Power piont ch2 phase-transformation-in-metals (1)
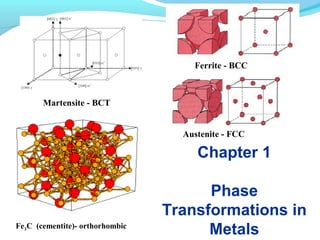
- 1. Chapter 1 Phase Transformations in MetalsFe3C (cementite)- orthorhombic Martensite - BCT Austenite - FCC Ferrite - BCC
- 2. Why do we study phase transformations? The tensile strength of an Fe-C alloy of eutectoid composition can be varied between 700-2000 MPa depending on HT process adopted. Desirable mechanical properties of a material can be obtained as a result of phase transformations using the right HTprocess. In order to design a HT for some alloy with desired RT properties, time & temperature dependencies of some phase transformations can be represented on modified phase diagrams. Based on this, we will learn: A. Phase transformations in metals B. Microstructure and property dependence in Fe-C alloy system C. Precipitation Hardening
- 3. Topics to be covered: Transformation rate Kinetics of Phase Transformation Nucleation: homogeneous, heterogeneous Free Energy, Growth Isothermal Transformations (TTT diagrams) Pearlite, Martensite, Spheroidite, Bainite Continuous Cooling Mechanical Behavior Precipitation Hardening
- 4. Phase Transformations Phase transformations – change in the number or character of phases. Simple diffusion-dependent No change in # of phases No change in composition Example: solidification of a pure metal, allotropic transformation, re- crystallization, grain growth More complicated diffusion-dependent Change in # of phases Change in composition Example: eutectoid reaction Diffusion-less Example: meta-stable phase : martensite
- 5. Phase Transformations -Stages Most phase transformations begin with the formation of numerous small particles of the new phase that increase in size until the transformation is complete. Nucleation is the process whereby nuclei (seeds) act as templates for crystal growth. 1. Homogeneous nucleation - nuclei form uniformly throughout the parent phase; requires considerable supercooling (typically 80- 300°C). 2. Heterogeneous nucleation - form at structural in-homogeneities (container surfaces, impurities, grain boundaries, dislocations) in liquid phase much easier since stable “nucleating surface” is already present; requires slight super-cooling (0.1-10ºC).
- 6. Supercooling During the cooling of a liquid, solidification (nucleation) will begin only after the temperature has been lowered below the equilibrium solidification (or melting) temperature Tm. This phenomenon is termed super-cooling or under-cooling. The driving force to nucleate increases as ∆T increases Small super-cooling slow nucleation rate - few nuclei - large crystals Large super-cooling rapid nucleation rate - many nuclei - small crystals
- 7. Kinetics of Solid State Reactions Transformations involving diffusion depend on time. Time is also necessary for the energy increase associated with the phase boundaries between parent and product phases. Moreover, nucleation, growth of the nuclei, formation of grains and grain boundaries and establishment of equilibrium take time. As a result we can say the transformation rate is a function of time. The fraction of reaction completed is measured as a function of time at constant T. Tranformation progress can be measured by microscopic examination or measuring a physical property (e.g., conductivity). The obtained data is plotted as fraction of the transformation versus logarithm of time.
- 8. 2 • Fraction transformed depends on time. fraction transformed time y = 1− e −ktn Avrami Eqn. • Transformation rate depends on T. 1 10 102 1040 50 100 135°C 119°C113°C102°C 88°C 43°C y (%) log (t) min Ex: recrystallization of Cu r = 1 t 0.5 = Ae −Q /RT activation energy • r often small: equil not possible y log (t) Fixed T 0 0.5 1 t 0.5 FRACTION OF TRANSFORMATION
- 9. Percent recrystallization as a function of time and at constant temperature for pure copper.
