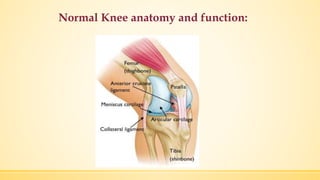
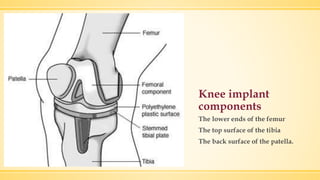
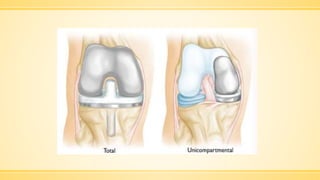
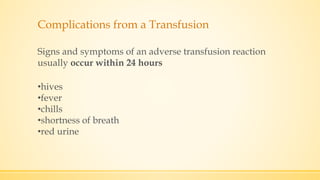
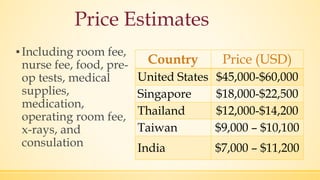
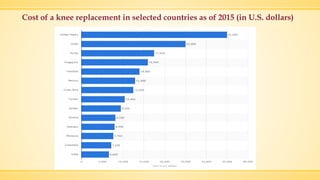
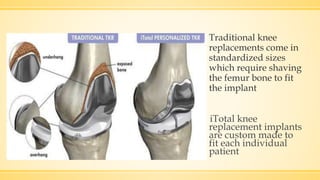
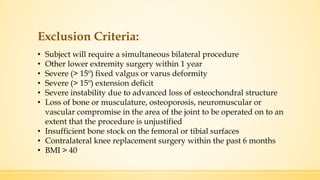
The document discusses a knee implant developed for total and partial knee replacements, including its materials, indications for surgery, risks and complications, and cost estimates across various countries. Additionally, it details a clinical trial comparing a customized knee implant to a traditional off-the-shelf version, outlining the study's design and inclusion/exclusion criteria. The document highlights both the benefits, such as pain relief and improved joint function, and the risks involved with surgery, emphasizing the importance of careful patient selection.