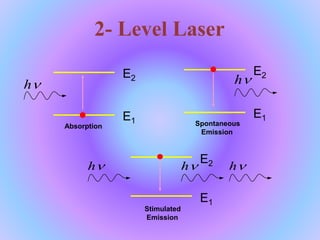
1. The document discusses the basics of laser technology, including definitions and key components. It describes how population inversion, stimulated emission, and optical pumping allow for laser action.
2. Various laser types are described, including ruby, He-Ne, and CO2 lasers. Ruby was the first demonstrated laser and uses optical pumping of chromium ions. He-Ne uses optical pumping to excite neon atoms which then transfer energy to the lasing level.
3. CO2 lasers operate on molecular gas transitions between vibrational states of carbon dioxide molecules. It is a four level system and produces a continuous wave output in the far infrared region.
























![Stimulated Emission
The probability of occurrence of stimulated emission transition from
the upper level 2 to the lower level 1 is proportional to the energy
density u(v) of the radiation and is given by
Thus the total probability of emission transition from the upper level
2 to the lower level 1 is
(P21 )st
P21 (P21)sp
P21 N2[ A21 B21 ρ()]
P21 α N2
P21α ρ(v)
(P21 )st B21N2ρ(v)
Where B21 is known as the Einstein’s coefficient of stimulated Emission of
radiation.](https://image.slidesharecdn.com/laserforengineeringstudents-200413141853/85/Laser-for-engineering-students-25-320.jpg)
![Relation between Einstein’s Coefficients
Let N1 and N2 be the number of atoms at any instant in the state 1
and 2, respectively. The probability of absorption transition for
atoms from state 1 to 2 per unit time is
P12 N1B12 ρ(v)
The probability of transition of atoms from state 2 to
1,either by spontaneously or by stimulated emission per unit time
is
P21 N2[ A21 B21 ρ( )]
at temperature t, the emission and absorption probabilities are
equal and thus
P12 P21](https://image.slidesharecdn.com/laserforengineeringstudents-200413141853/85/Laser-for-engineering-students-26-320.jpg)
![N1B12 ρ( ) N2[ A21 B21 ρ( )]
N1B12
N2 A21
N2 B21
ρ( )
N1B21 N2 B21
N2 A21
ρ( )
But Einstein proved that probability of (stimulated)
absorption is equal to the probability of stimulated
emission, So
B12 B21
B21 (N1 / N2 )1
1
ρ( )
A21](https://image.slidesharecdn.com/laserforengineeringstudents-200413141853/85/Laser-for-engineering-students-27-320.jpg)