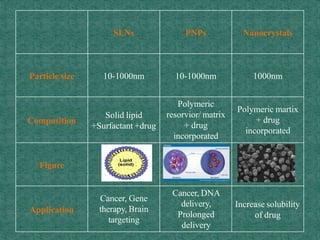
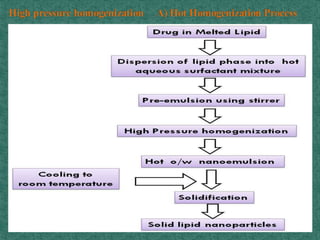
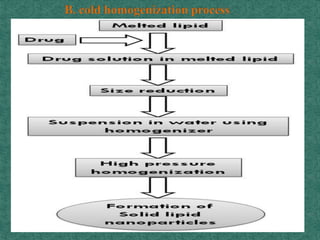
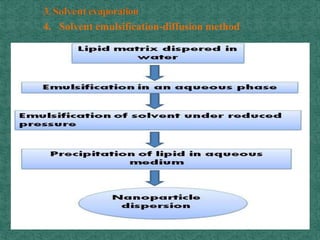
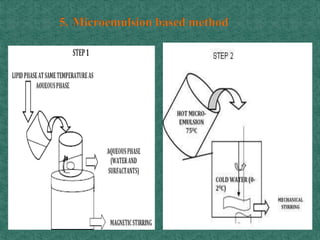
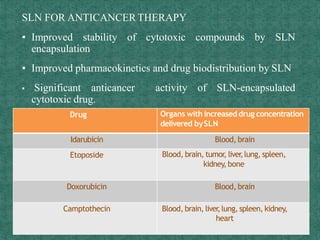
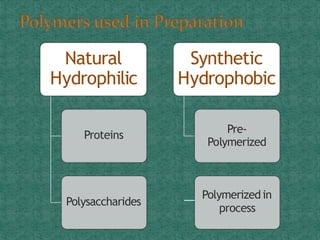
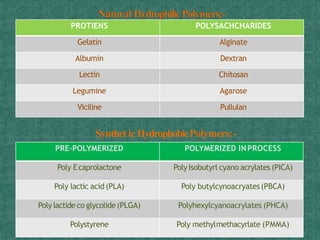
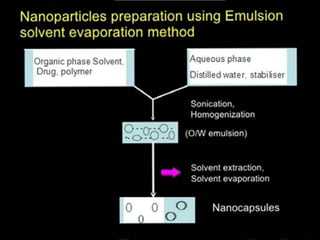
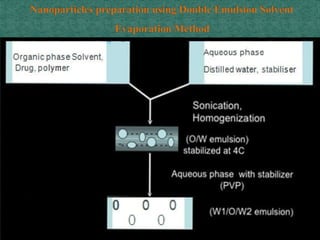
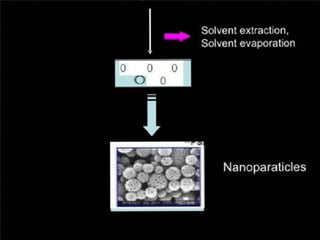
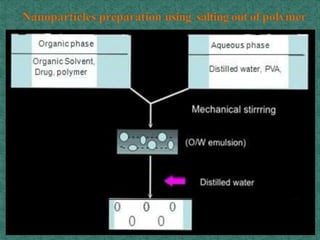
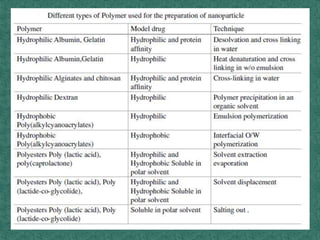
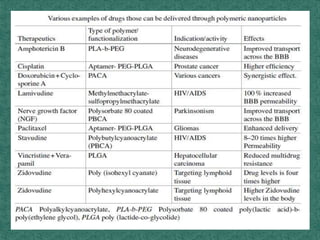
This document discusses different types of nanoparticles used for pharmaceutical applications. It begins by defining nanoparticles as structures between 1-100 nm in size. It then discusses various types of nanoparticles including solid lipid nanoparticles (SLNs), polymeric nanoparticles (PNPs), and nanocrystals. SLNs are described as 10-1000 nm particles composed of solid lipids and surfactants that can encapsulate both hydrophilic and lipophilic drugs. PNPs are similarly sized particles composed of polymeric matrices that can encapsulate or absorb drugs. The document discusses various pharmaceutical applications of these nanoparticles including cancer therapy, drug delivery, and increased drug solubility.