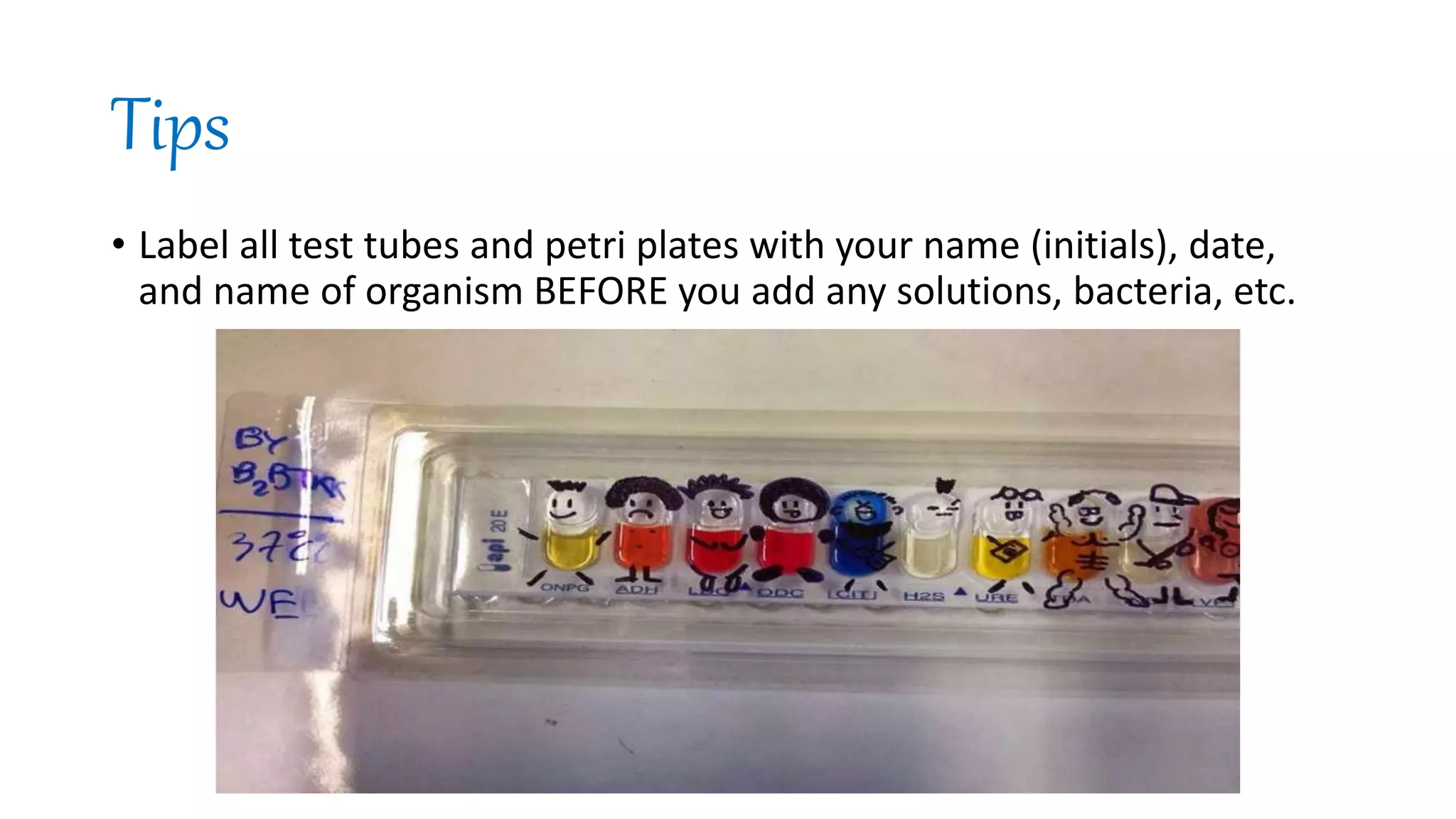
This document provides guidelines for practicing safe and aseptic techniques in a microbiology laboratory. It discusses wearing protective clothing and equipment like gloves and goggles. Maintaining cleanliness and orderliness in the lab is emphasized to avoid contamination and accidents. Common equipment used includes autoclaves for sterilization, incubators for culture growth, and laminar flow hoods. Proper labeling and disposal of materials is also covered.