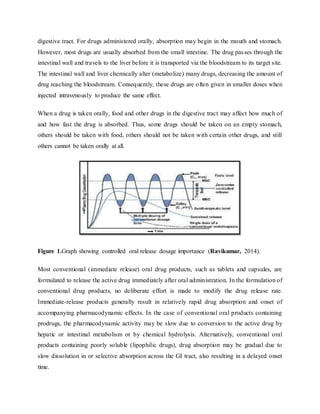
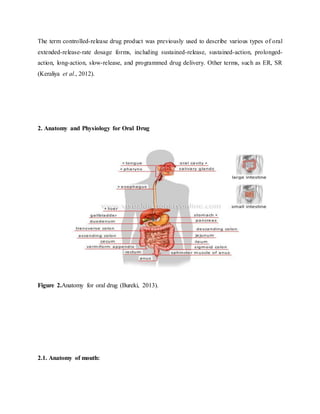
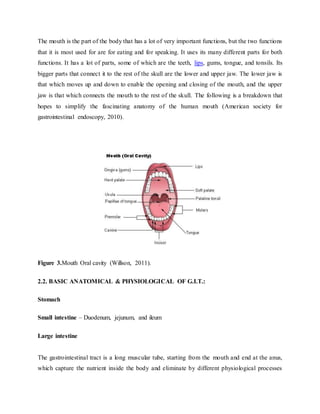
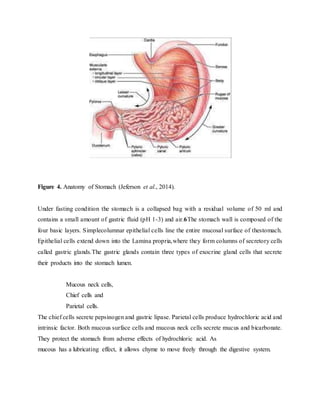
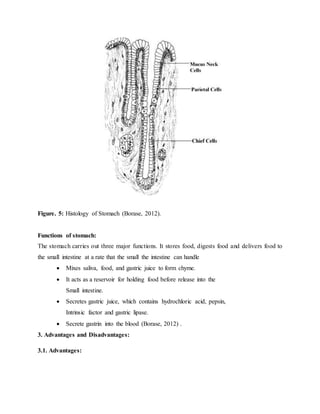
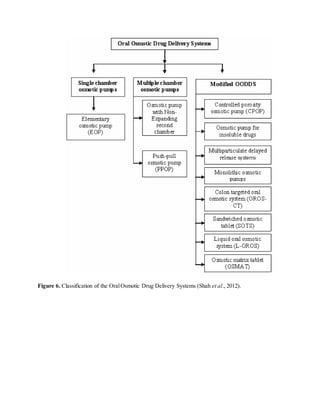
The document provides an overview of oral controlled release drug delivery systems. It discusses the anatomy and physiology related to oral drug administration. Some key advantages of controlled release oral dosage forms are enhanced bioavailability, reduced dosing frequency, and more consistent drug levels in the body. The document also covers various mechanisms for controlled release, including osmotic pumps, floating systems, and coating technologies. It provides examples of commercially available controlled release drugs and classifications of these systems.