Microencapsulation by Mali vv
- 1. Mr’s Vidhya V. Mali Asst. Prof. R P College of Pharmacy Osmanabad
- 2. Contents Introduction Definition Advantages Disadvantages Reasons for Microencapsulation Formulation Consideration Release Mechanisms Microencapsulation Techniques Applications of Microencapsulation
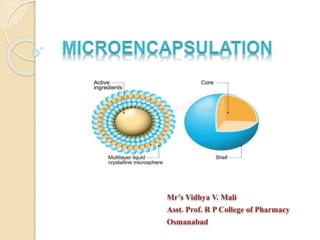
- 3. Introduction Microencapsulation is a process in which small particles or droplets (core material) is surrounded by a shell or coat (coating material) to give small capsules. Their diameters generally range from a few microns to a few millimetres . (1µm-1000µm) Solids, liquids or gasses may be enclosed by thin coating of wall or polymeric material. Increase safety and efficacy of drug. Maintain the desired concentration at the site of action. Process of surrounding or enveloping one substance within another substance on a very small scale.
- 4. Definition Microencapsulation is a process by which very tiny droplets or particles of liquid or solid material are surrounded or coated with a continuous film of polymeric material. The product obtained by this process is called as micro particles, microcapsules, microspheres, coated granules and pellets . Particles having diameter between 3 - 800µm are known as micro particles or microcapsules or microspheres. Particles larger than 1000µm are known as Macro particles .
- 5. Microcapsules and Microspheres Microcapsules are small particles that contain an active agent (core material) surrounded by a shell or coating. Their diameters generally range from a few microns to a few milimetres.
- 6. Microcapsules and Microspheres Microcapsules can have many different types and structures: a) Simple droplets of liquid core material surrounded by a spherical shell (Microcapsules) b) Irregularly-shaped particles containing small particles of solid core material dispersed in a continuous polymer shell matrix )Microspheres).
- 7. Advantages To Improve bioavailability and reduce untoward effects. To extend or alter the drug release. To improve the safety and patient’s compliance. To produce a targeted drug delivery. To reduce the reactivity of the core in relation to the outside environment. To decrease evaporation rate of the core material. To convert liquid to solid form & To mask the core taste. Microspheres are used for targeting of anticancer drugs to the Tumors
- 8. Disadvantages It is an expensive process. Difficult to achieve continuous and uniform film. Shelf life of hygroscopic drugs is reduced. Polymer may produce toxic effect. Require skills.
- 10. Formulation Consideration Generally Micro particles consist of two components a) Core material b) Coat or wall or shell material. Core Material Coating Material Vehicle Aqueous Non- aqueous Polymers Waxes Resins Proteins Polysaccharide s Solid Liquid Microencapsulation
- 11. Formulation Consideration a) Core material: The material to be coated. It may be liquid or solid or gas. Composition : 1. Drug or active constituents. 2. Additive like diluents. 3. Stabilizers. b) Coat or wall or shell material: Inert substance which coated on core with desired thickness. Composition : 1. Inert Polymer. 2. Plasticizers. 3. Coloring agents. 4. Resins, waxes and lipids. 5. Release rate enhancers or retardants.
- 12. Formulation Consideration Role of Polymers: Polymers are used widely in pharmaceutical system as: adjuvant, coating material and a component of controlled and site specific drug delivery system. Coating Material Properties: ◦ Stabilization of core material. ◦ Inert toward active ingredient. ◦ Controlled release under specific condition. ◦ Film-forming, pliable, tasteless and stable. ◦ Non-hygroscopic, no high viscosity, economic. ◦ Soluble in aqueous media or solvent or melting. ◦ The coating can be flexible, brittle, hard, thin etc Core Coating
- 15. Air suspension Techniques Air suspension technique is also called Wruster Process and Fluidized bed coating. Principle : 1. The Wrurster process consists of the dispersing of solid particulate core materials in a supporting air stream and the spray-coating of the air suspended particles. 2. Within the coating chamber, particles are suspended on an upward moving air stream as indicated in the drawing. 3. The design of the chamber and its operating parameters provide a recirculating flow of the particles through the coating zone portion of the chamber, where a coating material, usually a polymer solution, is spray-applied to the moving particles.
- 16. Air suspension Techniques 4. During each pass through the coating zone, the core material receives an increment of coating material. 5. The cyclic process is repeated several times during processing, depending on the purpose of microencapsulation, the coating thickness desired. 6. The air stream also serves to dry the product while it is being encapsulated.
- 17. Air suspension TechniquesWorking : Fine solid core materials are suspended by a vertical current of air Evaporation of the solvent The encapsulating material is deposited onto the core material Achieved the desired film thickness The encapsulated product is dried by passing the stream air
- 19. Air suspension TechniquesWorking : Disadvantage: Agglomeration of the particles to some larger size is normally achieved.
