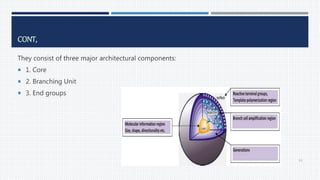
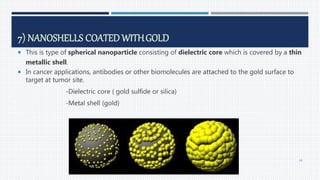
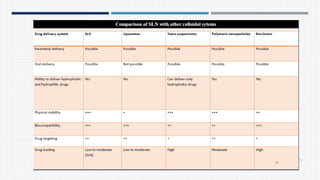
This document provides an overview of nanoparticles, including:
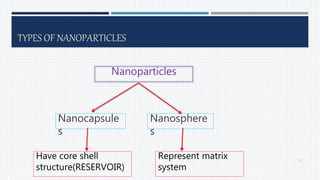
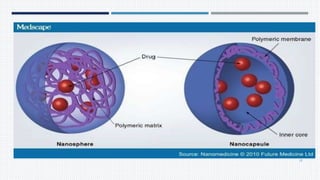
- A definition of nanoparticles as sub-nanosized colloidal drug delivery systems ranging from 1-100nm in diameter.
- The history of nanoparticles dating back to Richard Feynman in the 1960s.
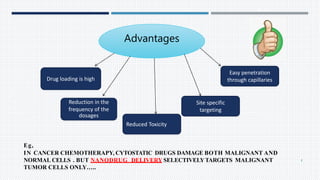
- The need for and advantages of nanoparticles for site-specific drug targeting and reduced toxicity compared to traditional drugs.
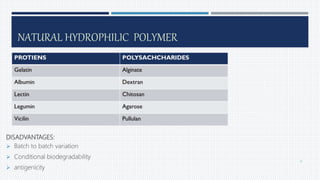
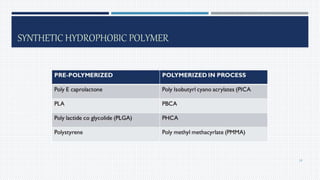
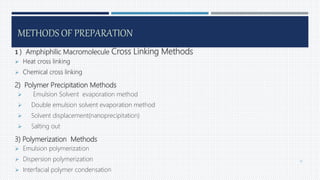
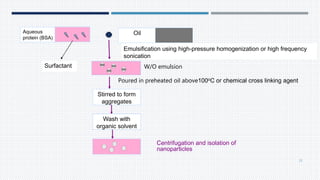
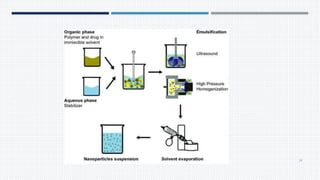
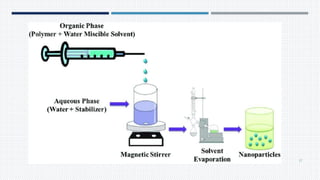
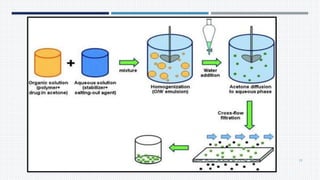
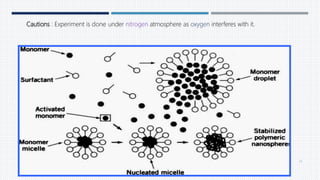
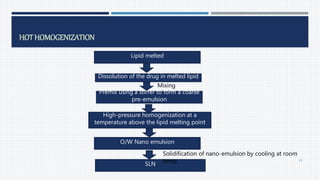
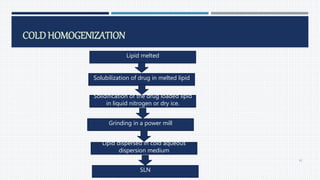
- Common polymers and preparation methods used to produce nanoparticles, including emulsion solvent evaporation, salting out, and high pressure homogenization.
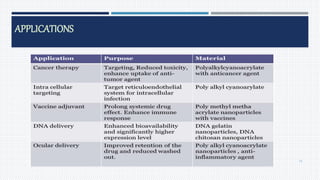
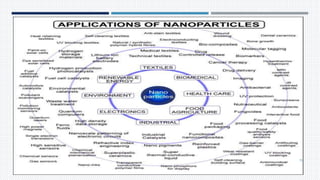
- Applications of nanoparticles in cancer chemotherapy and other areas.














































![CLASSIFICATION
Based on size & no. of bilayer:
1. Small unilamellar vesicles [ SUV ]
2. Large unilamellar vesicles [ LUV ]
3. Multilamellar vesicles [ MLV ]
.
61](https://image.slidesharecdn.com/nanoparticlessimbu200420-210529160302/85/Pharmaceutical-Nanoparticles-61-320.jpg)