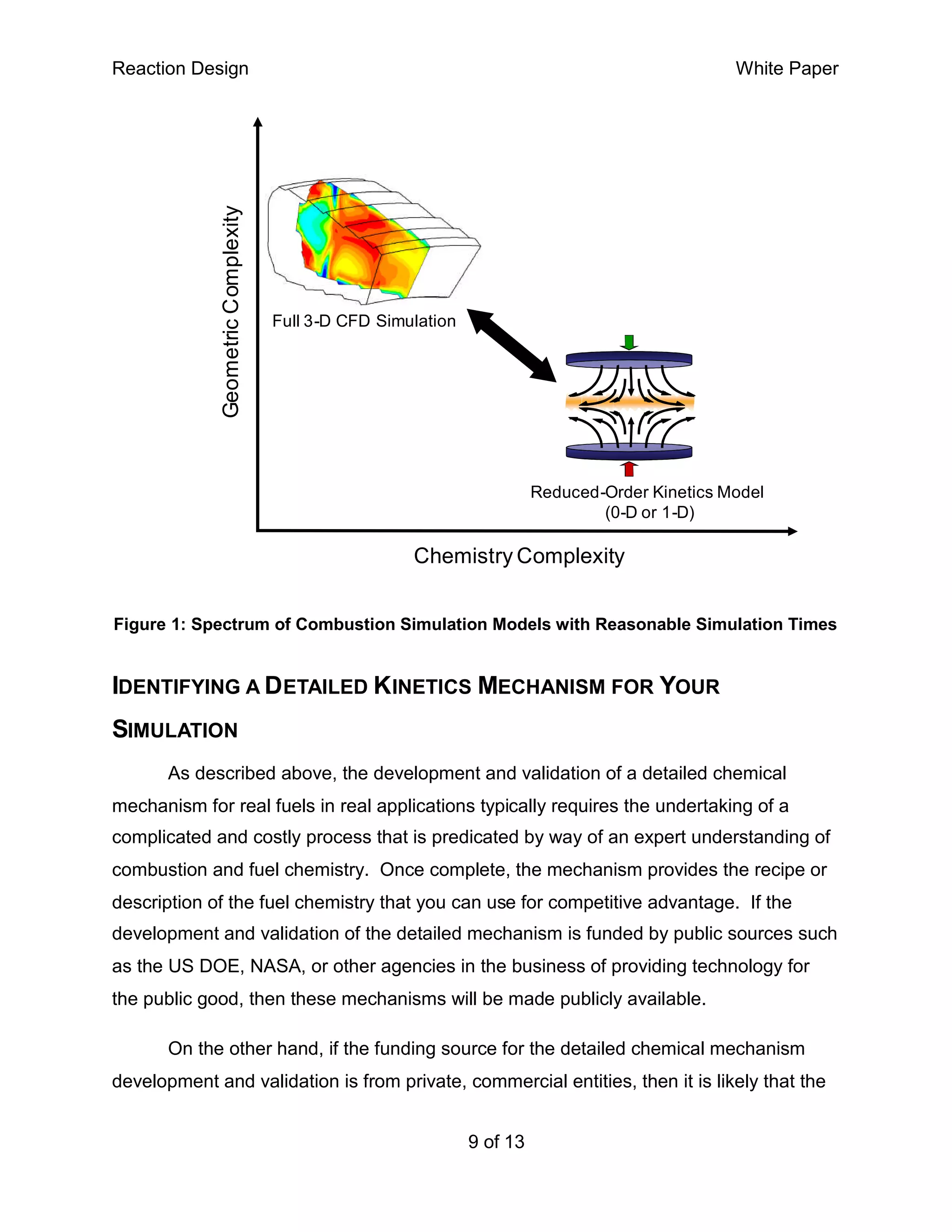
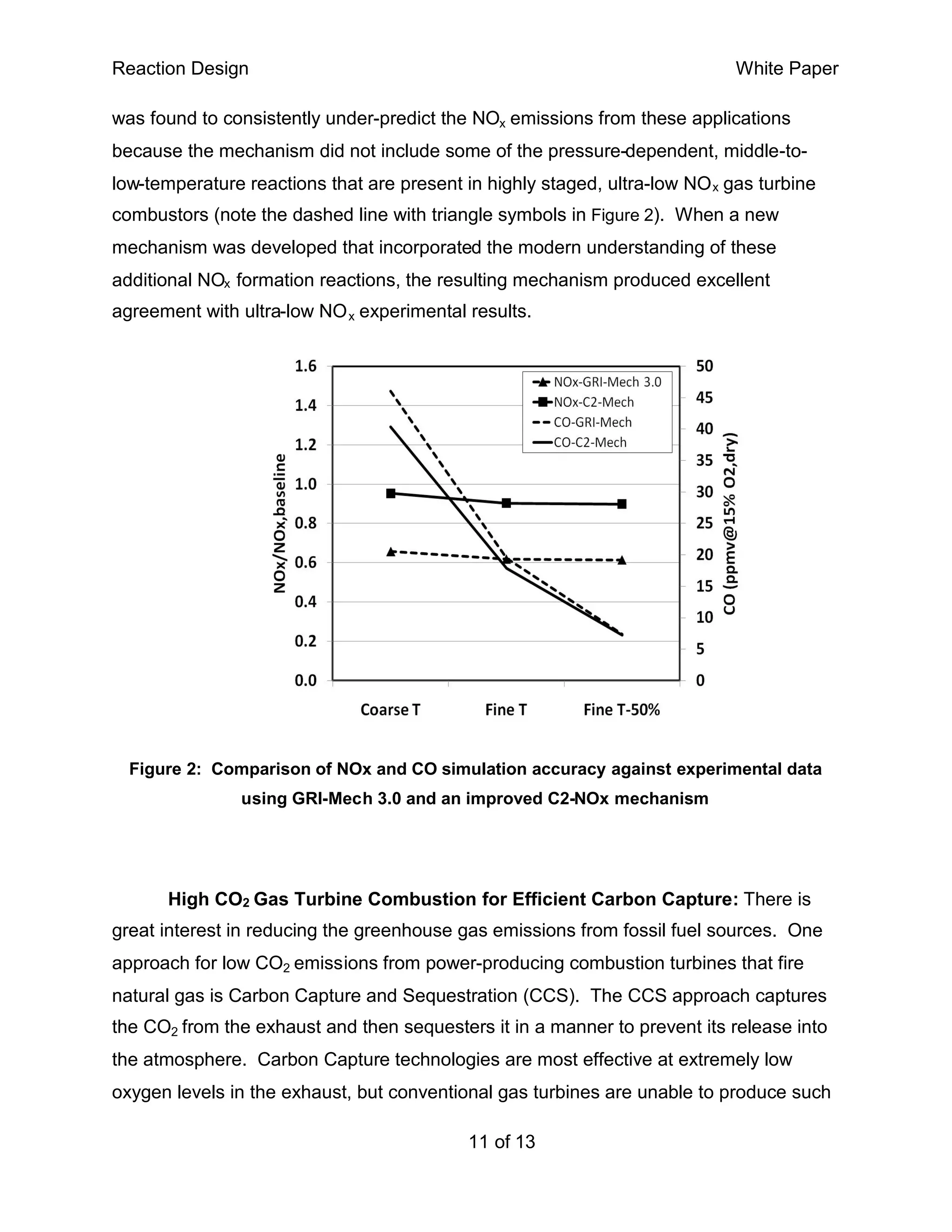
This white paper discusses the importance of detailed chemical kinetics mechanisms for accurate combustion simulation, emphasizing their role in improving efficiency, reducing emissions, and understanding combustion instability. It outlines the development, validation, and application of these mechanisms, highlighting the complexities involved in accurately modeling the combustive processes of various fuels. The document also notes that while public mechanisms exist for common fuel applications, novel or advanced technologies may require the development of proprietary mechanisms tailored to specific conditions.