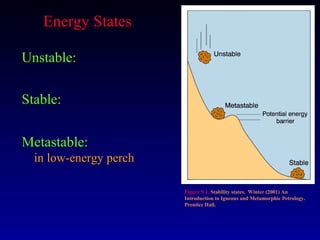
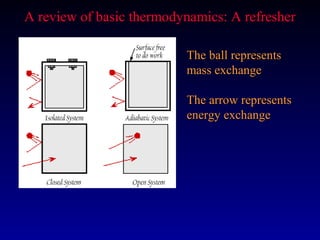
Thermodynamics describes the transfer of energy and changes in states of matter. The first law of thermodynamics states that energy is conserved during any process. The second law states that the entropy of the universe increases over time as energy disperses and becomes less useful. Natural systems evolve towards stable, low-energy states. Thermodynamic properties like enthalpy, heat capacity, and entropy can be used to characterize physical and chemical changes.