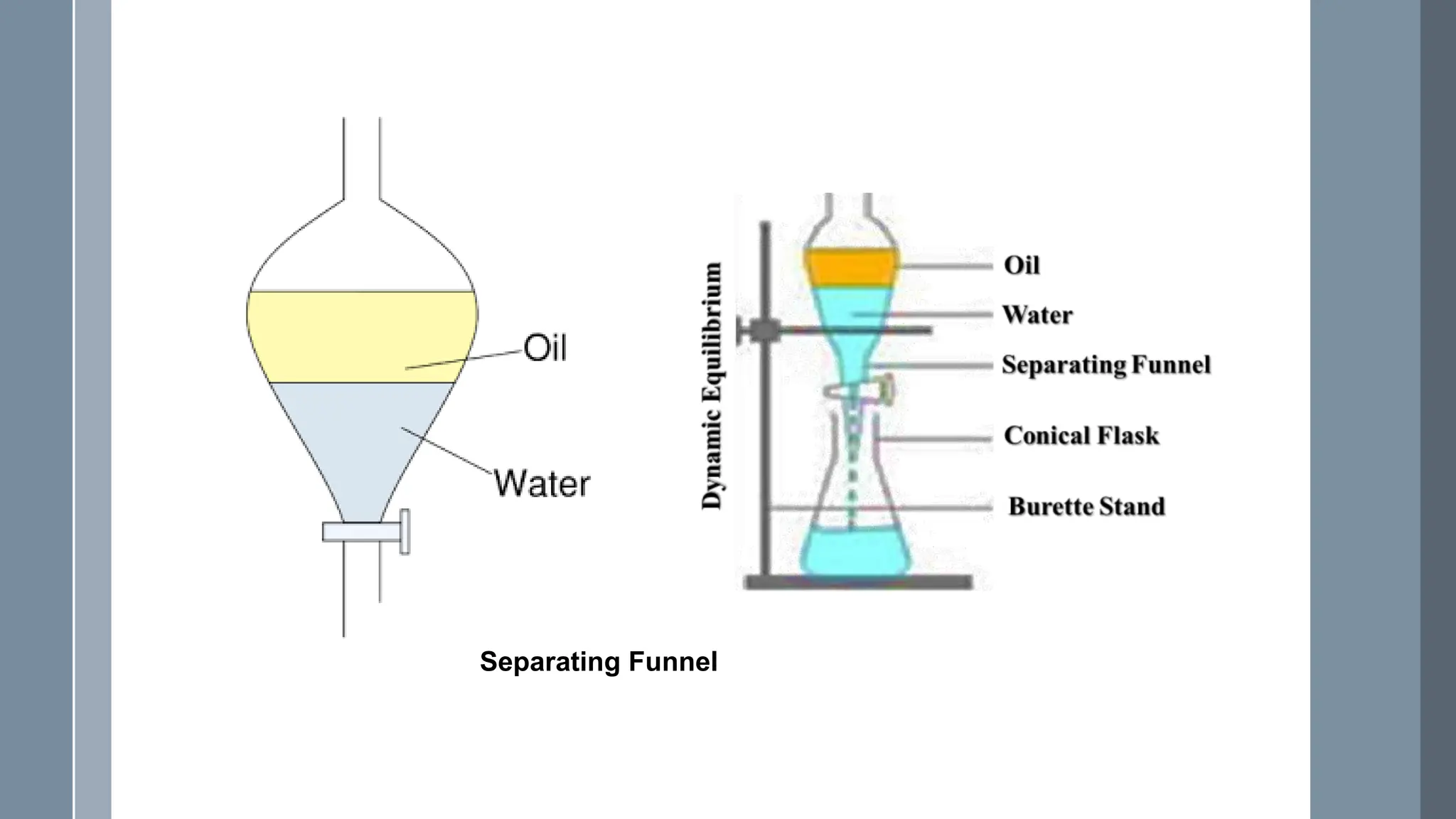
The document discusses the concept of partition coefficients in pharmaceutics, defining it as the ratio of a drug's distribution between organic and aqueous phases at equilibrium. It outlines methods for measuring partition coefficients, including the shake flask method and the HPLC method, highlighting the advantages of HPLC for more accurate results. The document also provides formulas for calculating partition coefficients and related parameters.