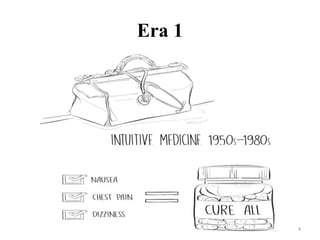
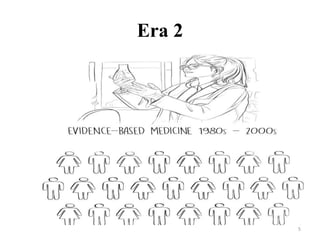
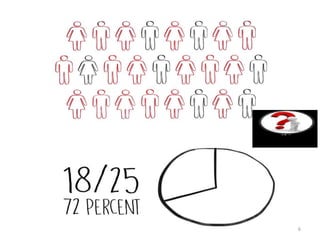
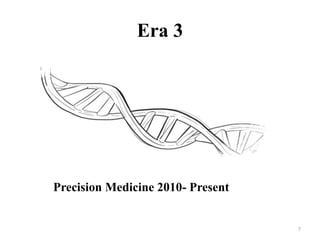
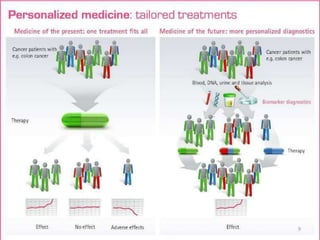
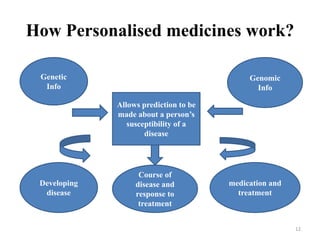
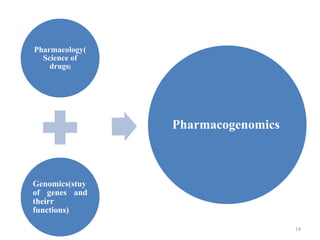
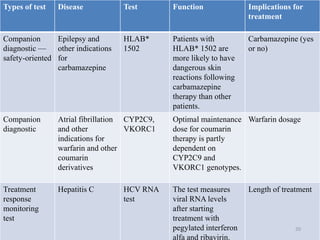
This document discusses personalised medicine, which tailors medical treatments based on individual patient characteristics, largely driven by advances in genetics and genomics. It covers the definition, need, mechanisms, benefits, limitations, and examples of personalised medicines, emphasizing that while its aim is tailored treatment, it often addresses sub-populations rather than individuals. The conclusion raises questions about the true personalization of these medicines, suggesting that current practices do not fully achieve individualized care.






















![Internet Links
NHMRC. 2011. Personalised medicine and genetics.
National Health and Medical Research Council,
http://www.nhmrc.gov.au/you/health/egenetics/personal
isedmedicine.(Accessed December 25, 2019)
Personalized Medicine E-Symposium (archived).
Available at: www.pharmacogenomicsociety.org.in
https://en.m.wikipedia.org/wiki/personalised_medicine
[Accessed December 23, 2019]
https://www.annualreviews.ord/doi/full/10.1146/annuar
ev-genom (Accessed December 23, 2019)
33](https://image.slidesharecdn.com/personalisedmedicinesseminarbyalakeshbharalifinal-200319161640/85/Personalised-medicines-pharmacogentics-and-pharmacogenomics-33-320.jpg)
