MICROENCAPSULATION (Definition, advantages and disadvantage, microspheres or microcapsules, and methods of Microencapsulation.
- 1. MICROENCAPSULATION By: Ankita Kanshide Maratha Mandal’s College of Pharmacy
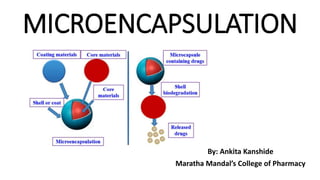
- 2. MICROENCAPSULATION • Microencapsulation is defined as a process of enclosing or enveloping solids, liquids or even gases with a continuous coating of polymeric materials yielding microscopic particles for the purposes of providing environmental protection and controlling the release characteristics. • The particle size ranges from 1-1000 µm. • Two general micro morphologies of microparticles are microcapsules and microspheres.
- 3. • The term “microcapsule” is used to describe particles in which a drug- containing core is completely surrounded by a polymer shell. • While, “microspheres” are defined as microparticles in which the drug substance is homogenously dispersed in a polymeric matrix.
- 4. FORMULATION CONSIDERATIONS 1. Core Material: The material to be coated is known as core material. It may be liquid or solid or gas. Liquid core may be dissolved or dispersed material. Composition of core material: a. Drug or active constituent b. Additive like diluents c. Stabilizers.
- 5. 2. Coating Material: Inert substance which coats on core with desired thickness. Composition of coating: a. Inert polymer b. Plasticizer c. Coloring agent d. Resins, waxes and lipids e. Release rate enhancers or retardants. Example: Gelatin, Ethyl cellulose, cellulose nitrate, Paraffin, bees wax, Shellac.
- 6. ADVANTAGES OF MICROENCAPSULATION 1. Formulation of sustained release and controlled release dosage forms. 2. Providing environmental protection to the encapsulated active agents or core materials. 3. Liquids and gases can be converted into solid particles in the form of microcapsules. 4. Masking of bitter taste of the drugs. 5. Reduce hygroscopic nature of the substance. 6. Reduction of vaporization of volatile drugs.
- 7. DISADVANTAGES OF MICROENCAPSULATION 1. Expensive techniques. 2. Microencapsulation coating may not be uniform and this can influence the release of encapsulated materials. 3. Cross reaction between core and shell material is possible. 4. More skill and knowledge is required for formulation of dosage forms by utilizing microencapsulation techniques.
- 8. METHODS OF MICROENCAPSULATION 1. Air suspension/Fluidized-bed technique 2. Pan coating 3. Coacervation phase separation 4. Spray drying 5. Solvent Evaporation
- 9. 1. Air suspension/Fluidized bed technique • Used for solid core particles, which are greater than 35 μm in size. • Solid, particulate core materials are dispersed in a supporting air stream through a fluidized bed. • The coating material is sprayed on the air suspended particles. • Within the coating chamber, particles are suspended on an upward moving air stream.
- 10. • The design of the chamber and its operating parameters effect a recirculating flow of the particles through the coating zone portion of the chamber, where a coating material, usually a polymer solution, is spray applied to the moving particles. • During each pass through the coating zone, the core material receives an increment of coating material. • The cyclic process is repeated, perhaps several hundred times during processing, depending on: The purpose of microencapsulation. The coating thickness desired. Until the core material particles are thoroughly encapsulated. • The supporting air stream also serves to dry the product while it is being encapsulated. Drying rates are directly related to the volume temperature of the supporting air stream.
- 11. 2. Pan coating • For relatively large solid core particles, which are greater than 600 μm in size, microencapsulation can be done by pan coating method, which is being widely used in pharmaceutical industry for the preparation of controlled release dosage forms. • The drug particles are tumbled in a pan, while the coating material is generally applied slowly with the moving of the pan.
- 12. • The coating is applied as a solution or as an atomized spray to the desired solid core material in the coating pan. • Warm air is passed over the coated materials as the coatings are being applied in the coating pans to remove the coating solvent. In some cases, the process of final solvent removal is accomplished in the drying oven.
- 13. 3. Coacervation phase separation Membrane formation Involves three steps: i. Formation of 3 immiscible phases (a core material phase, a coating material phase, and a liquid manufacturing phase) ii. Deposition of coating iii. Rigidization of coating.