- 20. Air suspension Techniques Variables for Efficient, Effective Encapsulation by Air Suspension Techniques: 1. Density, surface area , melting point, solubility, friability, volatility, crystallinity and flow ability of core material. 2. Concentration of coating material. 3. Rate of coating material application. 4. Volume of air for fluidization of core material. 5. Amount of coating material. 6. Inlet and outlet operating temperature.
- 21. Pan Coating Principle: Pan coating process is and widely used in pharmaceutical industry. Oldest method for coating. Particles are tumbled in a pan or a device while the coating material is applied slowly. Solid particles greater than 600µ are effectively coated. It is used for preparation of controlled release beads. Coating can be applied as solution by atomized spray.
- 22. Pan Coating Working: Core material is tumbled in a pan and Coating material is applied slowly . Coating is applied as a solution or as an atomized spray. Warm air is passed over a coated material for removing the coating solvent. In some cases solvent removal can be accomplished by drying oven.
- 23. Pan Coating Working : Solid particles are mixed with a dry coating material Temperature is raised The coating material melts and encloses the core particles Achieved the desired film thickness Then is solidified by cooling
- 24. Spray drying Coating is followed by rapid evaporation of solvent in which coating material is dissolved. Removal of solvent is done by Absorption, Evaporation and Extraction technique. Principle: Dispersing the core material into coating substance and spraying for solidification. Formation of microcapsules is followed by following two different methods Spray drying Spray congealing(cooling/chilling)
- 25. Spray drying Rapid evaporation of solvent in which the coating material is dissolved. Steps: 1. Core particles are dispersed in a polymer solution and sprayed into a hot chamber. 2. The shell material solidifies onto the core particles as the solvent evaporates. 3. The microcapsules obtained are of polynuclear or matrix type.
- 26. Spray drying Equipment components of a standard spray dryer include: 1. An air heater 2. Atomizer 3. Main spray chamber 4. Blower or fan 5. Cyclone 6. Product collector Advantages: Low cost commercial process Mostly used for encapsulation of fragrances , oils and flavors
- 27. Spray congealing(cooling/chilling) Core material is dispersed in a coating material melt rather than coating solution. Coating solidification (and microencapsulation ) is accomplished by spraying the hot mixture into a cool air stream. Steps: 1. Preparation of dispersion or emulsion. 2. Homogenization of the dispersion. 3. Atomization of mass into a dry cylinder.
- 28. Spray congealing(cooling/chilling) Spray drying: Spray = Aqueous solution/ Hot air Spray Congealing: Spray= Hot melt/ Cold air
- 29. Spray congealing(cooling/chilling) Advantages: 1. Least expensive encapsulation technology. 2. Used for encapsulation of organic and inorganic salts, textural ingredients, enzymes, flavors and other functional ingredients. 3. It improves heat stability, delay release in wet environment and convert liquid into free flowing powders. Coating material used in spray drying: Gum acacia, maltodextrins, hydrophobically modified starch and mixtures. Other polysaccharides like alginate, carboxy methyl cellulose and guar gum. Proteins : Whey proteins, soy proteins, sodium caseinate etc.
- 30. Coacervation Coacervation microencapsulation is the phase separation of one or more hydrocolloids from the initial solution and subsequent deposition of the newly formed coacervate around the active ingredient. Typically used for encapsulate flavor oil, fish oils, nutrients, vitamins, preservatives and enzymes. There are two methods of coacervation microencapsulation. 1. Simple coacervation 2. Complex coacervation The mechanism for both processes is identical except for the way in which the phase separation is carried out.
- 33. Ionotropic Gelation Technique In this method, Polysaccharides(Sod. Alginate, gellan gum, pectin) are dissolved in water or in weak acidic medium (chitosan). This solution is added drop wise under a constant stirring to the solution containing other counter/multivalent ions. Due to complexation between oppositely charged ions polysaccharide undergoes ionic gelation and precipitate to form spherical particles. Beads are removed by filtration, washed with distilled water and dried.
- 34. Ionotropic Gelation Technique The counterions used in ionotropic gelation method : Low molecular weight counter ions- (e.g. CaCl2, BaCl2, MgCl2, CuCl2, ZnCl2) High molecular weight counter ions- (Octyl sulphate, Lauryl sulphate, hexadecyl sulphate)
- 35. Solvent Evaporation (Chemical process) In this case the core material is dissolved in a coating polymer solution. Polymer shrinks around core material. Evaporation of solvent. A matrix type microcapsules are formed. Core material may be water-soluble or water-insoluble.
