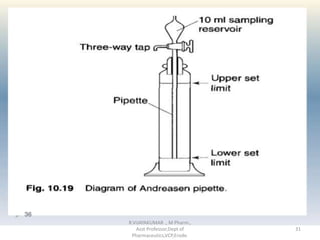
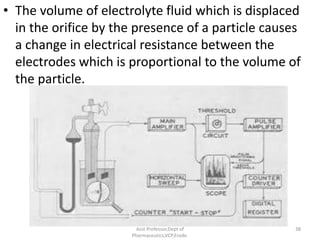
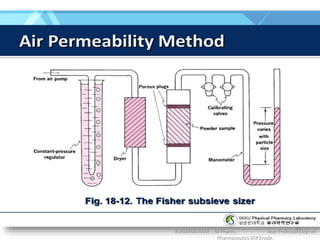
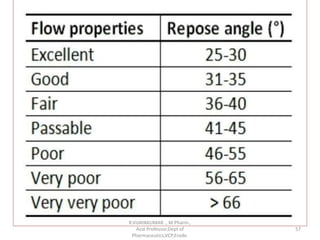
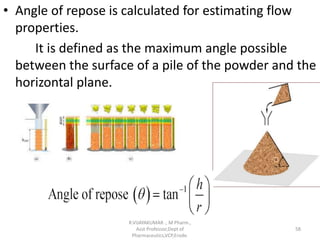
The document discusses micromeritics, the science of small particles, emphasizing the importance of particle size and distribution in pharmacy for drug release, absorption, and stability. It outlines methods for particle size determination including optical microscopy, sieving, sedimentation, and conductivity methods, describing their principles, advantages, and disadvantages. Additionally, it covers properties of powders and the significance of surface area in drug bioavailability.