- 10. Transformations & Undercooling For transformation to occur, must cool to below 727°C Eutectoid transformation (Fe-Fe3C system): γ ⇒ α + Fe3C 0.76 wt% C 0.022 wt% C 6.7 wt% C Fe3C(cementite) 1600 1400 1200 1000 800 600 400 0 1 2 3 4 5 6 6.7 L γ (austenite) γ+L γ +Fe3C α +Fe3C L+Fe3C δ (Fe) C, wt% C 1148°C T(°C) α ferrite 727°C Eutectoid: Equil. Cooling: Ttransf. = 727ºC ∆T Undercooling by Ttransf. < 727°C 0.76 0.022
- 11. Generation of Isothermal Transformation Diagrams • The Fe-Fe3C system, for Co = 0.76 wt% C • A transformation temperature of 675°C. 100 50 0 1 102 104 T = 675°C%transformed time (s) 400 500 600 700 1 10 102 103 104 105 0%pearlite 100% 50% Austenite (stable) TE (727°C)Austenite (unstable) Pearlite T(°C) time (s) isothermal transformation at 675°C
- 12. Coarse pearlite formed at higher temperatures – relatively soft Fine pearlite formed at lower temperatures – relatively hard • Transformation of austenite to pearlite: γα α α α α α pearlite growth direction Austenite (γ) grain boundary cementite (Fe3C) Ferrite (α) γ • For this transformation, rate increases with ( ∆T) [Teutectoid – T ]. 675°C (∆T smaller) 0 50 %pearlite 600°C (∆T larger) 650°C 100 Diffusion of C during transformation α α γ γ α Carbon diffusion Eutectoid Transformation Rate ~ ∆T
- 13. Eutectoid Transformation Rate At T just below 727°C, very long times (on the order of 105 s) are required for 50% transformation and therefore transformation rate is slow. The transformation rate increases as T decreases, for example, at 540°C 3 s is required for 50% completion. This observation is in clear contradiction with the equation of This is because in T range of 540°C-727°C, the transformation rate is mainly controlled by the rate of pearlite nucleation and nucleation rate decreases with increasing T. Q in this equation is the activation energy for nucleation and it increases with T increase. It has been found that at lower T, the austenite decomposition is diffusion controlled and the rate behavior can be calculated using Q for diffusion which is independent of T. r = 1 t 0.5 = Ae −Q /RT activation energy
- 14. 5 • Reaction rate is a result of nucleation and growth of crystals. • Examples: % Pearlite 0 50 100 Nucleation regime Growth regime log (time)t50 Nucleation rate increases w/ ∆T Growth rate increases w/ T Nucleation rate high T just below TE T moderately below TE T way below TE Nucleation rate low Growth rate high γ γ γ pearlite colony Nucleation rate med . Growth rate med. Growth rate low Nucleation and Growth
- 15. Isothermal Transformation Diagrams solid curves are plotted: one represents the time required at each temperature for the start of the transformation; the other is for transformation completion. The dashed curve corresponds to 50% completion. The austenite to pearlite transformation will occur only if the alloy is supercooled to below the eutectoid temperature (727 C).˚ Time for process to complete depends on the temperature.
- 16. • Eutectoid iron-carbon alloy; composition, Co = 0.76 wt% C • Begin at T > 727˚C • Rapidly cool to 625˚C and hold isothermally. Isothermal Transformation Diagram Austenite-to-Pearlite
- 17. Transformations Involving Noneutectoid Compositions Hypereutectoid composition – proeutectoid cementite Consider C0 = 1.13 wt% C Fe3C(cementite) 1600 1400 1200 1000 800 600 400 0 1 2 3 4 5 6 6.7 L γ (austenite) γ +L γ +Fe3C α +Fe3C L+Fe3C δ (Fe) C, wt%C T(°C) 727°C ∆T 0.76 0.022 1.13
- 18. Strength DuctilityMartensite T Martensite bainite fine pearlite coarse pearlite spheroidite General Trends Possible Transformations
- 19. Coarse pearlite (high diffusion rate) and (b) fine pearlite - Smaller ∆T: colonies are larger - Larger ∆T: colonies are smaller
- 20. 10 103 105 time (s) 10-1 400 600 800 T(°C) Austenite (stable) 200 P B TE 0% 100% 50% A A Bainite: Non-Equilibrium Transformation Products elongated Fe3C particles in α-ferrite matrix diffusion controlled α lathes (strips) with long rods of Fe3C 100% bainite 100% pearlite Martensite Cementite Ferrite
- 21. Bainite Microstructure Bainite: formed as a result of transformation of austenite. Bainite consists of ferrite and cementite and diffusion processes take place as a result. This structure looks like needles or plates. There is no proeutectoid phase in bainite. Bainite consists of acicular (needle-like) ferrite with very small cementite particles dispersed throughout. The carbon content is typically greater than 0.1%. Bainite transforms to iron and cementite with sufficient time and temperature.
