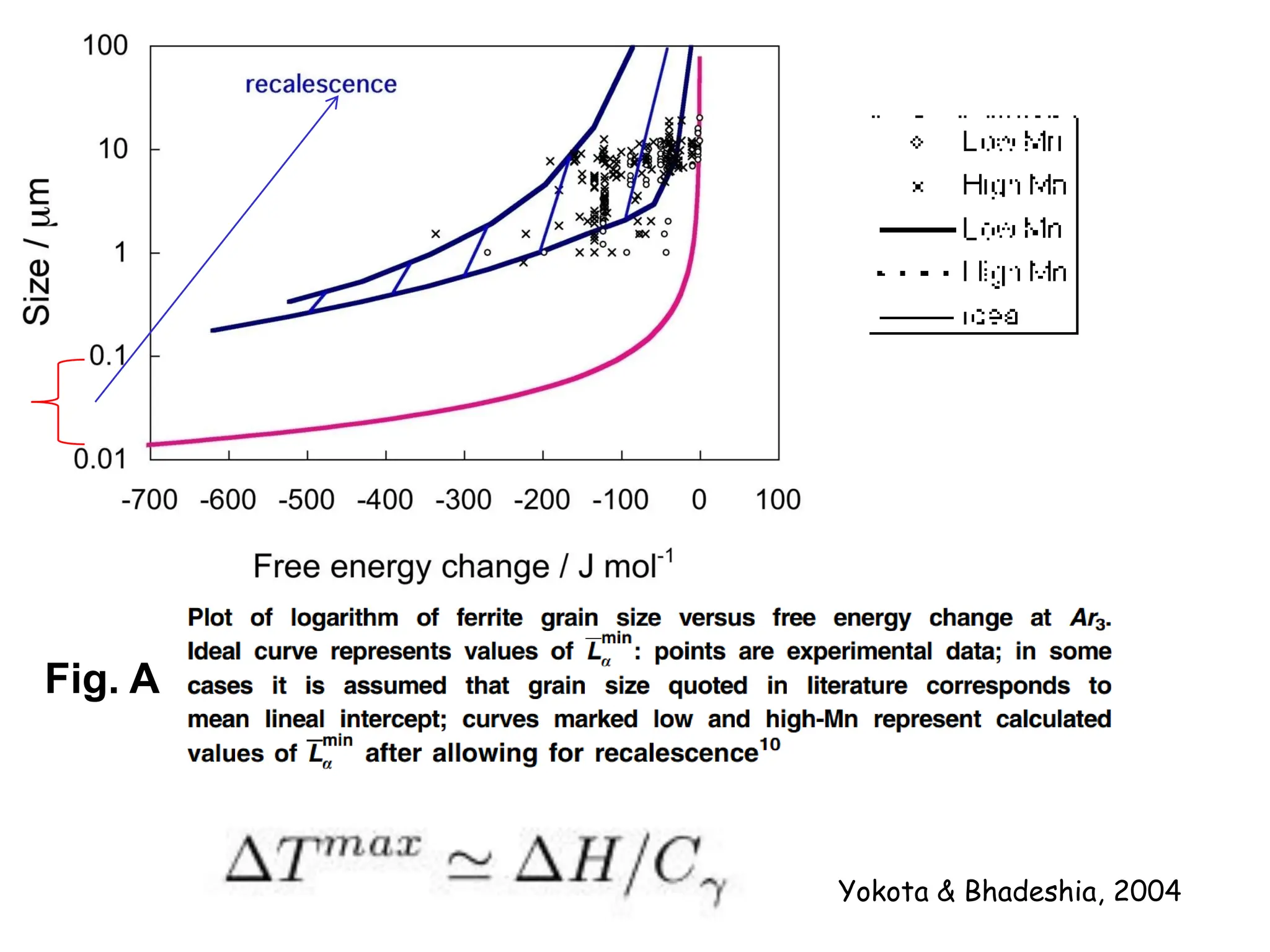
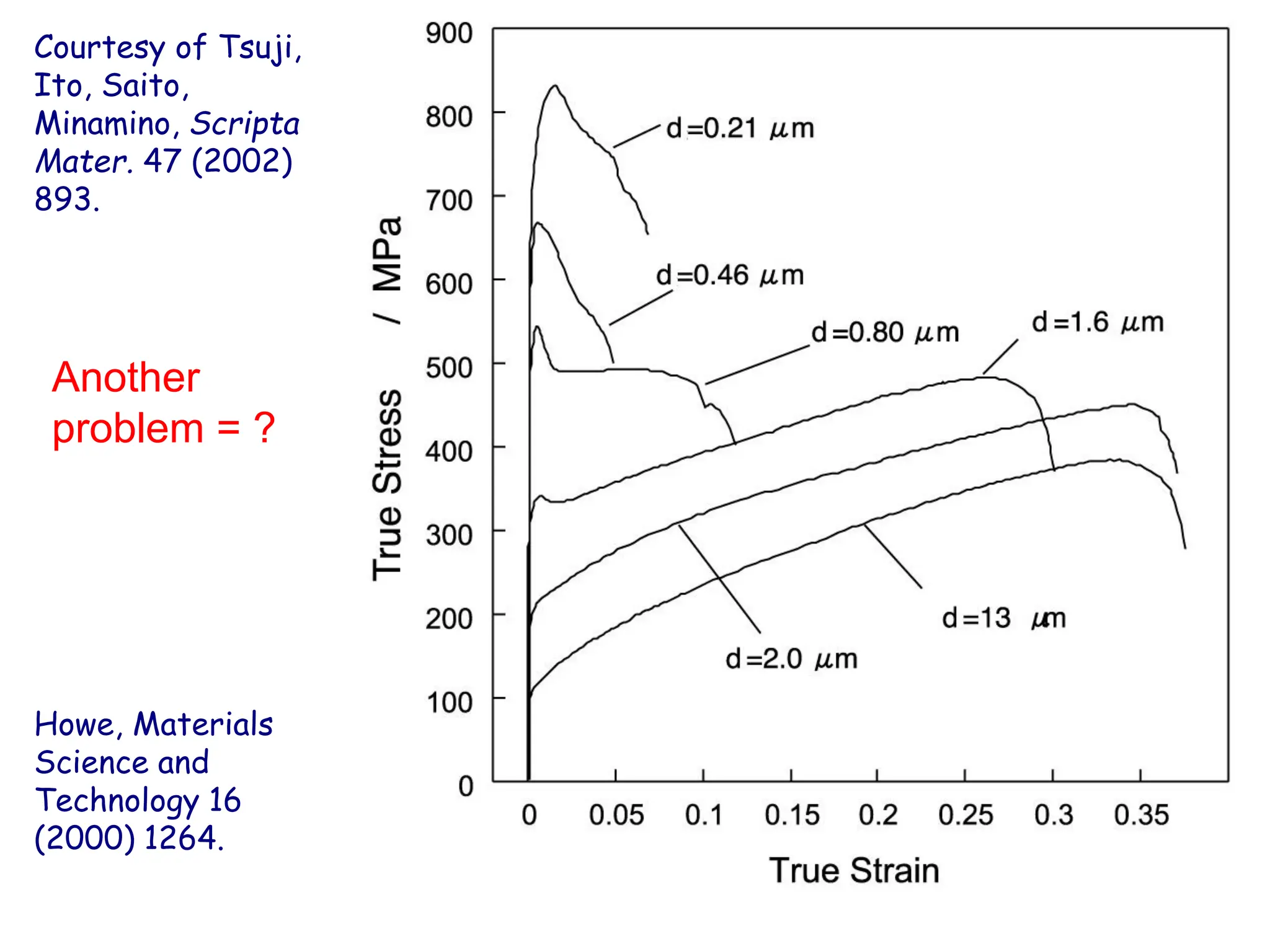
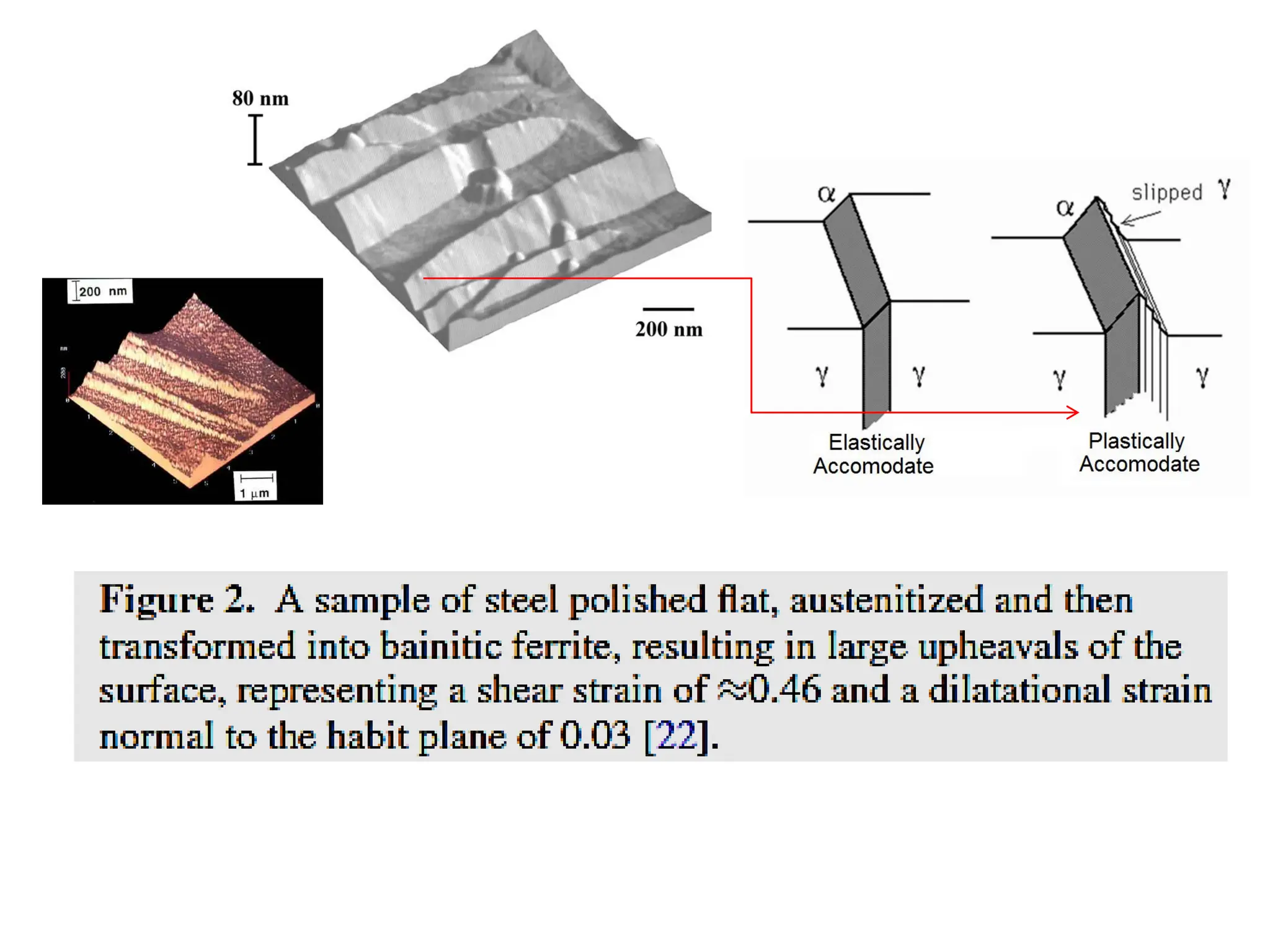
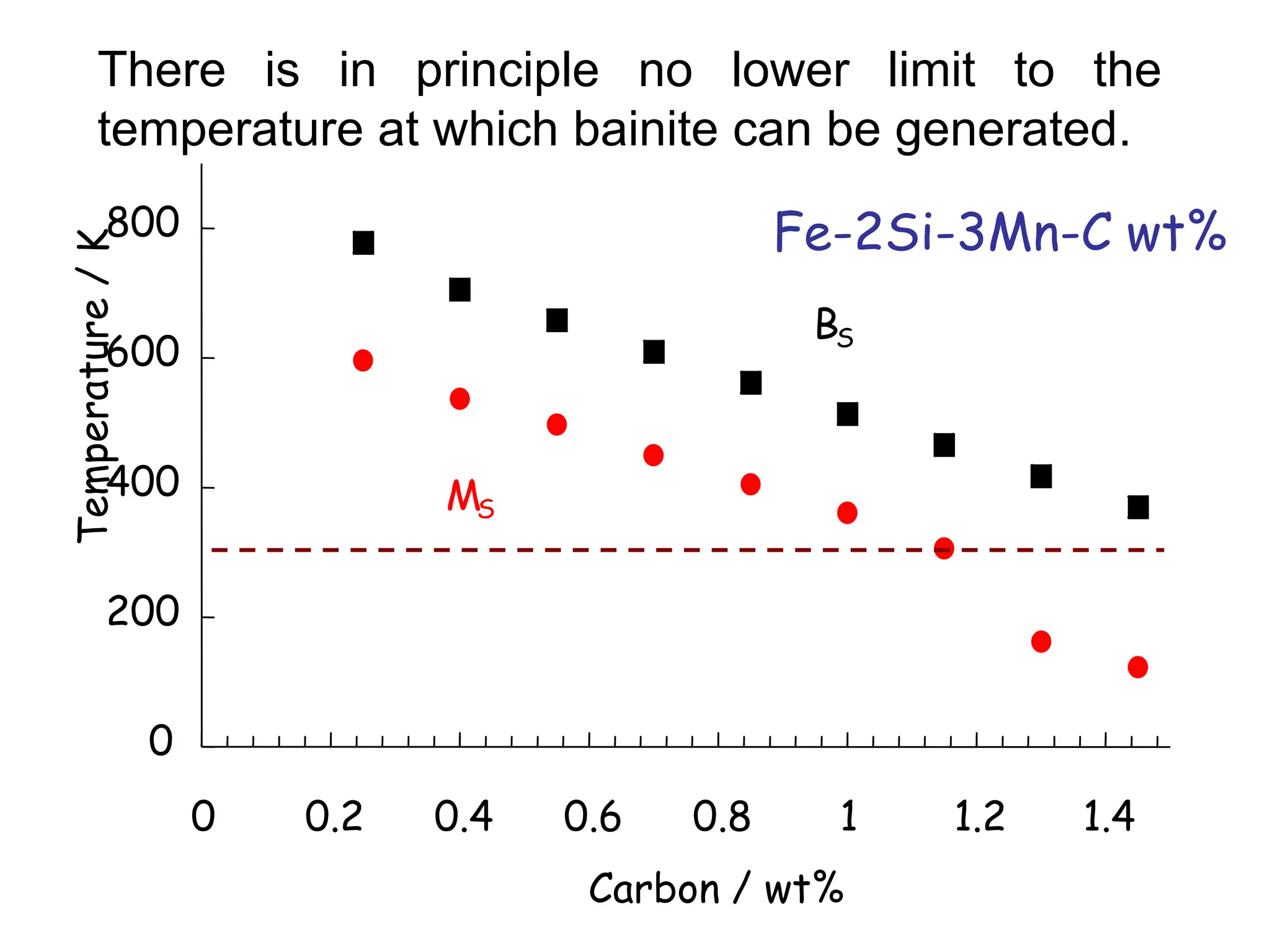
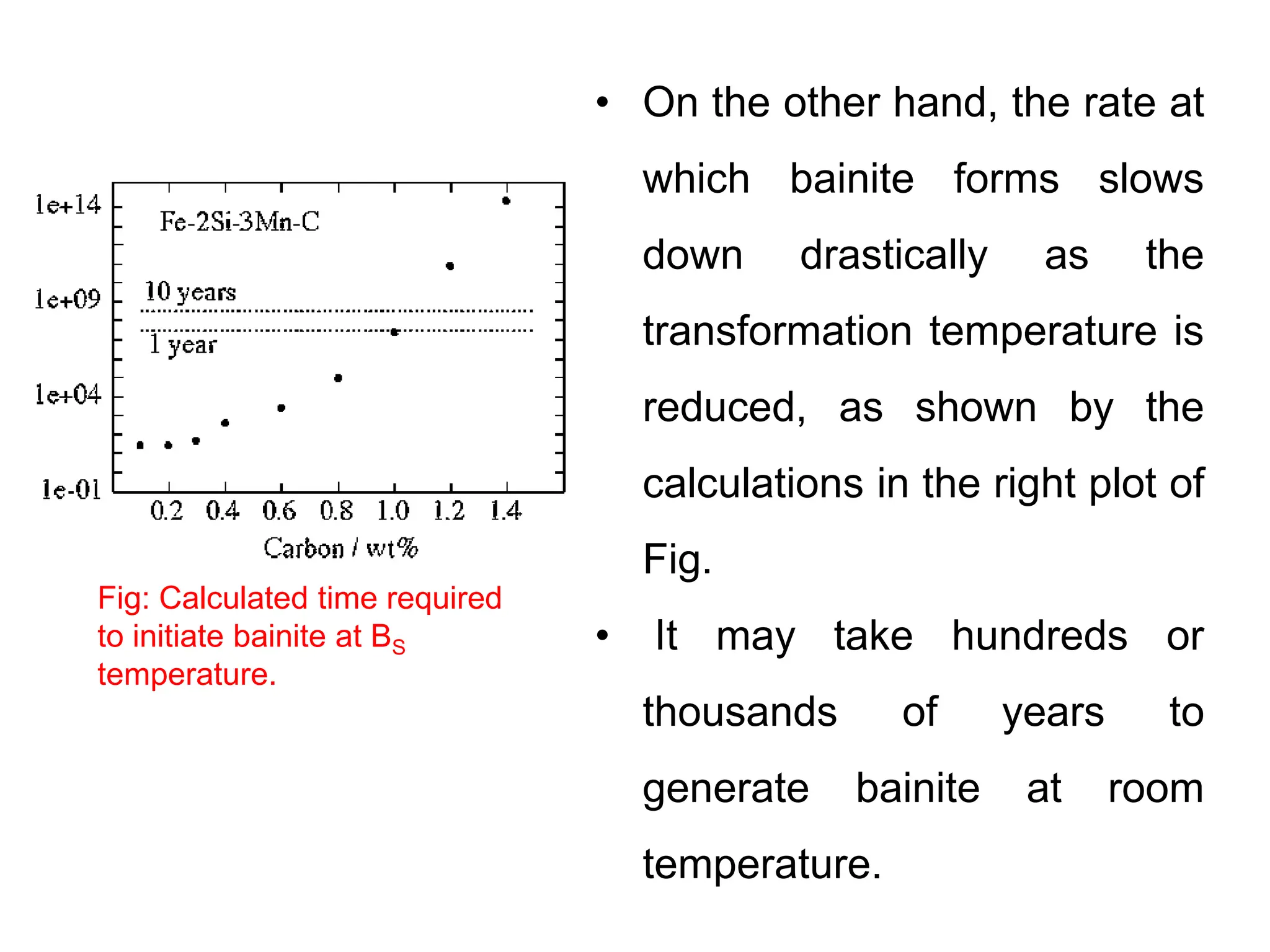
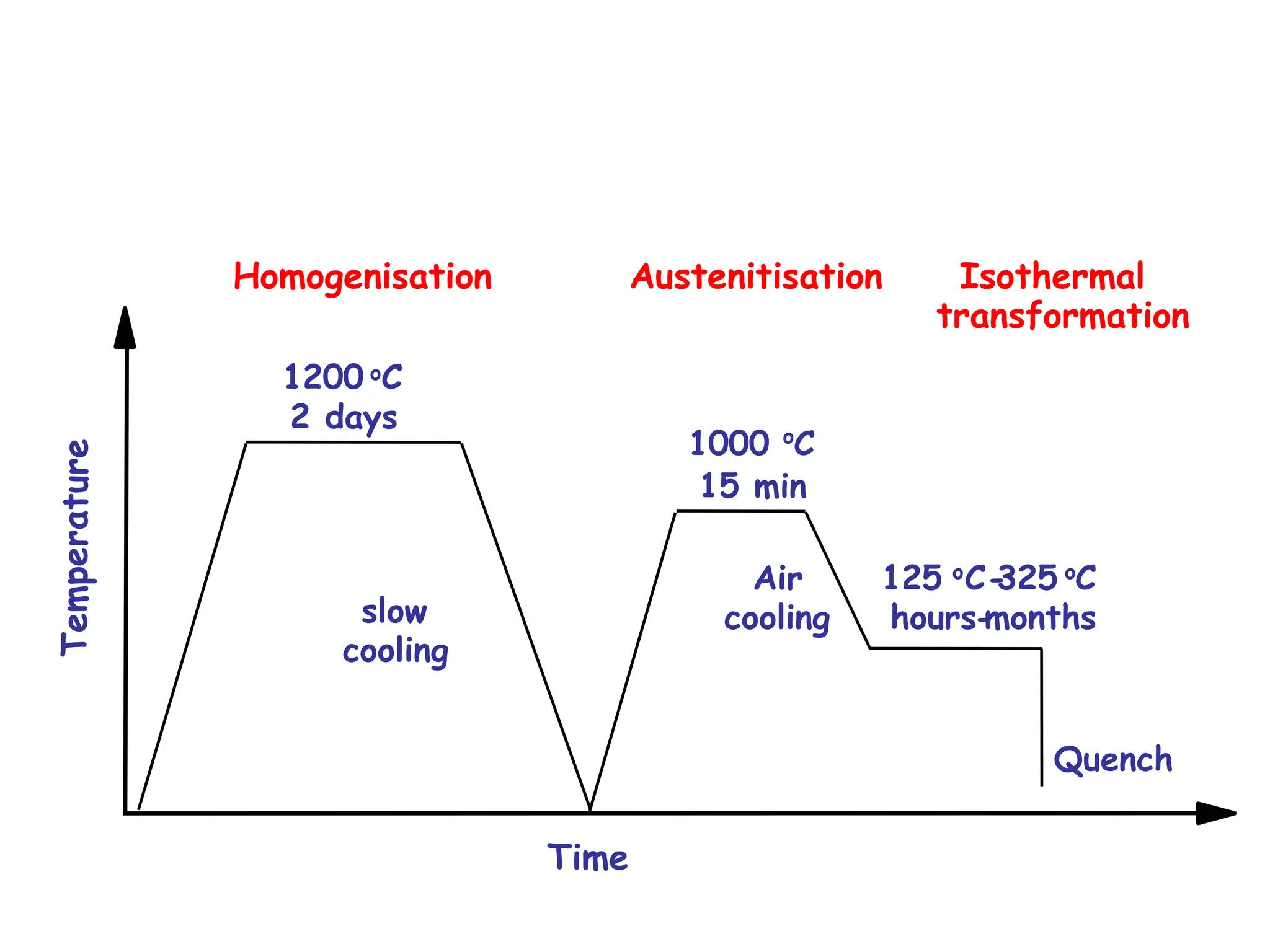
