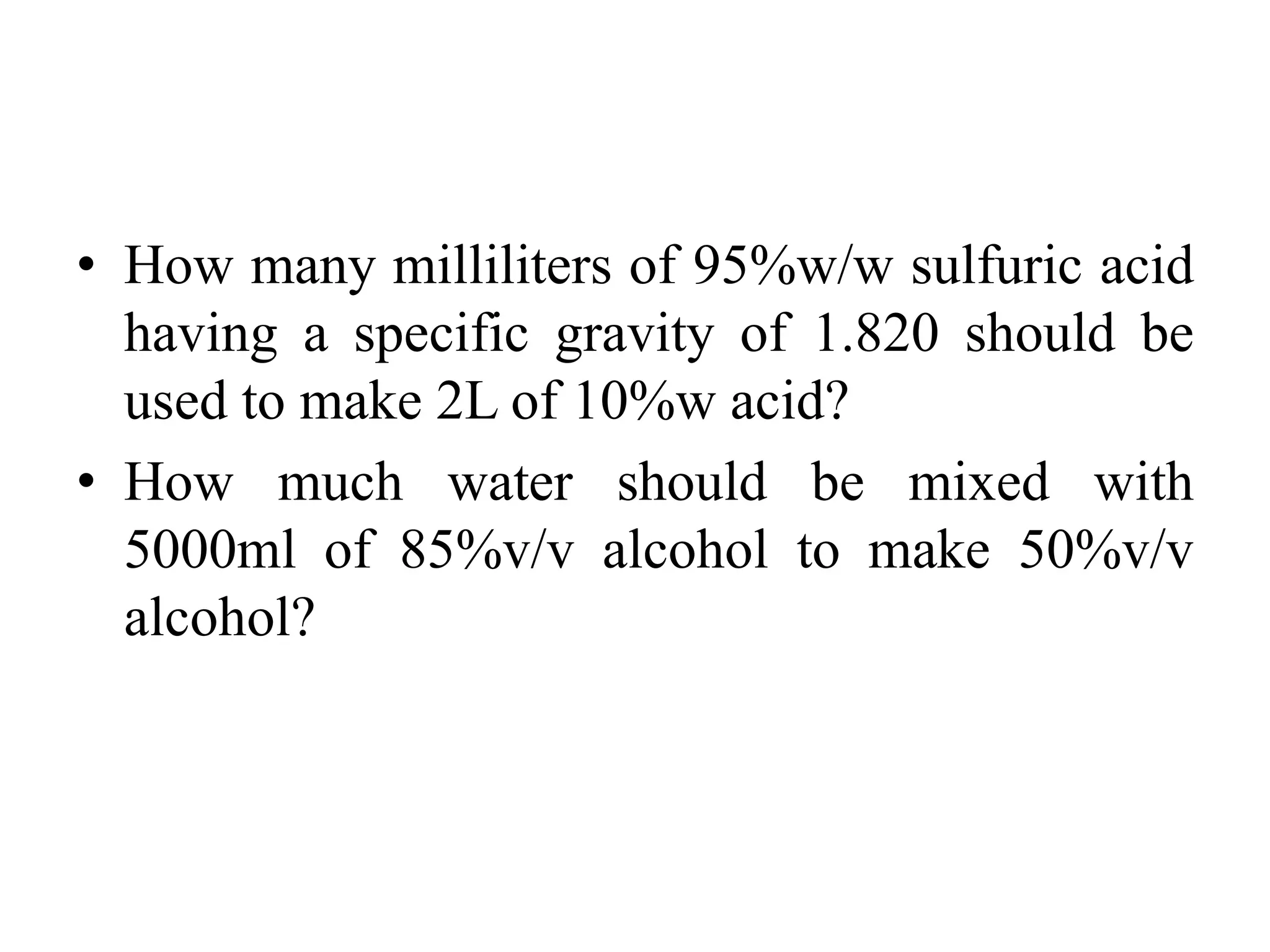The document discusses the aliquot method for weighing and measuring solids and liquids when the desired quantity is below the measurement device's limits. It involves weighing a known quantity of the material and diluting it with an inert substance to obtain the desired amount. Examples are provided for weighing specific amounts of various drugs using this method. Calculations for adjusting doses based on factors like age, weight, and surface area are also described.