How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, and Beyond—Therapeutic Selection, Sequencing, and Next Steps.
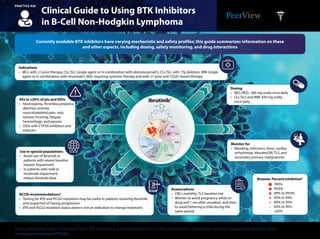
- 1. Clinical Guide to Using BTK Inhibitors in B-Cell Non-Hodgkin Lymphoma PRACTICE AID Access the activity,“How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, and Beyond—Therapeutic Selection, Sequencing, and Next Steps,” at www.peerview.com/FKM40. AEs in ≥20% of pts and DDIs • Neutropenia, thrombocytopenia, diarrhea, anemia, musculoskeletal pain, rash, nausea, bruising, fatigue, hemorrhage, and pyrexia • DDIs with CYP3A inhibitors and inducers Use in special populations • Avoid use of ibrutinib in patients with severe baseline hepatic impairment • In patients with mild or moderate impairment, reduce ibrutinib dose Dosing • MCL/MZL: 560 mg orally once daily • CLL/SLL and WM: 420 mg orally once daily Monitor for • Bleeding, infections, fever, cardiac arrhythmias, elevated BP, TLS, and secondary primary malignancies Assess/advise • CBCs monthly; TLS baseline risk • Women to avoid pregnancy while on drug and 1 mo after cessation, and men to avoid fathering a child during the same period Kinome: Percent Inhibition5 NCCN recommendations4 • Testing for BTK and PLCG2 mutations may be useful in patients receiving ibrutinib and suspected of having progression • BTK and PLCG2 mutation status alone is not an indication to change treatment Indications • MCL with ≥1 prior therapy, CLL/SLL (single agent or in combination with obinutuzumab2 ), CLL/SLL with 17p deletion, WM (single agent or in combination with rituximab3 ), MZL requiring systemic therapy and with ≥1 prior anti-CD20–based therapy Currently available BTK inhibitors have varying mechanistic and safety profiles; this guide summarizes information on these and other aspects, including dosing, safety monitoring, and drug interactions Ibrutinib1 TKL STE CK1 AGC CAMKCMGC Other TK N N N N N H2 N O O 100% 99.9% 99% to 99.9% 95% to 99% 90% to 95% 65% to 90% <65%
- 2. Clinical Guide to Using BTK Inhibitors in B-Cell Non-Hodgkin Lymphoma PRACTICE AID Access the activity,“How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, and Beyond—Therapeutic Selection, Sequencing, and Next Steps,” at www.peerview.com/FKM40. AEs in ≥20% of pts and DDIs • Neutropenia, thrombocytopenia, diarrhea, anemia, musculoskeletal pain, rash, nausea, bruising, fatigue, hemorrhage, and pyrexia • DDIs with CYP3A inhibitors and inducers, and gastric acid–reducing agents (PPIs, H2-receptor antagonists, and antacids) Use in special populations • Advise women not to breastfeed Dosing • 100 mg orally approximately every 12 h; swallow whole with water, with or without food • Advise not to break, open, or chew capsule Monitor for • Bleeding, signs and symptoms of infections, secondary primary malignancies, atrial fibrillation, and trial flutter Assess/advise • CBCs monthly • Patients to use sun protection Kinome: Percent Inhibition5 NCCN recommendations4 • NCCN also recommends acalabrutinib for relapsed/refractory CLL (except ibrutinib-refractory CLL with BTK C481S mutations) • Patients with ibrutinib intolerance have been successfully treated with acalabrutinib without recurrence of these symptoms Indications • Adult patients with MCL who have received ≥1 prior therapy Currently available BTK inhibitors have varying mechanistic and safety profiles; this guide summarizes information on these and other aspects, including dosing, safety monitoring, and drug interactions Acalabrutinib6 TKL STE CK1 AGC CAMKCMGC Other TK O N N N NH2 N O NH N 100% 99.9% 99% to 99.9% 95% to 99% 90% to 95% 65% to 90% <65%
- 3. AE: adverse event; BTK: Bruton's tyrosine kinase; CAMK: calcium/calmodulin-dependent protein kinase; CBC: complete blood count; CK1: casein kinase 1; CLL: chronic lymphocytic leukemia; CYP3A: cytochrome P450 3A; DDI: drug–drug interaction; MCL: mantle cell lymphoma; MZL: marginal zone B-cell lymphoma; NCCN: National Comprehensive Cancer Network; PPI: proton pump inhibitor; SLL: small lymphocytic lymphoma; STE: sulfotransferase, estrogen-preferring; TK: tyrosine kinase; TKL: tyrosine kinase–like; TLS: tumor lysis syndrome; WM: Waldenström’s macroglobulinemia. 1. Imbruvica (ibrutinib) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/205552s002lbl.pdf. Accessed November 12, 2019. 2. http://www.ascopost.com/News/59690. Accessed November 12, 2019. 3. http://www.ascopost.com/News/59204. Accessed November 12, 2019. 4. NCCN Clinical Practice Guidelines in Oncology. Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma. Version 2.2020. 5. Kaptein A et al. 60th American Society of Hematology Meeting and Exposition (ASH 2018). Abstract 1871. 6. Calquence (acalabrutinib) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/210259s000lbl.pdf. Accessed November 12, 2019. 