- 36. Solvent Evaporation (Chemical process) Step 1: Formation of emulsion/dispersion (Drug and organic polymer phase). Step 2: Emulsification of polymer phase into aqueous phase (Containing suitable stabilizer thus forming o/w emulsion). Step 3: Removal of the organic solvent from the dispersed phase by extraction or evaporation (polymer precipitation to form
- 37. Single Emulsion Method The natural polymers (proteins and carbohydrates) are dissolved/dispersed In aqueous medium followed by non-aqueous medium (oil). In second step crosslinking of dispersed globules is carried out either by means of heat or by using chemical cross linkers. Chemical crosslinking agents: gluteraldehyde, formaldehyde, terepthalate chloride, diacidchloride etc.
- 38. Double Emulsion Method This type involves formation of the multiple emulsions or the double emulsion of type o/w/o. Suitable for water-soluble drugs, peptides, proteins and the vaccines. The aqueous protein solution is dispersed in organic coating polymer solution which encapsulate protein (primary emulsion). The primary emulsion is then subjected to homogenization before addition to aqueous solution of PVA. This will result in formation of double emulsion. Removal or evaporation of solvent by maintaining at reduced pressure or by stirring continuously.
- 39. Polymerization It is relatively new microencapsulation process utilizes polymerization techniques to form protective microcapsule coating in-situ. This method involves reaction of monomeric unit located at the interface between core material and continuous phase in which core material is dispersed. The core material supporting phase is liquid or gas and therefore polymerization reaction occurs at liquid-liquid, liquid-gas, solid-liquid or solid-gas interface. E.g. in the formation of polyamide (Nylon) polymeric reaction occurring at liquid- liquid interface existing between aliphatic diamine and dicarboxylic acid halide.
- 40. Polymerization There are three different methods of polymerization: 1. Interfacial polymer 2. In-situ polymerization 3. Matrix polymer
- 41. Polymerization
- 42. Polymerization
- 43. Multiorific-centrifugal Process Developed by- Southwest Research Institute (SWRI) Encapsulate solid particle and liquid droplets Mechanical process that utilizes centrifugal forces to hurl a core particle through an enveloping microencapsulation membrane there by effecting mechanical microencapsulation.
- 44. Multiorific-centrifugal Process Production rate is about 50 to 75 pounds per hour Processing variables include; The rationale speed of the cylinder The flow rate of the core The flow rate of coating material Concentration, viscosity and surface tension of core material. Advantages: Capable for microencapsulating solid and liquid of varied size ranges with diverse coating material. Production rate is higher.
- 45. Vibrational Nozzle The fluid stream of liquid core and coating material is pumped through concentric tubes by a laminar flow through a nozzle and forms droplets under the influence of vibration. The vibration has to be done in resonance of Rayleigh stability and leads to form very uniform droplets. Solidification done by gelation process Particle between 100-5000 µm encapsulated.
- 47. Applications of Microencapsulation Agricultural application Catalysis Food industry Pharmaceutical application Other applications
- 48. Applications of Microencapsulation Prolonged release dosage forms. Prepare enteric coated dosage forms. Mask the taste of bitter drugs. To reduce gastric irritation. Aid in the addition of oily medicine into the tablet dosage forms. To overcome problems inherent in producing tablets from otherwise tacky granulations. To protect drugs from environmental hazards such as humidity, light, oxygen or heat. e.g. - Vitamin A and K have been shown to be protected from moisture and oxygen through microencapsulation. The separation of incompatible substances e.g. pharmaceutical eutectics.
- 49. Applications of Microencapsulation To improve flow properties. e.g. Thiamine, Riboflavin. To enhance the stability. e.g. Vitamins. To reduce the volatility of materials. e.g. peppermint oil, methyl salicylate. To avoid incompatibilities. e.g. Aspirin and chloramphenicol. To mask the unpleasant taste and odour. e.g. Aminophylline, Castor oil. To convert liquid into solid. e.g. Castor oil, Eprazinone. To reduce gastric irritation. e.g. Nitrofuranation, Indomethacin. To Protect core material from atmosphere. e.g. Vitamin A palmitate.
- 50. Question’s 1. Define microencapsulation. List out the advantages and disadvantages of microencapsulation. 2. What are the different reasons for microencapsulation? 3. Explain the formulation consideration of microencapsulation. 4. Explain the release mechanism of microcapsules. 5. Explain the different techniques of microencapsulation. 6. Explain the coacervation phase separation technique of microencapsulation. 7. Explain the solvent evaporation technique of microencapsulation. 8. Explain the polymerization technique of microencapsulation. 9. Explain the Wruster process of microencapsulation. 10. Write down the applications of microencapsulation.
- 51. Thank you…!!!