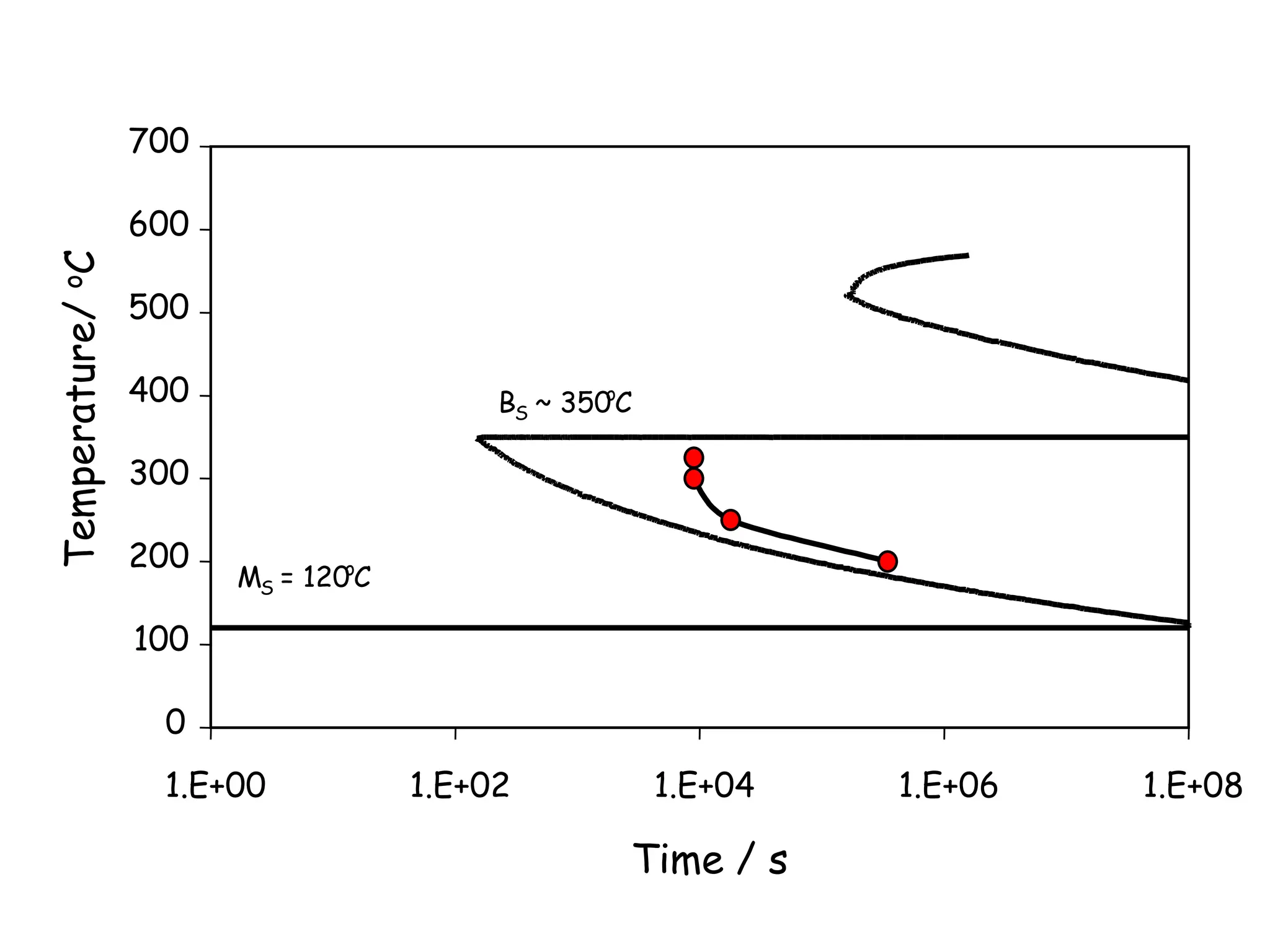
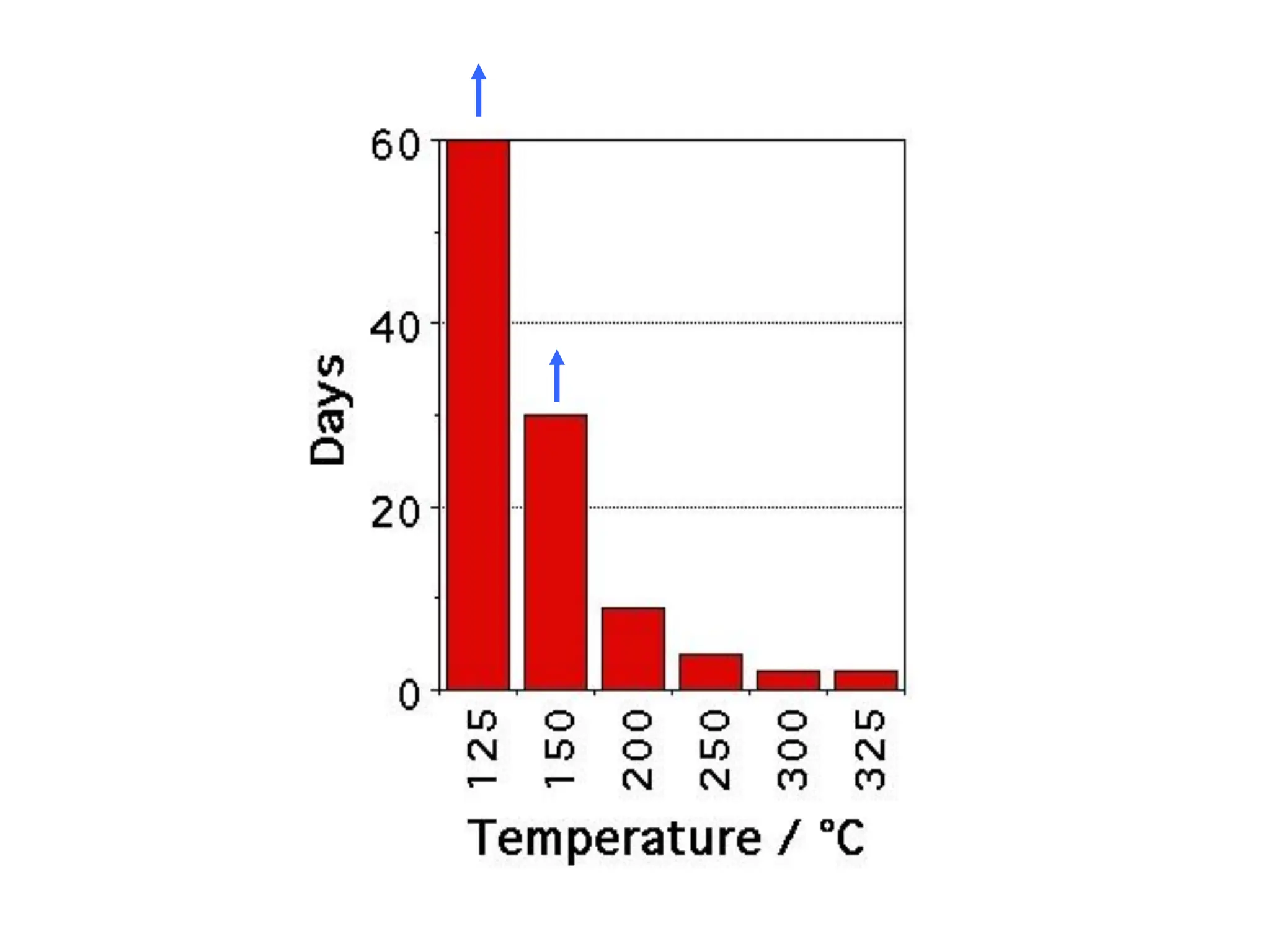
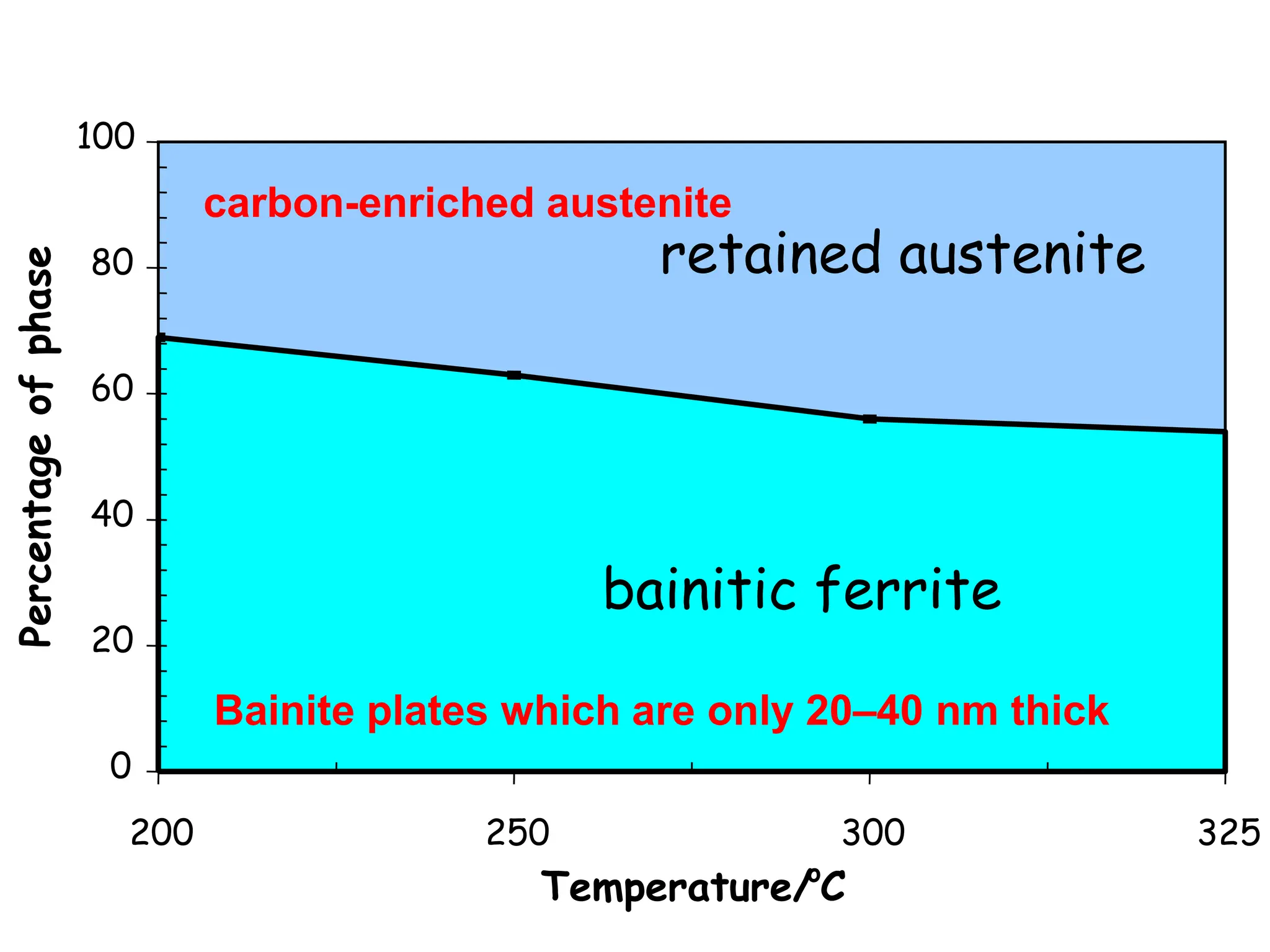
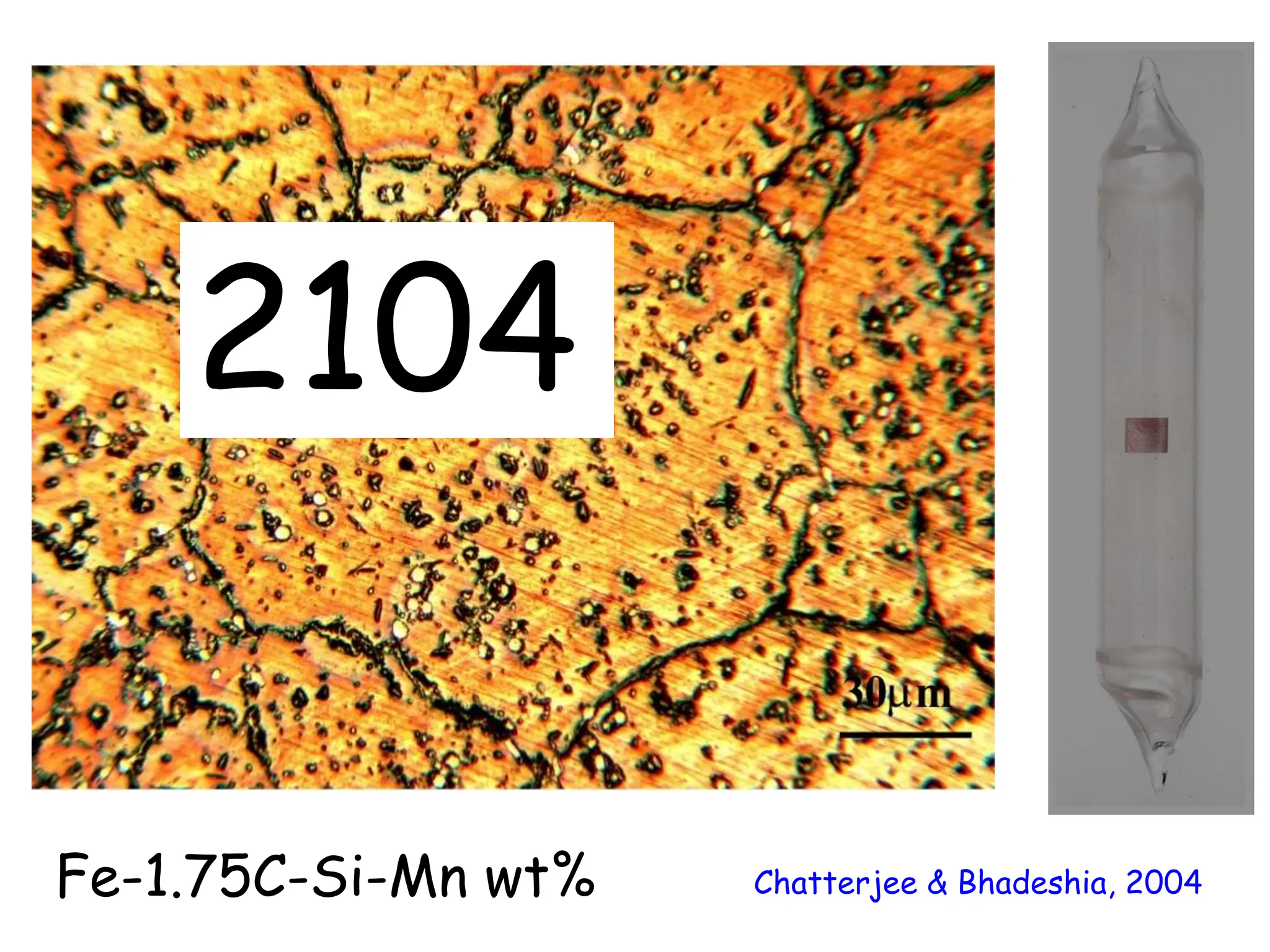
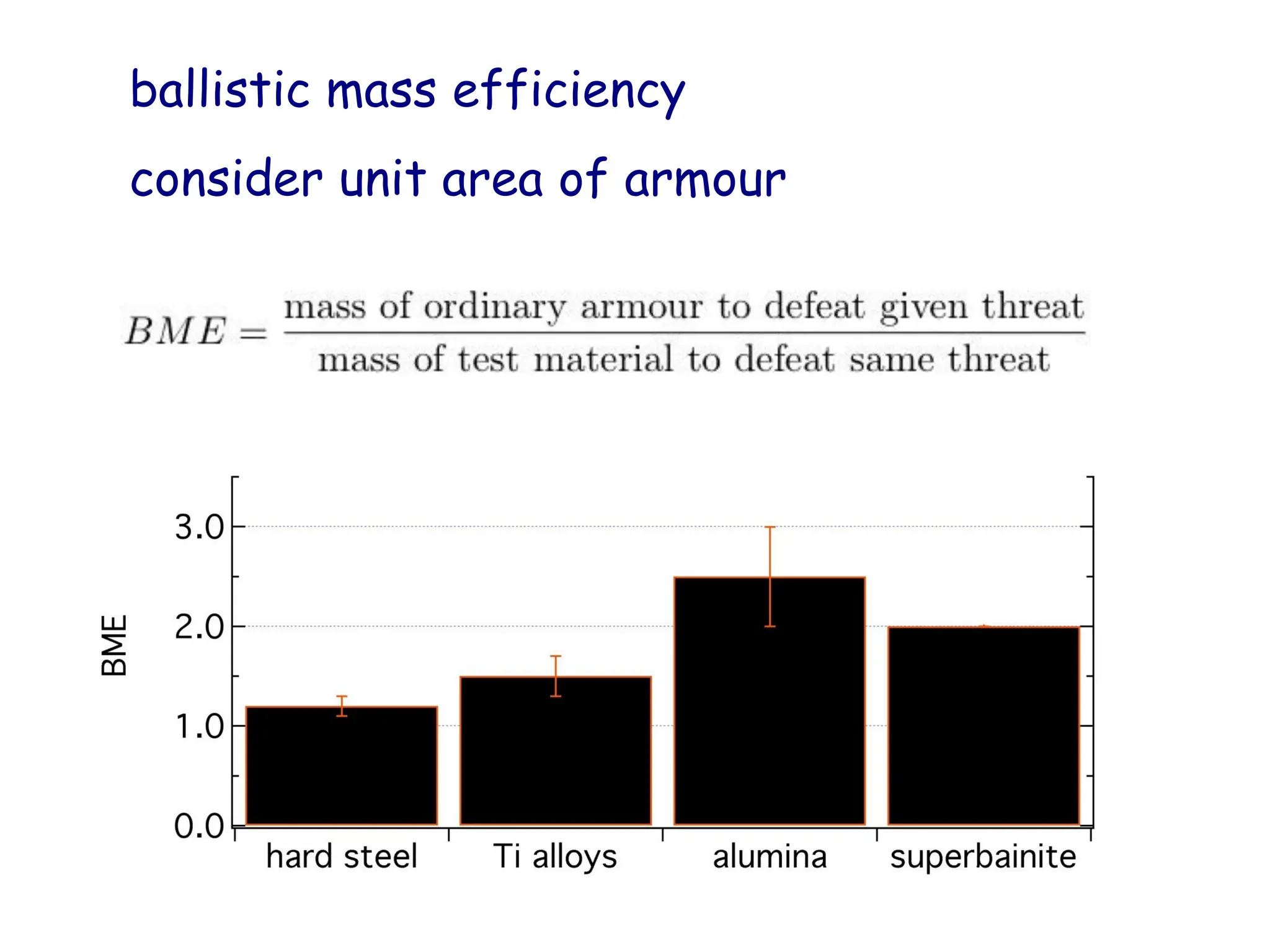
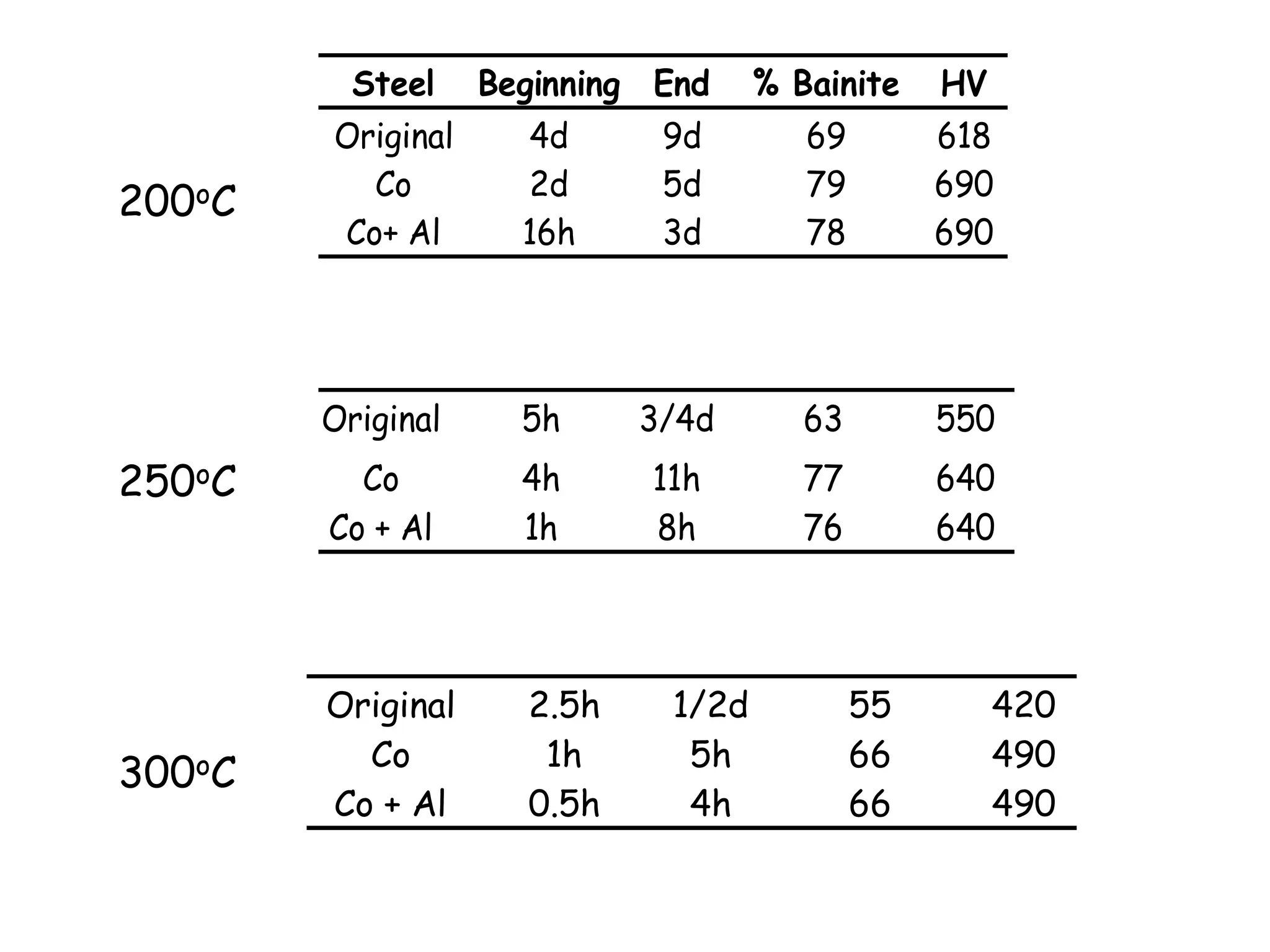