- 22. 10 Fe3C particles within an α-ferrite matrix diffusion dependent heat bainite or pearlite at temperature just below eutectoid for long times driving force – reduction of α-ferrite/Fe3C interfacial area Spheroidite: Nonequilibrium Transformation 10 103 105time (s)10-1 400 600 800 T(°C) Austenite (stable) 200 P B TE 0% 100% 50% A A Spheroidite 100% spheroidite 100% spheroidite
- 23. Pearlitic Steel partially transformed to Spheroidite
- 24. Design a heat treatment to produce the pearlite structure shown in Figure micrograph of the pearlite lamellae (x 2000) The effect of the austenite transformation temperature on the interlamellar spacing in pearlite. 7.14
- 25. A B C D E
- 26. A B C
- 27. The Martensitic Reaction and Tempering Martensiteis : is result of a diffusionless solid-state transfomation . •The growth rate is so high → nucleation is controlling step. • is an athermal transformation (i.e. the reaction depends only on the temperature, not on the time) FCC austenite upon quenching, In steels (< 0.2% C) nonequilibrium supersaturated BCC martensite structure In steels (>0.2% C) FCC austenite upon quenching, BCT (body-centered tetragonal) martensite BCT caused by the Carbon atoms in the (1/2, 0, 0) site being trapped during the transformation to the BCC %C(martensite) = %C (the starting austenite)
- 29. single phase body centered tetragonal (BCT) crystal structure BCT if C0 > 0.15 wt% C Diffusion-less transformation BCT few slip planes hard, brittle % transformation depends only on T of rapid cooling Martensite Formation 10 10 3 10 5 time (s)10 -1 400 600 800 T(°C) Austenite (stable) 200 P B TE 0% 100%50% A A M + A M + A M + A 0% 50% 90% Martensite needles Austenite
- 30. An micrograph of austenite that was polished flat and then allowed to transform into martensite. The different colors indicate the displacements caused when martensite forms.
- 31. Iron-carbon alloy with eutectoid composition. A: Austenite P: Pearlite B: Bainite M: Martensite
- 32. Other elements (Cr, Ni, Mo, Si and W) may cause significant changes in the positions and shapes of the TTT curves: Change transition temperature; Shift the nose of the austenite-to- pearlite transformation to longer times; Shift the pearlite and bainite noses to longer times (decrease critical cooling rate); Form a separate bainite nose; Effect of Adding Other Elements 4340 Steel plain carbon steel nose Plain carbon steel: primary alloying element is carbon.
- 33. Example 1: Iron-carbon alloy with eutectoid composition. Specify the nature of the final microstructure (% bainite, martensite, pearlite etc) for the alloy that is subjected to the following time–temperature treatments: Alloy begins at 760˚C and has been held long enough to achieve a complete and homogeneous austenitic structure. Treatment (a) Rapidly cool to 350 ˚C Hold for 104 seconds Quench to room temperature Bainite, 100%
- 34. Martensite, 100% Example 2: Iron-carbon alloy with eutectoid composition. Specify the nature of the final microstructure (% bainite, martensite, pearlite etc) for the alloy that is subjected to the following time–temperature treatments: Alloy begins at 760˚C and has been held long enough to achieve a complete and homogeneous austenitic structure. Treatment (b) Rapidly cool to 250 ˚C Hold for 100 seconds Quench to room temperature Austenite, 100%
- 35. Bainite, 50% Example 3: Iron-carbon alloy with eutectoid composition. Specify the nature of the final microstructure (% bainite, martensite, pearlite etc) for the alloy that is subjected to the following time–temperature treatments: Alloy begins at 760˚C and has been held long enough to achieve a complete and homogeneous austenitic structure. Treatment (c) Rapidly cool to 650˚C Hold for 20 seconds Rapidly cool to 400˚C Hold for 103 seconds Quench to room temperature Austenite, 100% Almost 50% Pearlite, 50% Austenite Final: 50% Bainite, 50% Pearlite
- 36. Continuous Cooling Transformation Diagrams Isothermal heat treatments are not the most practical due to rapidly cooling and constant maintenance at an elevated temperature. Most heat treatments for steels involve the continuous cooling of a specimen to room temperature. TTT diagram (dotted curve) is modified for a CCT diagram (solid curve). For continuous cooling, the time required for a reaction to begin and end is delayed. The isothermal curves are shifted to longer times and lower temperatures.
