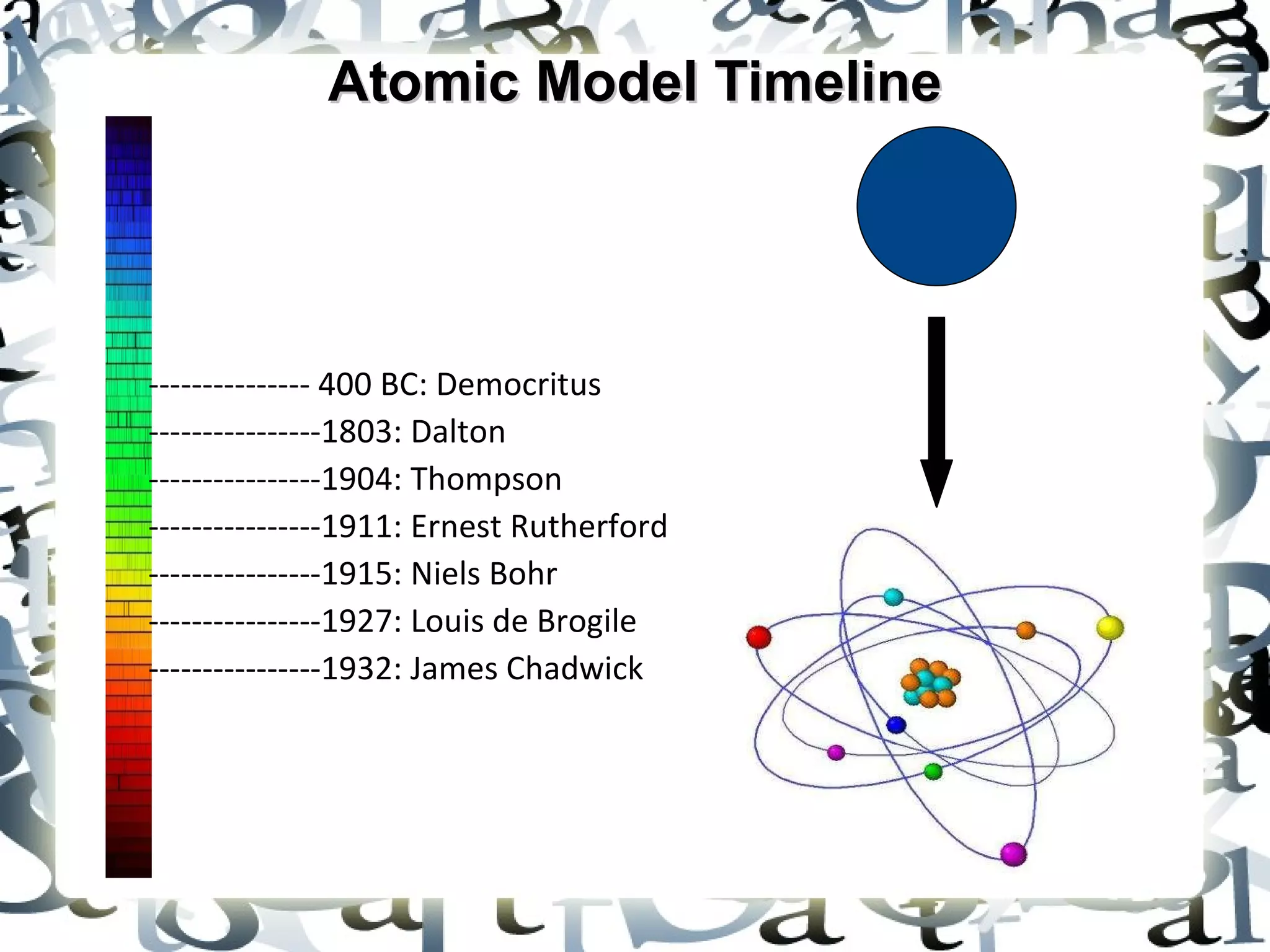
The document outlines the development of atomic models from ancient Greece to the early 20th century. It describes the models of Democritus (400 BC), Dalton (1803), Thomson (1904), Rutherford (1911), and Bohr (1915). Democritus proposed that all matter is composed of indivisible atoms. Dalton established the theory that atoms of different elements have different atomic weights. Thomson discovered electrons within atoms. Rutherford provided evidence of a dense nucleus through gold foil experiments. Bohr determined electrons orbit in shells around the nucleus.