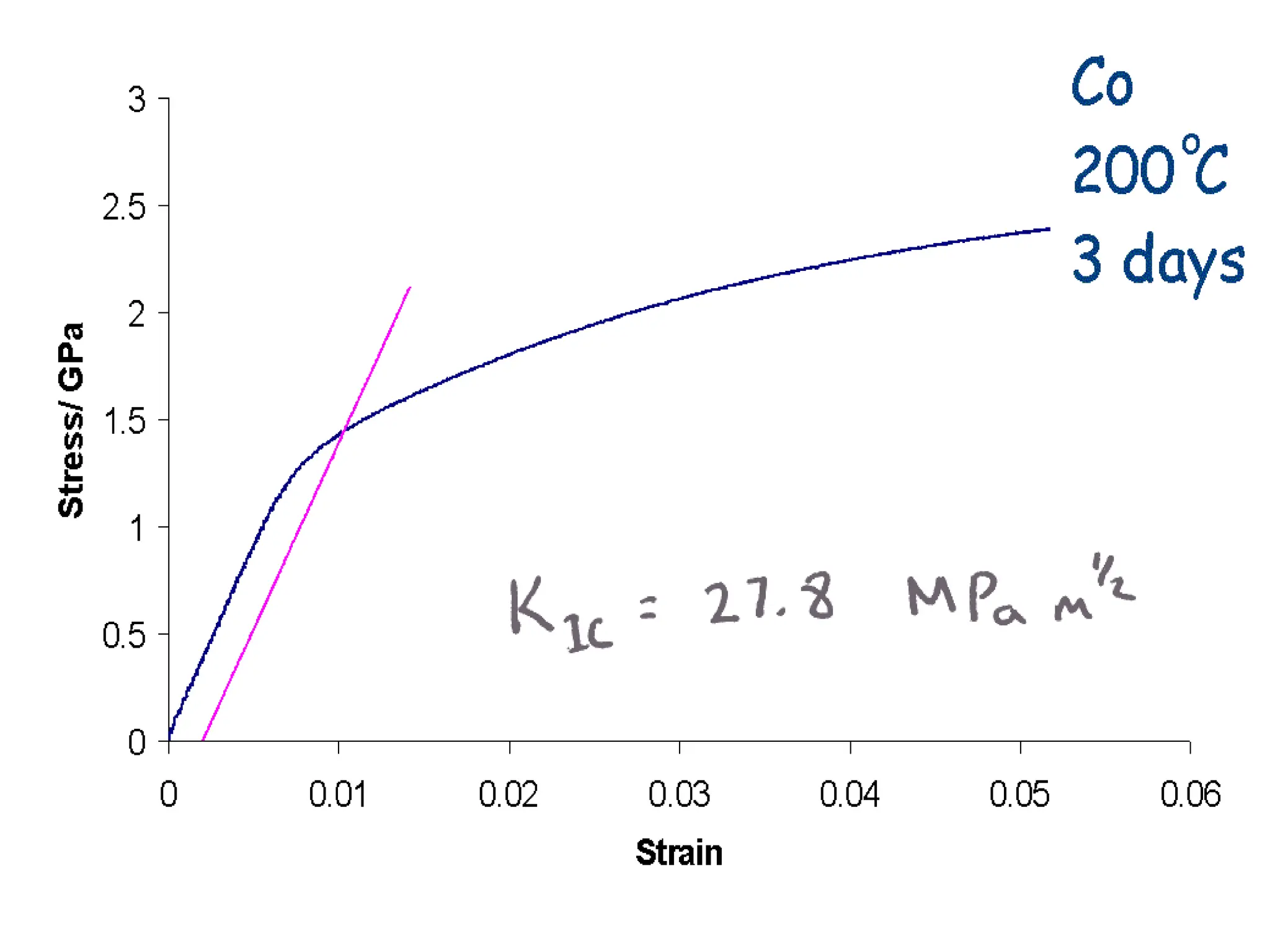
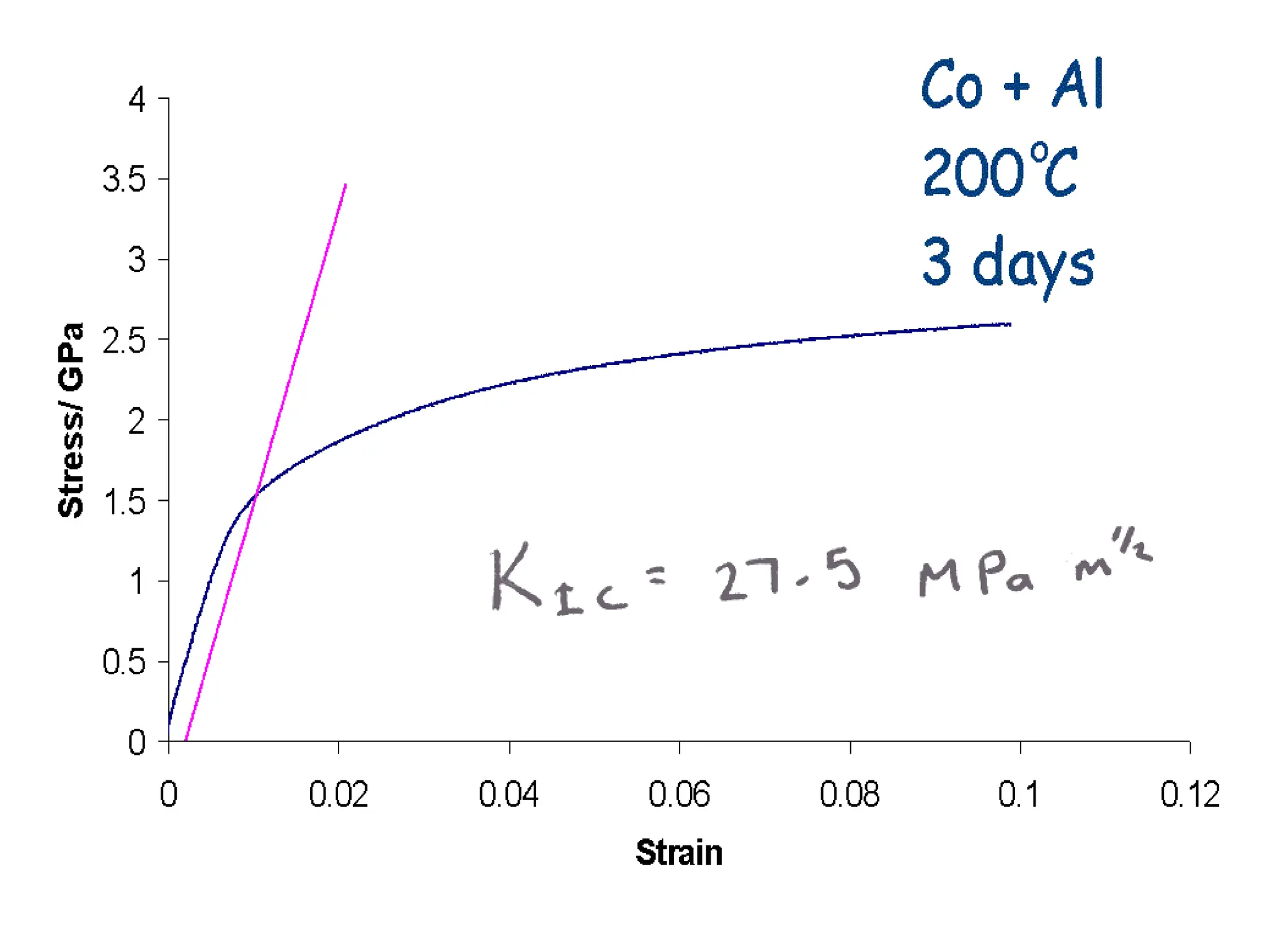
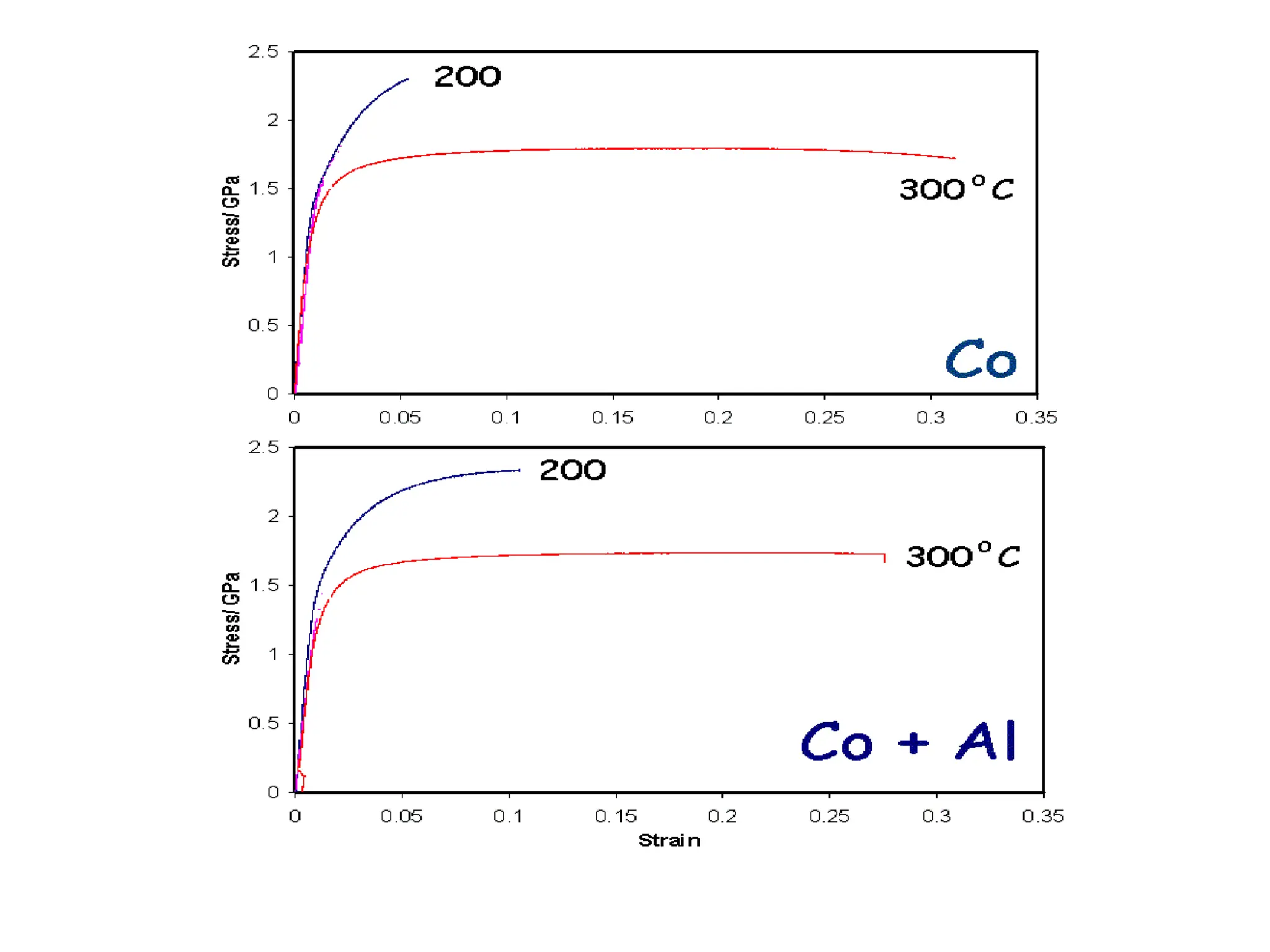
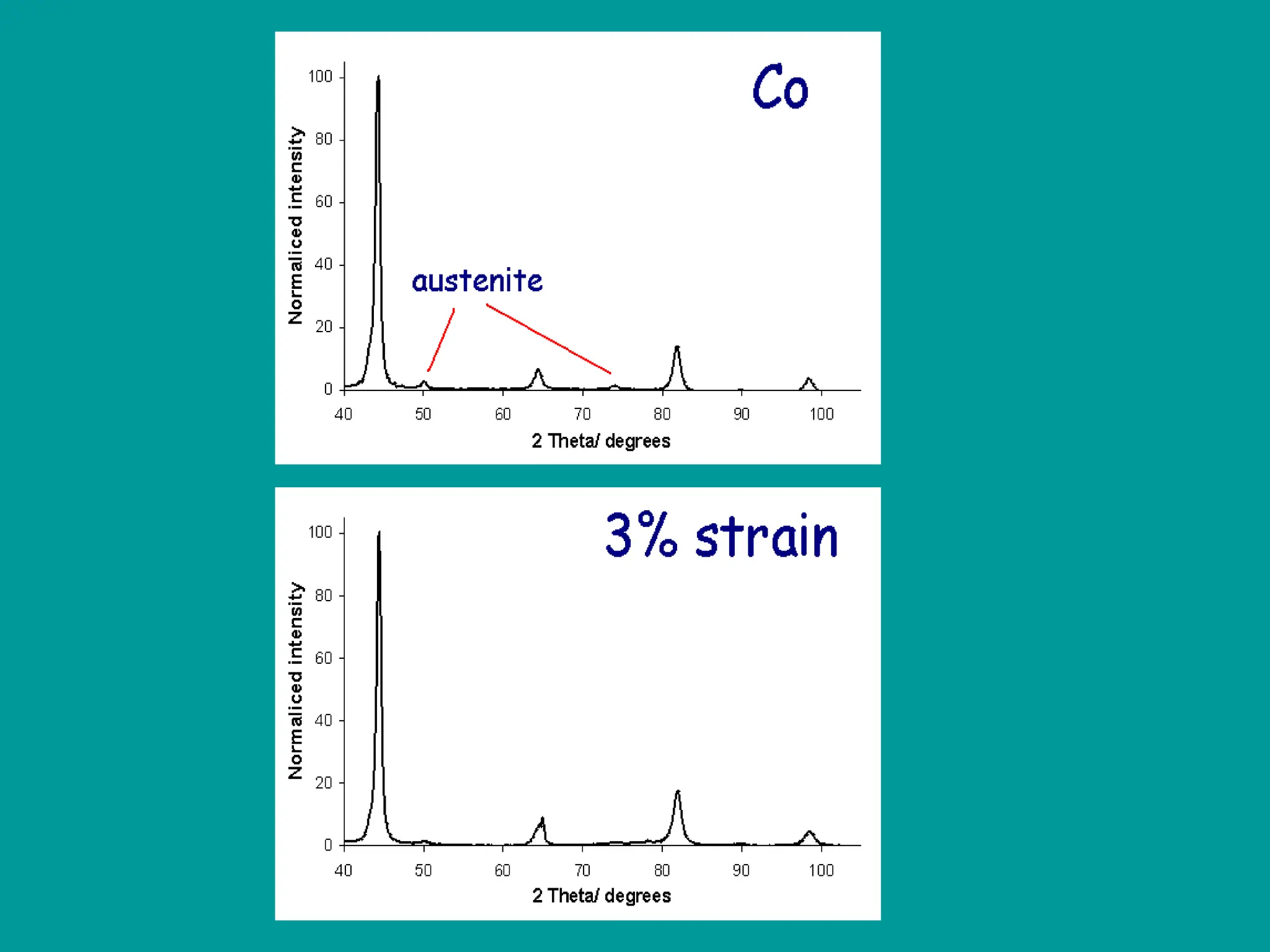
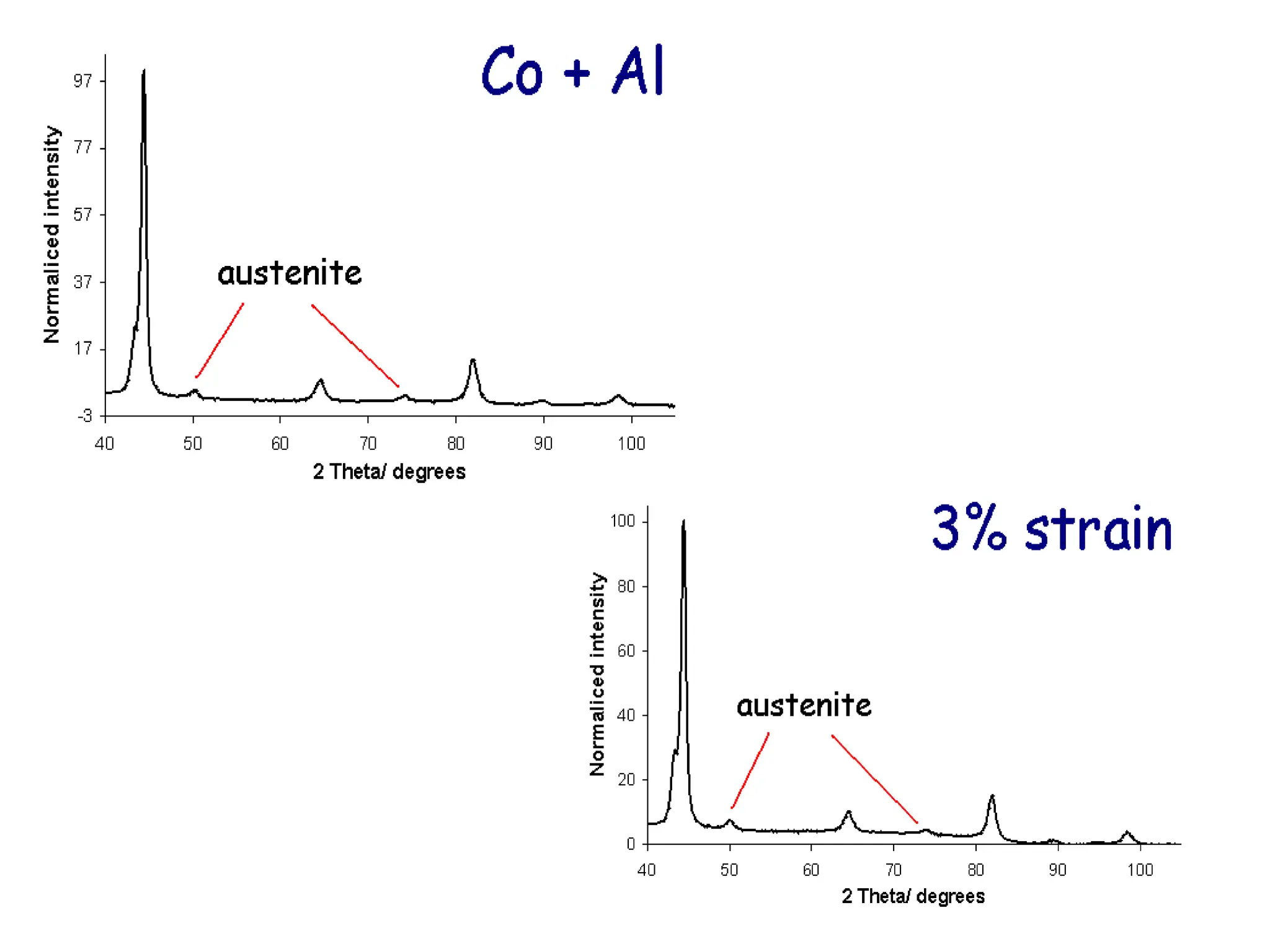
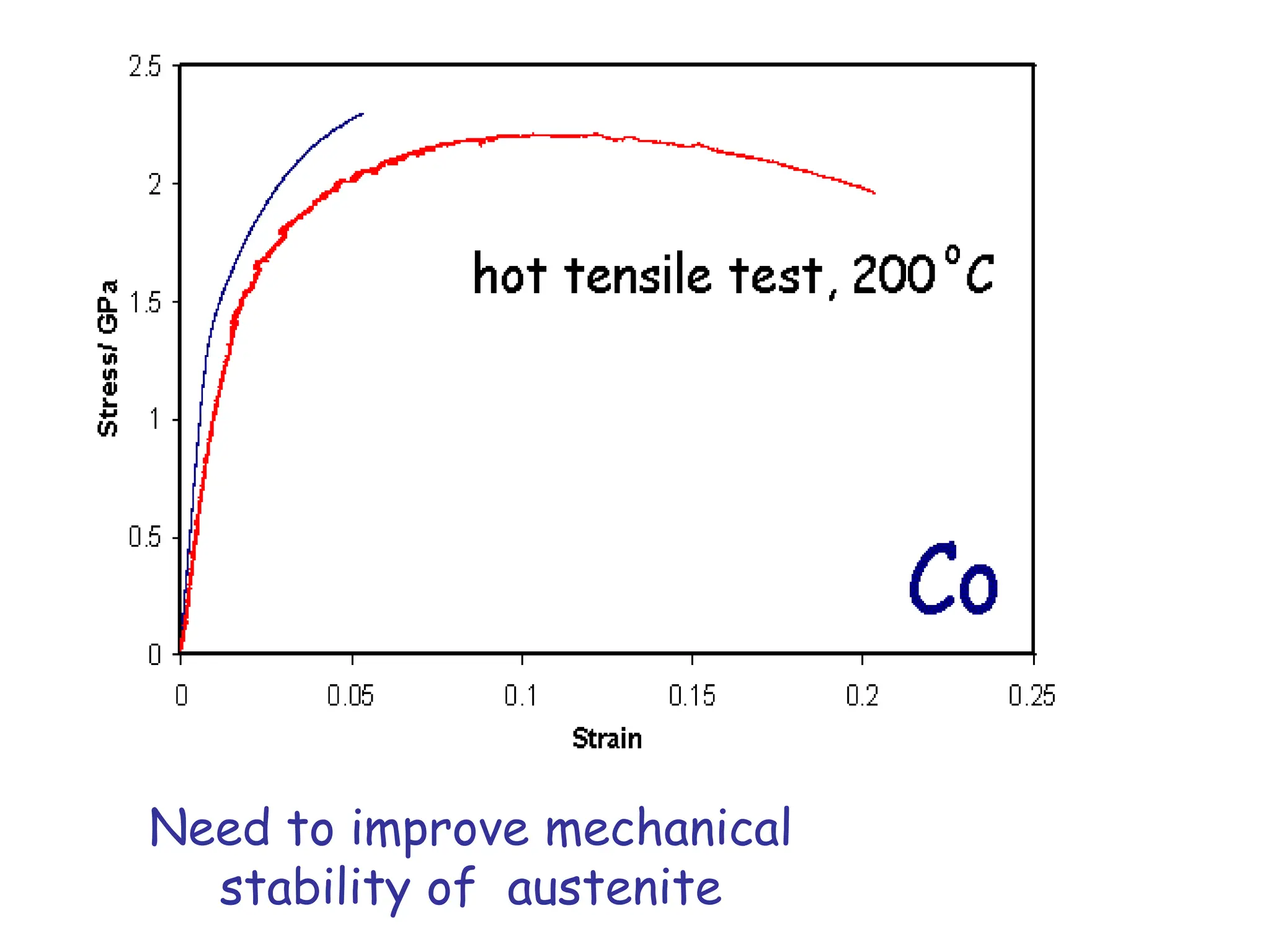
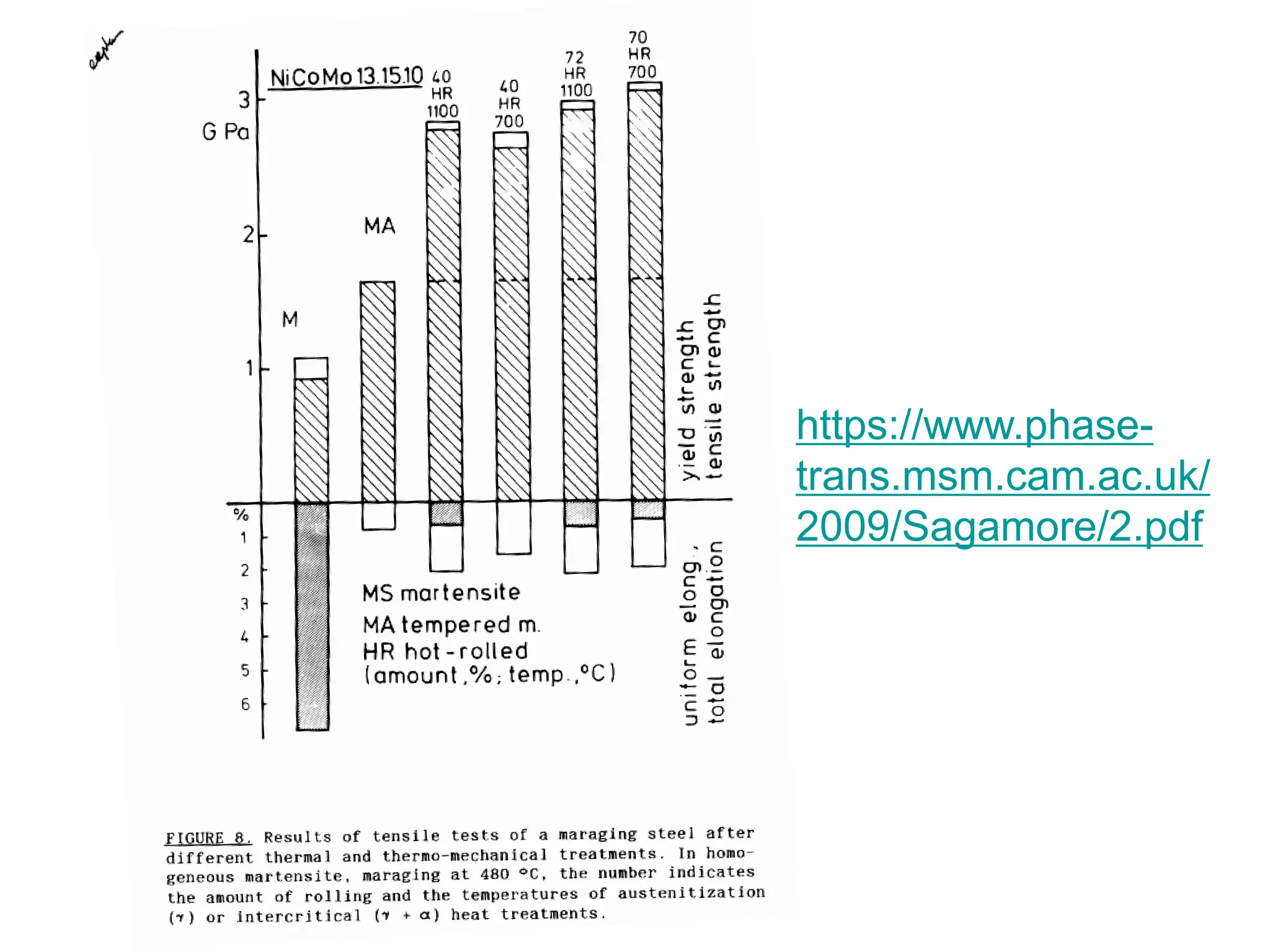