- 37. Moderately rapid and slow cooling curves are superimposed on a continuous cooling transformation diagram of a eutectoid iron-carbon alloy. The transformation starts after a time period corresponding to the intersection of the cooling curve with the beginning reaction curve and ends upon crossing the completion transformation curve. Normally bainite does not form when an alloy is continuously cooled to room temperature; austenite transforms to pearlite before bainite has become possible. The austenite-pearlite region (A--- B) terminates just below the nose. Continued cooling (below Mstart) of austenite will form martensite.
- 38. For continuous cooling of a steel alloy there exists a critical quenching rate that represents the minimum rate of quenching that will produce a totally martensitic structure. This curve will just miss the nose where pearlite transformation begins
- 39. Continuous cooling diagram for a 4340 steel alloy and several cooling curves superimposed. This demonstrates the dependence of the final microstructure on the transformations that occur during cooling. Alloying elements used to modify the critical cooling rate for martensite are chromium, nickel, molybdenum, manganese, silicon and tungsten.
- 40. Mechanical Properties Hardness Brinell, Rockwell Yield Strength Tensile Strength Ductility % Elongation Effect of Carbon Content
- 41. Mechanical Properties: Influence of Carbon Content C0 > 0.76 wt% C Hypereutectoid Pearlite (med) Cementite (hard) C0 < 0.76 wt% C Hypoeutectoid Pearlite (med) ferrite (soft)
- 42. Mechanical Properties: Fe-C System
- 43. Example: P10.37 For a eutectoid steel, describe isothermal heat treatments that would be required to yield specimens having the following Brinell Hardnesses: 1. 180 HB 2. 220 HB 3. 500 HB
- 44. Martensite is hard but also very brittle so that it can not be used in most of the applications. Any internal stress that has been introduced during quenching has a weakening effect. The ductility and toughness of the material can be enhanced by heat treatment called tempering. This also helps to release any internal stress. Tempering is performed by heating martensite to a T below eutectoid temperature (250°C-650°C) and keeping at that T for specified period of time. The formation of tempered martensite is by diffusion. Tempered Martensite
- 45. Tempered martensite is less brittle than martensite; tempered at 594 °C. Tempering reduces internal stresses caused by quenching. The small particles are cementite; the matrix is α-ferrite. US Steel Corp. Tempered Martensite 4340 steel
- 46. Tempered martensite may be nearly as hard and strong as martensite, but with substantially enhanced ductility and toughness. The hardness and strength may be due to large area of phase boundary per unit volume of the material. The phase boundary acts like a barrier for dislocations. The continuous ferrite phase in tempered martensite adds ductility and toughness to the material. The size of the cementite particles is important factor determining the mechanical behavior. As the cementite particle size increases, material becomes softer and weaker. The temperature of tempering determines the cementite particle size. Since martensite-tempered martensite transformation involves diffusion, Increasing T will accelerate the diffusion and rate of cementite particle growth and rate of softening as a result. Tempered Martensite
- 47. Hardness as a function of carbon concentration for steels
- 48. Hardness versus tempering time for a water-quenched eutectoid plain carbon steel (1080); room temperature. Rockwell C and Brinell Hardness
- 50. Example: P10.25 Briefly describe the simplest continuous cooling heat treatment procedure that would be used in converting a 4340 steel from microstructure to another. 1. (Martensite + Ferrite + Bainite) to (Martensite + Ferrite + pearlite + Bainite) 2. (Martensite + Ferrite + Bainite) to (spheroidite) 3. (Martensite + Ferrite + Bainite) to (tempered Martensite)
