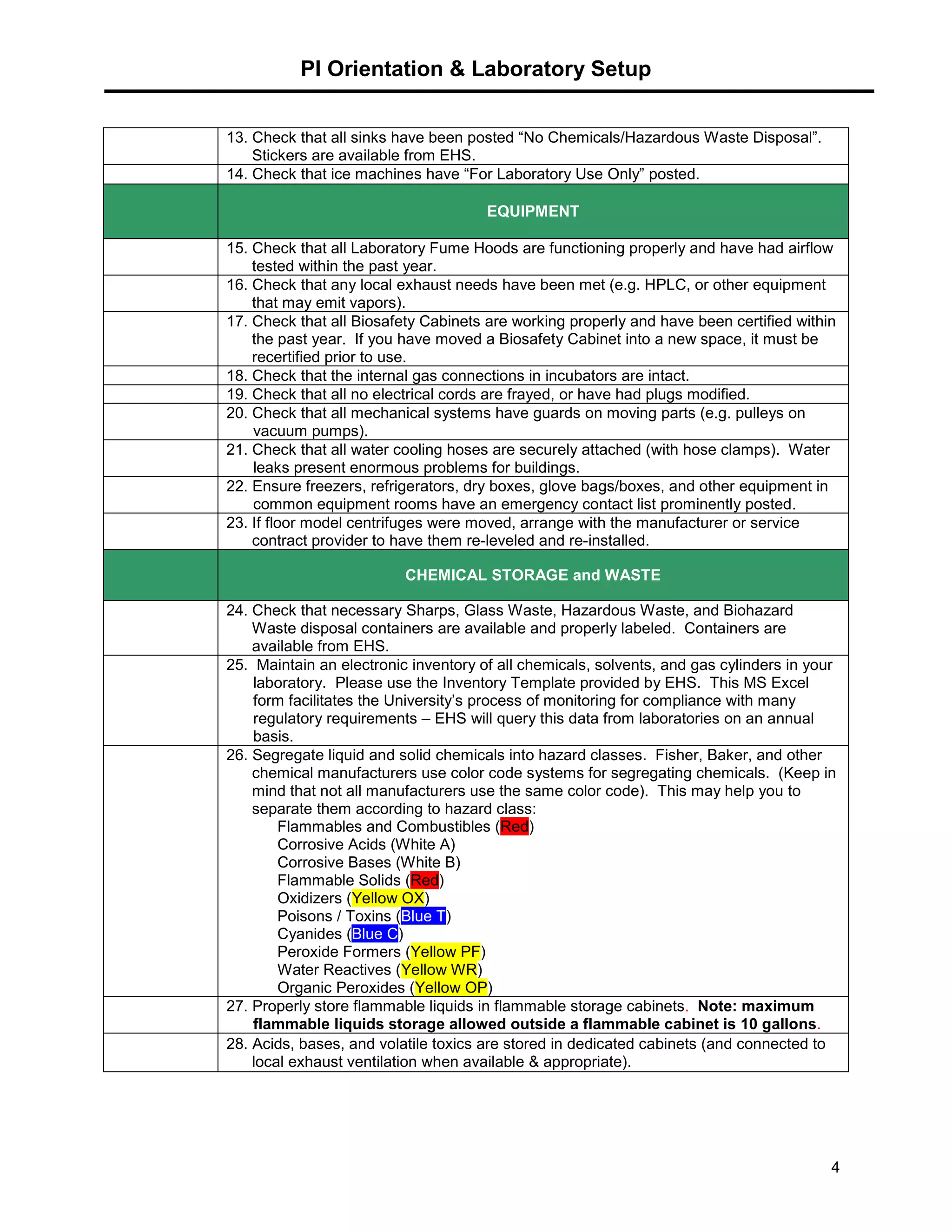
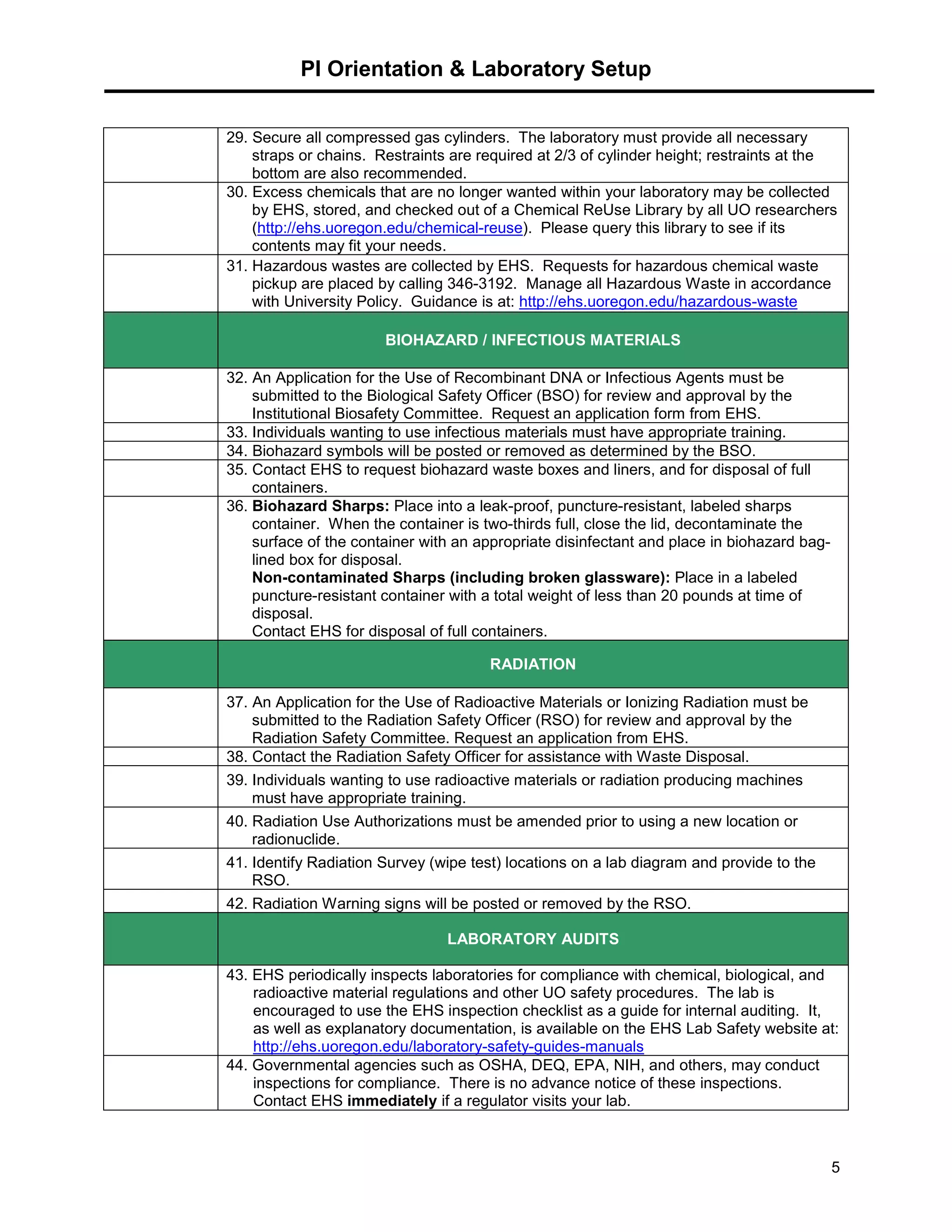
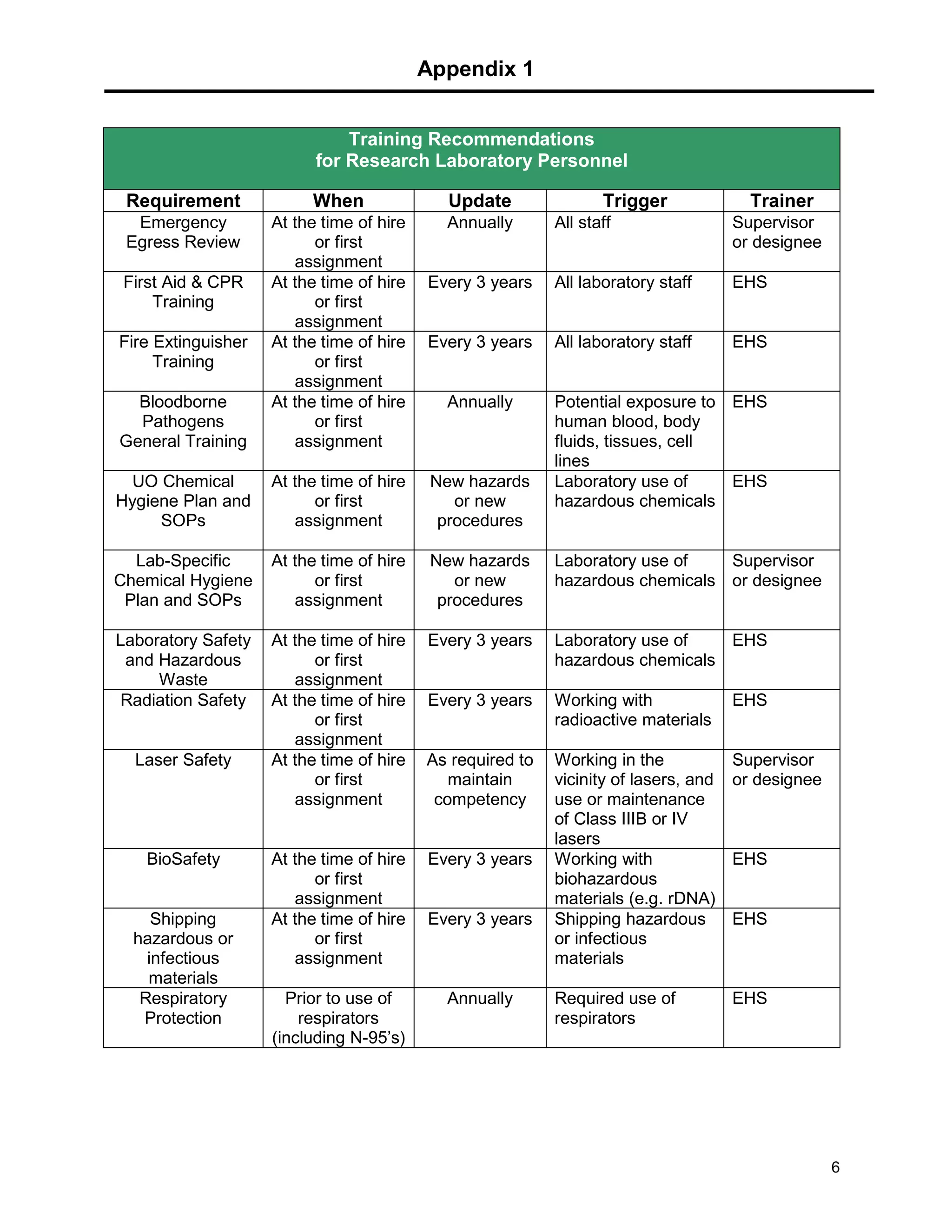
This document provides a checklist for laboratories moving into a new space or renovating their existing space to ensure proper safety protocols are followed. It outlines requirements for emergency equipment, signs, storage of chemicals and waste, and training. Laboratories are expected to maintain standards for safety, and the checklist provides steps to address requirements from the university's chemical hygiene plan, biosafety program, and other safety policies. Contact information is provided for the laboratory safety officer and other experts who can assist with the safety setup and compliance.