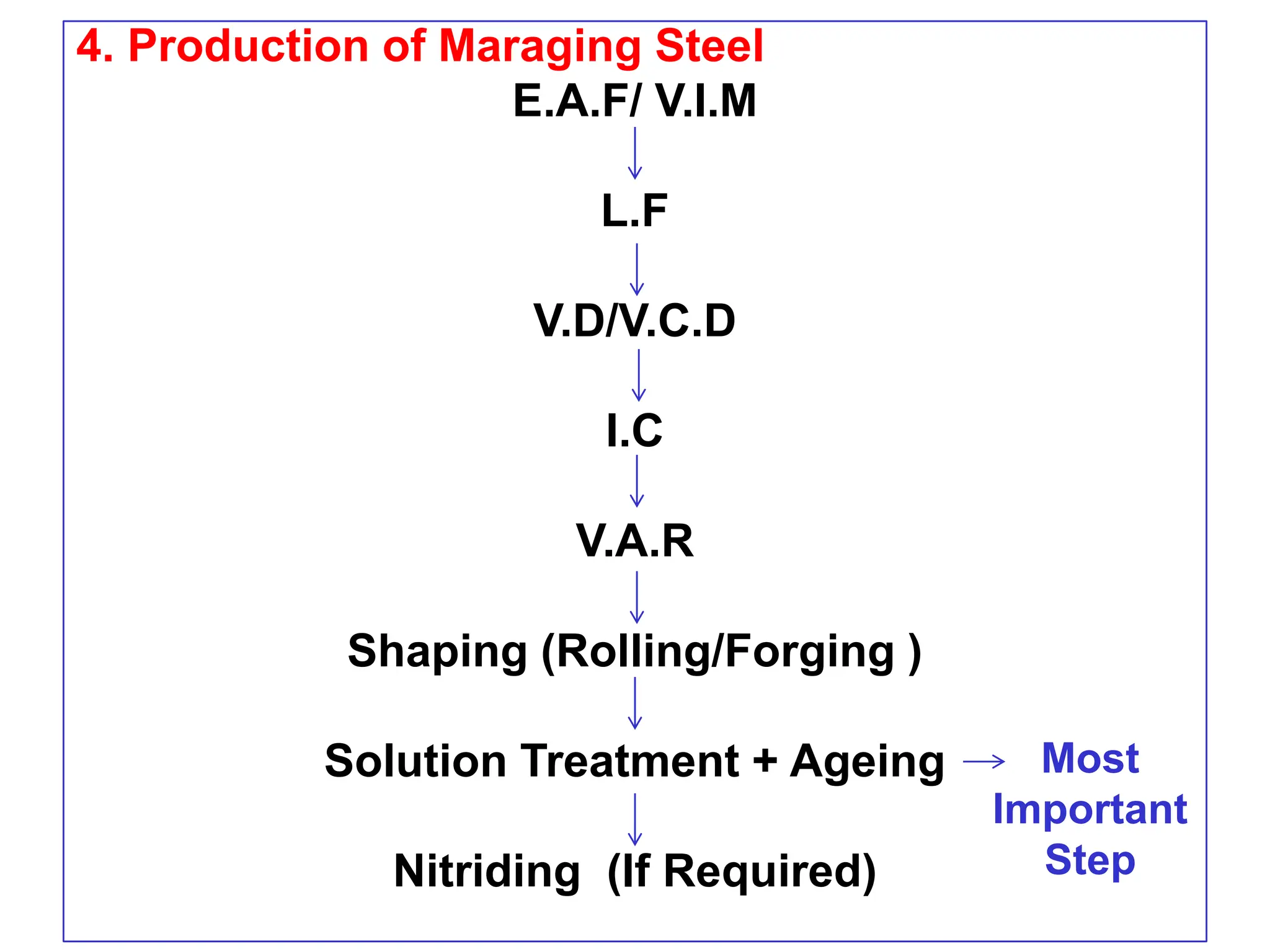
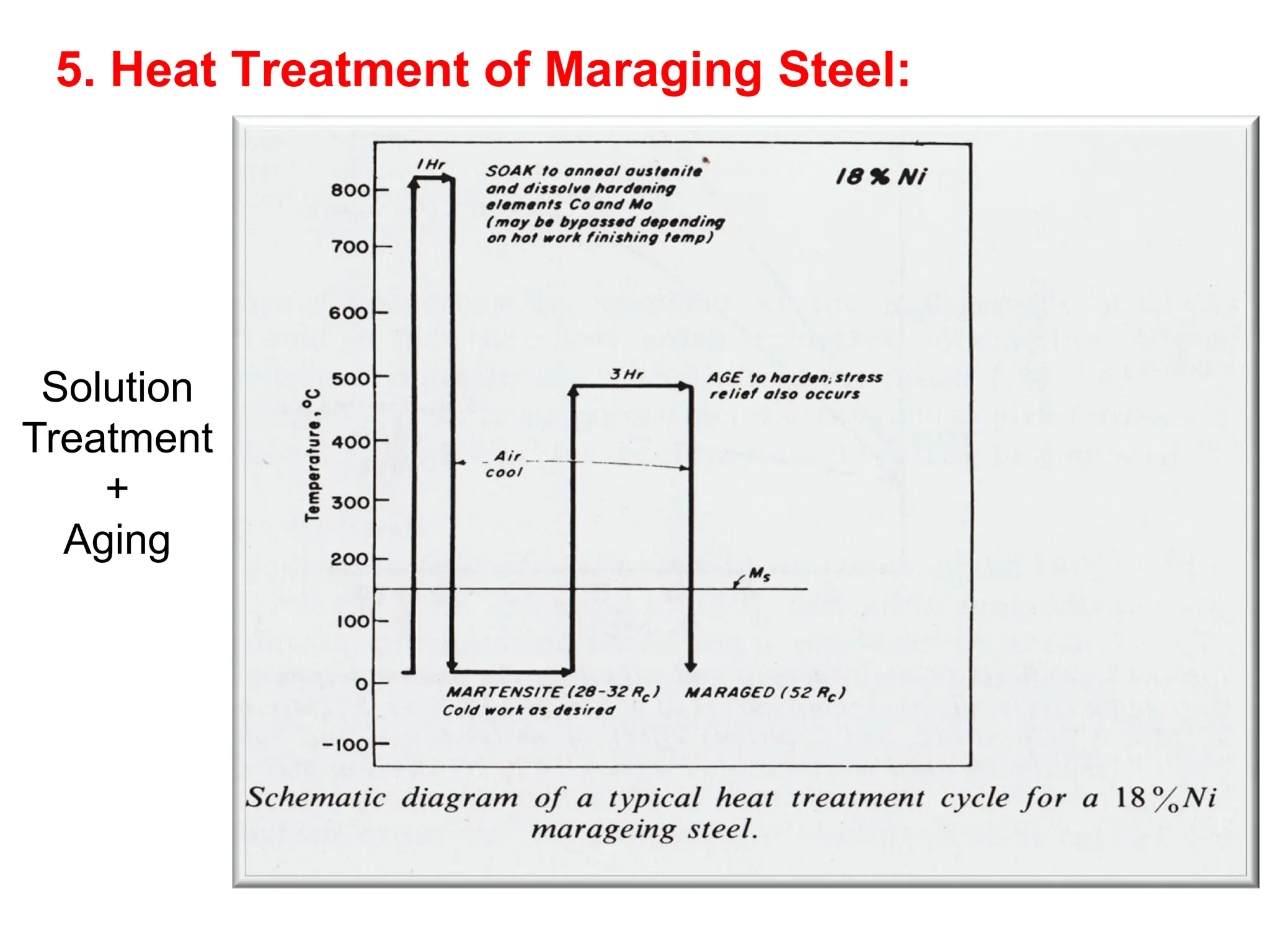
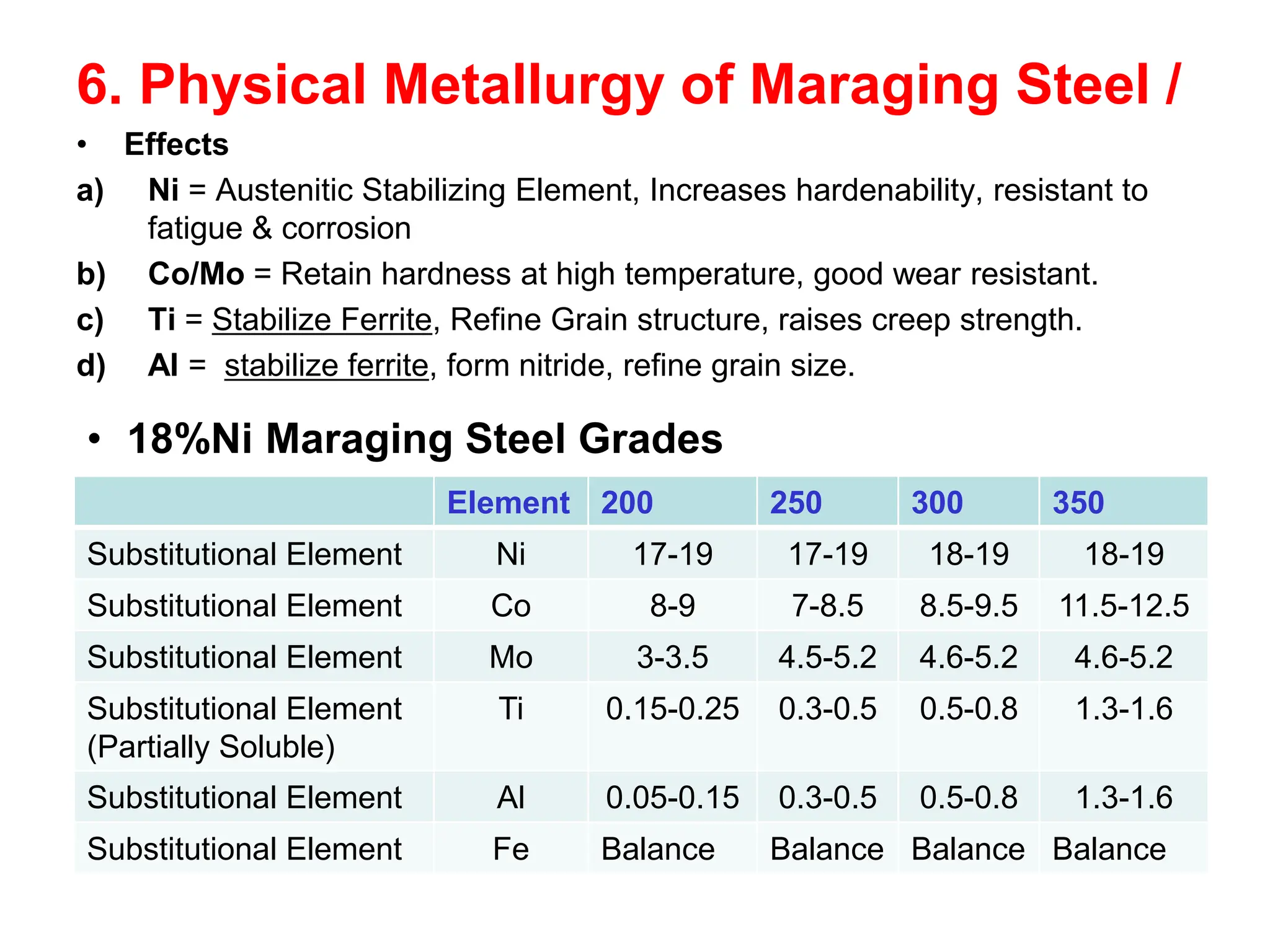
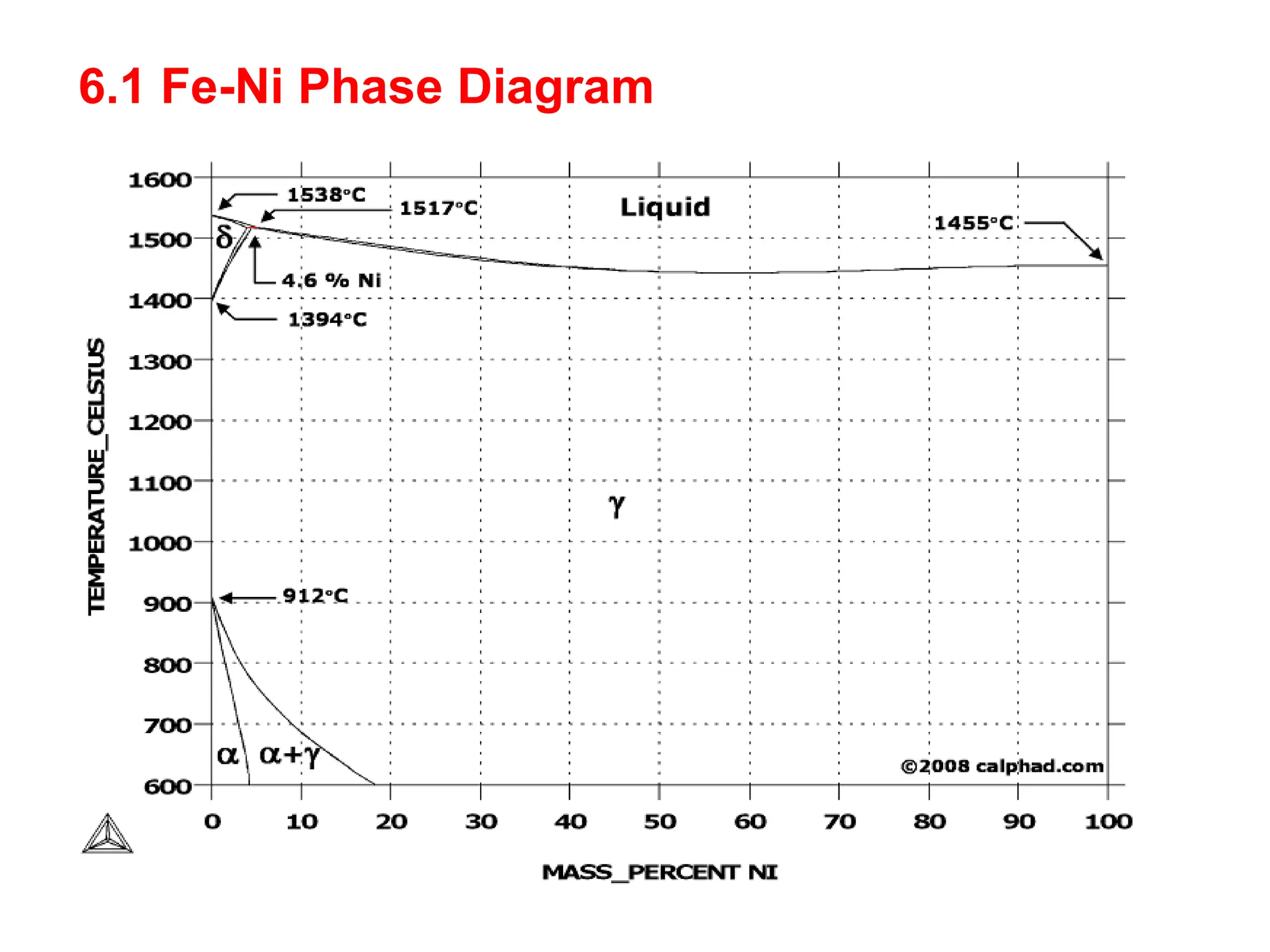
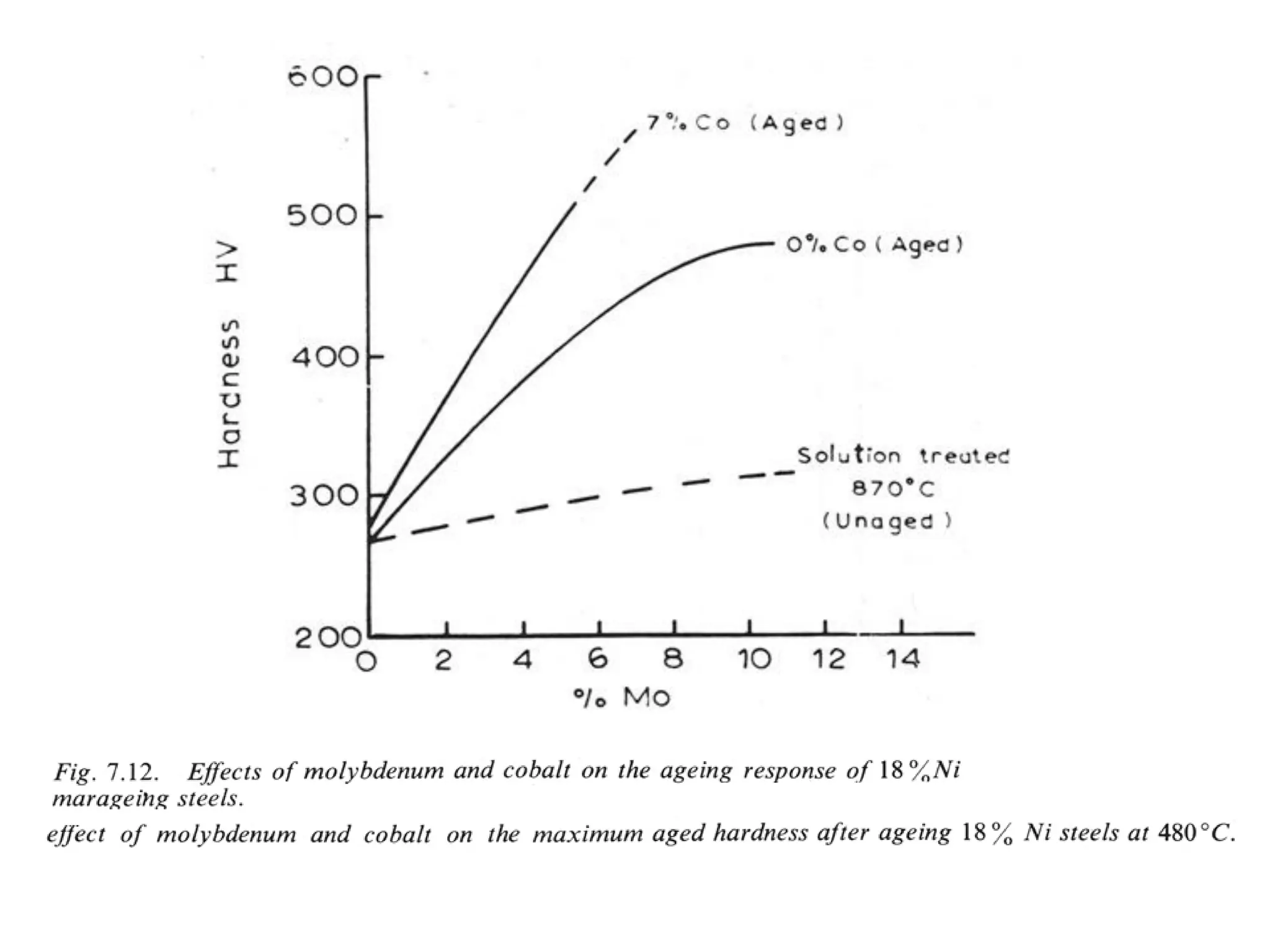
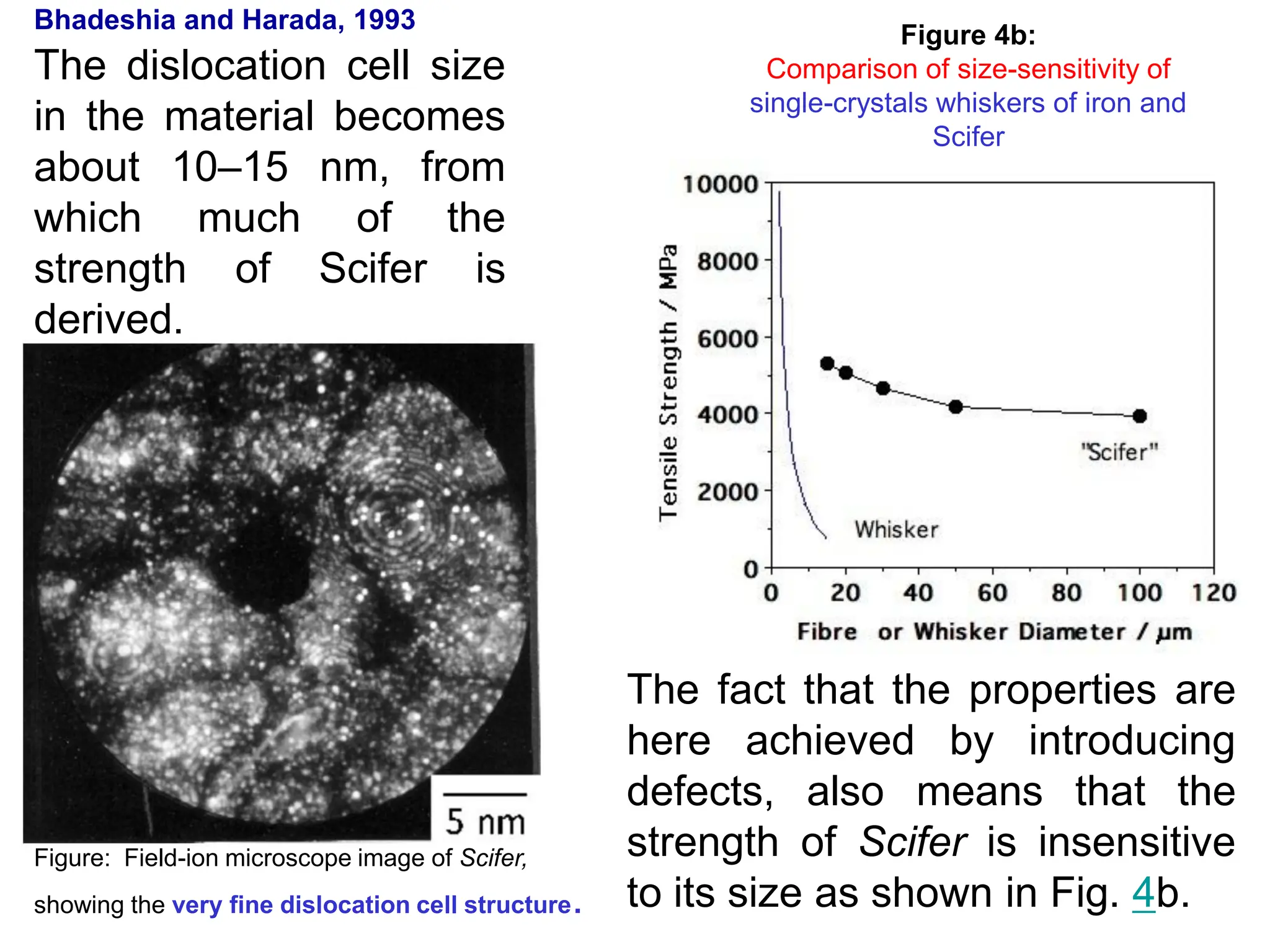