- 51. Precipitation Hardening The strength and hardness of some metal alloys may be improved by the formation of extremely small, uniformly dispersed particles (precipitates) of a second phase within the original phase matrix. Other alloys that can be precipitation hardened or age hardened: Copper-Beryllium (Cu-Be) Copper-Tin (Cu-Sn) Magnesium-Aluminum (Mg-Al) Aluminum-Copper (Al-Cu) High-strength Aluminum alloys
- 52. Criteria: Maximum solubility of 1 component in the other (M); Solubility limit that rapidly decreases with decrease in temperature (M→N). Process: Solution Heat Treatment – first heat treatment where all solute atoms are dissolved to form a single-phase solid solution. Heat to T0 and dissolve B phase. Rapidly quench to T1 Nonequilibrium state (α phase solid solution supersaturated with B atoms; alloy is soft, weak-no ppts). Phase Diagram for Precipitation Hardened Alloy
- 53. The supersaturated α solid solution is usually heated to an intermediate temperature T2 within the α+β region (diffusion rates increase). The β precipitates (PPT) begin to form as finely dispersed particles. This process is referred to as aging. After aging at T2, the alloy is cooled to room temperature. Strength and hardness of the alloy depend on the ppt temperature (T2) and the aging time at this temperature. Precipitation Heat Treatment – the 2nd stage
- 54. 0 10 20 30 40 50 wt% Cu L α+Lα α+θ θ θ+L 300 400 500 600 700 (Al) T(°C) composition range available for precipitation hardening CuAl2 A Precipitation Hardening • Particles impede dislocation motion. • Ex: Al-Cu system • Procedure: -- Pt B: quench to room temp. (retain α solid solution) -- Pt C: reheat to nucleate small θ particles within α phase. Temp. Time -- Pt A: solution heat treat (get α solid solution) Pt A (solution heat treat) B Pt B C Pt C (precipitate θ) At room temperature the stable state of an aluminum-copper alloy is an aluminum-rich solid solution (α) and an intermetallic phase with a tetragonal crystal structure having nominal composition CuAl2 (θ).
- 55. Precipitation Heat Treatment – the 2nd stage PPT behavior is represented in the diagram: With increasing time, the hardness increases, reaching a maximum (peak), then decreasing in strength. The reduction in strength and hardness after long periods is overaging (continued particle growth). Small solute-enriched regions in a solid solution where the lattice is identical or somewhat perturbed from that of the solid solution are called Guinier- Preston zones.
- 56. 24 • Hard precipitates are difficult to shear. Ex: Ceramics in metals (SiC in Iron or Aluminum). Large shear stress needed to move dislocation toward precipitate and shear it. Side View Top View Slipped part of slip plane Unslipped part of slip plane S Dislocation “advances” but precipitates act as “pinning” sites with spacing S. precipitate • Result: σy ~ 1 S PRECIPITATION STRENGTHENING
- 57. Several stages in the formation of the equilibrium PPT (θ) phase. (a)supersaturated α solid solution; (b)transition (θ”) PPT phase; (c)equilibrium θ phase within the α matrix phase.
- 58. • 2014 Al Alloy: • TS peak with precipitation time. • Increasing T accelerates process. Influence of Precipitation Heat Treatment on Tensile Strength (TS), %EL precipitation heat treat time tensilestrength(MPa) 200 300 400 100 1min 1h 1day 1mo 1yr 204°C non-equil. solidsolution manysmall precipitates“aged” fewerlarge precipitates “overaged” 149°C • %EL reaches minimum with precipitation time. %EL(2insample) 10 20 30 0 1min 1h 1day 1mo 1yr 204°C 149°C precipitation heat treat time
- 59. Effects of Temperature Characteristics of a 2014 aluminum alloy (0.9 wt% Si, 4.4 wt% Cu, 0.8 wt% Mn, 0.5 wt% Mg) at 4 different aging temperatures.
- 60. Alloys that experience significant precipitation hardening at room temp and after short periods must be quenched to and stored under refrigerated conditions. Several aluminum alloys that are used for rivets exhibit this behavior. They are driven while still soft, then allowed to age harden at the normal room temperature.