7. Brukinsa (zanubrutinib) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/ label/2019/213217s000lbl.pdf. Accessed November 12, 2019. Clinical Guide to Using BTK Inhibitors in B-Cell Non-Hodgkin Lymphoma PRACTICE AID Access the activity,“How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, and Beyond—Therapeutic Selection, Sequencing, and Next Steps,” at www.peerview.com/FKM40. AEs in ≥20% of pts and DDIs • Neutropenia, thrombocytopenia, upper respiratory tract infection, decreased white blood cells, decreased hemoglobin, rash, bruising, diarrhea, and cough • DDIs with CYP3A inhibitors and inducers Use in special populations • In patients with severe hepatic impairment, reduce zanubrutinib dose • Advise women not to breastfeed Dosing • 160 mg orally twice daily or 320 mg orally once daily; swallow whole with water, with or without food • Advise not to break, open, or chew capsule Monitor for • Bleeding, signs and symptoms of infections, secondary primary malignancies, atrial fibrillation, and atrial flutter Assess/advise • CBCs • Avoid use during pregnancy Indications • Adult patients with MCL who have received ≥1 prior therapy Currently available BTK inhibitors have varying mechanistic and safety profiles; this guide summarizes information on these and other aspects, including dosing, safety monitoring, and drug interactions TKL STE CK1 AGC CAMKCMGC Other TK Zanubrutinib7 N N N HN H2 N O O O Kinome: Percent Inhibition5 100% 99.9% 99% to 99.9% 95% to 99% 90% to 95% 65% to 90% <65%
- 4. Selected Studies With BTK Inhibitors Clinical Trial Landscape of B-Cell Non-Hodgkin Lymphoma1 PRACTICE AID Access the activity,“How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, and Beyond—Therapeutic Selection, Sequencing, and Next Steps,” at www.peerview.com/FKM40. Ibrutinib Zanubrutinib (BGB-3111) Key: ˜ Active, not recruiting ˜ Currently recruiting Phase 3 Clinical Trials Phase 3 Clinical Trials PFS NCT01776840 (SHINE) Ibrutinib + bendamustine + rituximab in patients with newly diagnosed MCL CR, VGPR NCT03053440 (BGB-3111-302 ASPEN) Zanubrutinib vs ibrutinib in patients with WM PFS NCT02947347 (PERSPECTIVE) Ibrutinib + rituximab in patients with treatment-naïve FL PFS NCT03112174 (SYMPATICO) Ibrutinib + venetoclax in previously treated patients with MCL PFS ISRCTN01844152 (FLAIR)2 Ibrutinib monotherapy vs ibrutinib + rituximab vs ibrutinib + venetoclax vs FCR in patients with treatment-naïve CLL/SLL TLS, DLT, PFS, CR NCT03462719 (GLOW/CLL 3011) Ibrutinib + venetoclax vs chlorambucil + obinutuzumab for patients with newly diagnosed CLL/SLL PFS NCT03336333 (BGB-3111-304 SEQUOIA) Zanubrutinib vs bendamustine + rituximab in patients with previously untreated CLL/SLL Approved BTK inhibitors currently include ibrutinib, acalabrutinib, and zanubrutinib Primary Endpoint(s) Primary Endpoint(s)
- 5. BTK: Bruton's tyrosine kinase; CLL: chronic lymphocytic leukemia; CR: complete response; DLT: dose-limiting toxicities; FCR: fludarabine, cyclophosphamide, and rituximab; FL: follicular lymphoma; MCL: mantle cell lymphoma; MRD-CR: minimal residual disease–negative complete response; R/R: relapsed/refractory; SLL: small lymphocytic lymphoma; TLS: tumor lysis syndrome; TN: treatment naïve; VGPR: very good partial response; WM: Waldenström’s macroglobulinemia. 1. https://clinicaltrials.gov/ct2/home. Accessed November 19, 2019. 2. https://www.cancerresearchuk.org/about-cancer/find-a-clinical-trial/a-trial-ibrutinib-rituximab-chronic-lymphocytic-leukaemia-flair#undefined. Accessed November 19, 2019. 3. Sharman JP et al. 61st Annual Meeting & Exposition of the American Society of Hematology (ASH 2019). Abstract 31. 4. NCCN Clinical Practice Guidelines in Oncology. B-Cell Lymphomas. Version 5.2019. https://www.nccn.org/professionals/physician_gls/pdf/b-cell.pdf. Accessed November 19, 2019. Selected Studies With BTK Inhibitors Clinical Trial Landscape of B-Cell Non-Hodgkin Lymphoma1 PRACTICE AID Access the activity,“How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, and Beyond—Therapeutic Selection, Sequencing, and Next Steps,” at www.peerview.com/FKM40. PFS Results available (ASH 2019): Acalabrutinib ± obinutuzumab significantly improved PFS vs obinutuzumab + chlorambucil3 NCT02475681 (Elevate CLL TN) Acalabrutinib monotherapy vs acalabrutinib + obinutuzumab vs obinutuzumab + chlorambucil in patients with previously untreated CLL/SLL PFS NCT02972840 (ACE-LY-308) Acalabrutinib + bendamustine/rituximab vs bendamustine/rituximab in patients with previously untreated MCL PFS NCT02970318 (ACE-CL-309) Acalabrutinib vs investigator's choice of idelalisib + rituximab or bendamustine + rituximab in R/R CLL/SLL MRD- CR, time to first therapy NCT03516617 (Phase 2) Acalabrutinib ± obinutuzumab in patients with early stage CLL/SLL MRD- CR NCT03580928 (AVO; Phase 2) Venetoclax + obinutuzumab + acalabrutinib in patients with newly diagnosed CLL/SLL PFS NCT02477696 (Elevate CLL R/R) Acalabrutinib vs ibrutinib in previously treated patients with high-risk CLL/SLL Key: ˜ Active, not recruiting ˜ Currently recruiting Acalabrutinib Primary Endpoint(s) Approved BTK inhibitors currently include ibrutinib, acalabrutinib, and zanubrutinib Phase 3 Clinical Trials NCCN Recommendation4 The NCCN believes that the best management for any patient with cancer is in a clinical trial. Participation in a clinical trial is especially encouraged.