This document discusses the development of bulk nanocrystalline steel. Previous attempts to create ultra-strong materials by decreasing crystal size were limited by strength reductions as size increased due to more defects. A new method is proposed using carbide-free bainite transformation which is displacive and can generate very fine grains down to 10 nanometers. This bainitic transformation meets the design criteria of allowing large components, ultra-fine grain size, and low-cost production method required for bulk nanocrystalline steel.














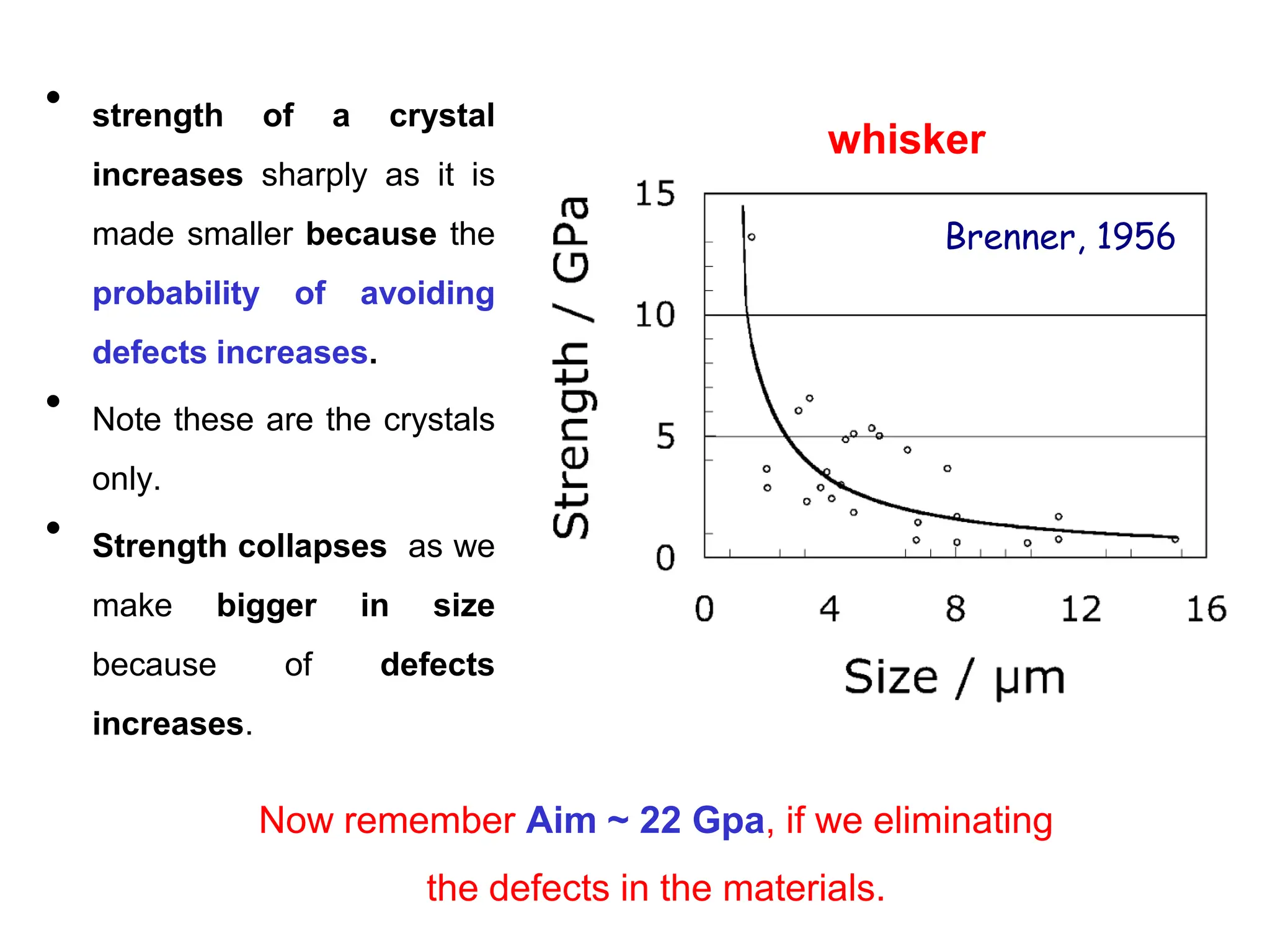







![as soon as make it big the strength
collapses due to increase in the
defects as we scale up
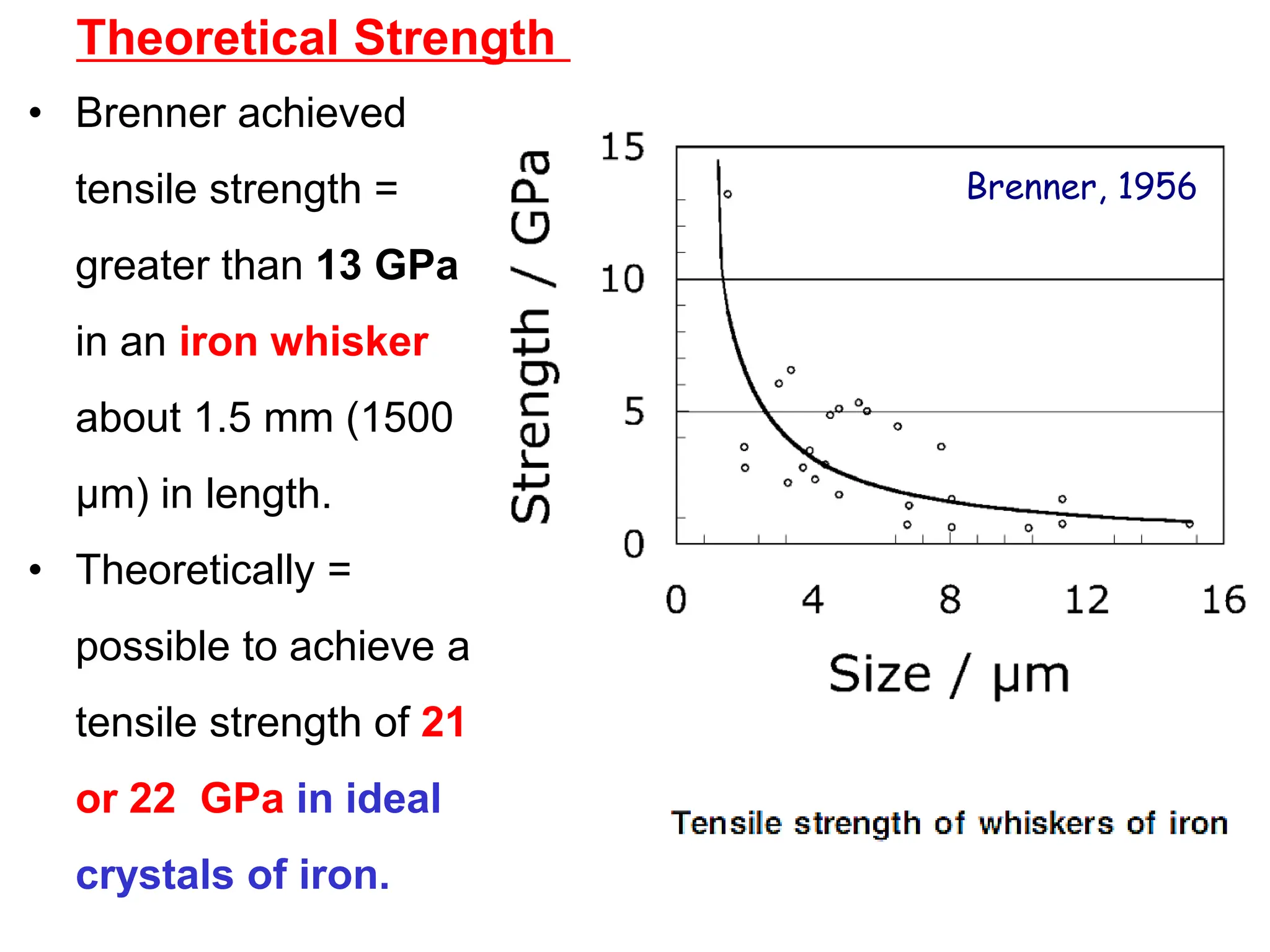
[as we know that about Fe in 1956.
(22 GPa) ]
Equilibrium number of defects (1020)
Strength of a nanotube rope 2 mm
long is less than 2000 MPa.](https://image.slidesharecdn.com/lecturec-240305115725-2cd8d6ee/75/Phase-Transformation-in-Steel-Lecture-C-pdf-35-2048.jpg)