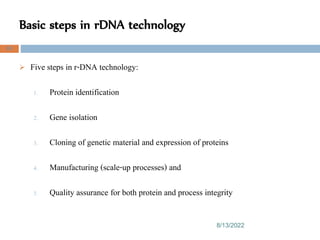
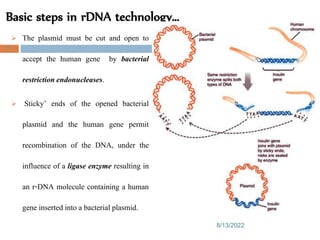
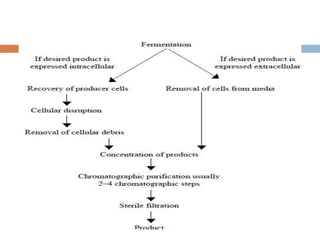
This document provides an introduction to biological products and pharmaceutical biotechnology. It defines biological products and describes some key types including proteins, blood factors, hormones, monoclonal antibodies, enzymes, and cytokines. The document outlines several technologies used in biotechnology including recombinant DNA, polymerase chain reaction, gene therapy, and monoclonal antibody production. It also summarizes the historical development of biotechnology from ancient uses of fermentation to modern discoveries of DNA and genetic coding.