- 14. • Used for solid/liquid core particles, which are greater than 2 μm in size. • Coacervation phase separation technique involves three major steps: i. Formation of 3 immiscible phases ii. Deposition of coating iii. Rigidization of coating. i. Formation of 3 immiscible phases (a core material phase, a coating material phase, and a liquid manufacturing phase): • Initially the core material is dispersed in a homogenous solution of coating polymer in which solvent for solubilizing the polymer is the liquid manufacturing vehicle. • The coating material is separated from the liquid manufacturing vehicle to form coacervates by application of any one of the following principles.
- 15. • Coacervation is done by: Changing the temperature of the polymer solution Addition of a salt Addition of non-solvent Addition of incompatible polymer to polymer solution Inducing polymer-polymer interaction. • Because of the application of the above principles, the solubility of the polymer in the liquid manufacturing vehicle decreases and the polymer starts separating in the form of small liquid droplets called as “Coacervates”. Thus, resulting in formation of three phases: a liquid manufacturing phase, a core material phase and a coating material phase
- 16. ii. Deposition of coating: The deposition of coating material takes place when the coacervates droplets (polymer) starts arranging themselves and gets adsorbed over the surface of core material resulting in formation of a coat around the core particles. iii. Rigidization of coating: Coating is made rigid by: Temperature rigidization Cross-linking Desolvation (evaporation of solvent)
- 17. 4. Spray drying • The pharmaceutical, and biochemical industries for is technique is widely used in food, chemical, various applications, including microencapsulation. • Basically, it is a continuous one-step and close- system process where drug is dissolved or dispersed in concentrated polymer solution and sprayed into hot gaseous medium of the spray dryer chamber. • As a consequence of solvent evaporation, the polymer rapidly solidifies around drug particles, which results in formation of either microspheres or polynuclear microparticles.
- 18. 5. Solvent Evaporation Addition of drug polymer solution to Aqueous phase Organic phase evaporation Encapsulated particles Polymer dissolved in organic solvent Drug particles Drug polymer solution &
- 19. • Used for solid/liquid core particles, which are greater than 5 μm in size. • In this method, drug is dissolved or dispersed in the polymer solution in volatile organic solvent. • The drug polymer solution is then slowly added to the aqueous phase with constant stirring. • Once the emulsion is formed, the organic solvent is removed by evaporation at elevated temperatures. • As a consequence of solvent evaporation, polymer shrinks around the drug forming microcapsules. • Several techniques can be used to achieve dispersion of the oil phase in the continuous phase. The most common method is the use of a propeller style blade attached to a variable speed motor.
- 20. EVALUATIONS OF MICROPARTICULATE DRUG DELIVERY SYSTEM 1. Microsphere recovery/yield 2. Drug Entrapment Efficiency 3. Surface Morphology 4. Particle Size Analysis 5. In vitro Release Studies 6. Differential Scanning Calorimetry (DSC) Analysis 7. In vivo Tissue Distribution Studies
- 21. APPLICATIONS • Microencapsulation can be used to formulate various sustained and controlled release dosage forms by modifying or delaying release of encapsulated active agents or core materials. • Microencapsulation can also be employed to formulate enteric- coated dosage forms, so that the drugs will be selectively absorbed in the intestine rather than the stomach. • Gastric irritant drugs are being microencapsulated to reduce the chances of gastric irritation. • The taste of bitter drugs can be masked by employing microencapsulation techniques. • Through microencapsulation, liquids and gases can be changed into solid particles in the form of microcapsules.
- 22. • Microencapsulation can employed to aid in the addition of oily medicines to tableted dosage forms to overcome the problems of tacky granulations and in direct compression. • Microencapsulation can be used to decrease the volatility. A microencapsulated volatile substance can be stored for longer times without any substantial evaporation. • Microencapsulation provides environmental protection to the encapsulated active agents from various environmental issues, such as light, heat, humidity, oxidation, etc. • Microencapsulation can reduce the hygroscopic characteristics of many core materials.
- 23. • The separations of incompatible substances can be achieved by microencapsulation. For example, pharmaceutical eutectics can be separated by microencapsulation. This is a case where direct contact of materials brings about liquid formation. The stability enhancement of incompatible aspirin-chlorpheniramine maleate mixture is accomplished by microencapsulating both of them before mixing. • Microencapsulation is used to lessen the potential danger of toxic substance handling. The toxicity owing to handling of herbicides, insecticides, pesticides and fumigants, etc., can be usefully lessened after microencapsulation.
- 24. THANK YOU