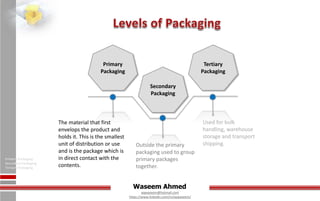
The document discusses various aspects of pharmaceutical packaging, detailing its functions, materials, and types including glass and plastic. It emphasizes the importance of packaging in protecting products, providing marketing appeal, and ensuring safety and convenience, while also outlining specific characteristics and applications of packaging materials. Additionally, it highlights the advantages and disadvantages of different types of glass and plastics used in the industry.