPETENCIES TOPIC No. Of Hours No. Of Items Percentage Item Numbers Knowledge Comprehension Application Analysis Evaluation Synthesis Describe how the Bohr model of the atom improved Rutherford’s atomic model S9MT-IIa-21 Electronic Structure of Matter 2 3 5 6, 7, 8 Explain how the Quantum Mechanical Model f the atom describes the energies and positions of the electrons S9MT-IIa-22 Electronic Structure of Matter 2 3 5 9 10 11 Explain the formation of ionic and covalent compunds S9MT-IIa-13 Chemical Bonding (Ionic and Covalent Bonding) 4 5 11 1, 26 2, 3 25
- 2. Recognize different types of compounds based on their properties such as melting point, hardness, polarity, and electrical and thermal conductivity S9MT-IIb-14 Chemical Bonding (Ionic and Covalent Bonding) 4 4 11 4 29, 38 28 Explain properties of metals in terms of their structure S9MT-IIc-d15 Chemical Bonding (Metallic Bonding) 4 4 11 5 39 36, 40 Explain how ions are formed S9MT-IIe-f16 Chemical Bonding (Metallic Bonding) 4 5 11 41 42 24 37 27 Explain how the structure of the carbon atom affects the type of bonds it forms S9MT-IIg-17 The Variety of Carbon Compounds 2 4 5 35 31 12 30 Recognize the general classes and uses of organic compounds S9MT-IIh-18 The Variety of Carbon Compounds 2 4 5 32, 13 33, 34
- 3. Use the mole concept to express mass of substances S9MT-IIi-19 The Mole Concept (Mass, Moles) 7 9 18 43 44, 49 16, 17 14, 21 22 15 Determine the percentage composition of a compound given its chemical formula and vice versa S9MT-IIj-20 The Mole Concept (Percentage Composition of a Compound) 7 9 18 48 45, 50 23, 46, 47 20 18, 19 TOTAL 38 50 100 Prepared by: PHILIP B. GALIZA T-III Reviewed and Checked: MARITA F. GARILLES OIC- HT Noted:
- 4. VILMA B. AGUAS, Ed.D Principal III