Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL
•
1 like•104 views
Nathan H. Fowler, MD, Matthew S. Davids, MD, MMSc, and Sonali M. Smith, MD, prepared useful practice aids pertaining to B-cell malignancies for this CME activity titled "Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2PZOP99. CME credit will be available until October 8, 2019.
Report
Share
Report
Share
Download to read offline
Recommended
Nathan H. Fowler, MD, prepared useful Practice Aids pertaining to lymphoma management for this CME/CNE activity titled "Building Innovative Treatment Options for Patients With B-Cell Malignancies: Practical Insights on Clinical Evidence and Integration Strategies." For the full presentation, monograph, complete CME/CNE information, and to apply for credit, please visit us at http://bit.ly/2PfAYe2. CME/CNE credit will be available until February 14, 2020.Building Innovative Treatment Options for Patients With B-Cell Malignancies: ...

Building Innovative Treatment Options for Patients With B-Cell Malignancies: ...PVI, PeerView Institute for Medical Education
Recommended
Nathan H. Fowler, MD, prepared useful Practice Aids pertaining to lymphoma management for this CME/CNE activity titled "Building Innovative Treatment Options for Patients With B-Cell Malignancies: Practical Insights on Clinical Evidence and Integration Strategies." For the full presentation, monograph, complete CME/CNE information, and to apply for credit, please visit us at http://bit.ly/2PfAYe2. CME/CNE credit will be available until February 14, 2020.Building Innovative Treatment Options for Patients With B-Cell Malignancies: ...

Building Innovative Treatment Options for Patients With B-Cell Malignancies: ...PVI, PeerView Institute for Medical Education
The Importance of Biomarkers in Hematology/Oncology Drug Development - Presentation by Steven Fruchtman, Former Chief Medical Officer, Syndax Pharmaceuticals, at the marcus evans Evolution Summit 2014 held in Palm Beach, FL May 7-9The Importance of Biomarkers in Hematology/Oncology Drug Development - Steven...

The Importance of Biomarkers in Hematology/Oncology Drug Development - Steven...Life Sciences Network marcus evans
LLA 2011 - L. Mazzucchelli - Principles of pathology and microscopic diagnosi...

LLA 2011 - L. Mazzucchelli - Principles of pathology and microscopic diagnosi...European School of Oncology
Justin F. Gainor, MD; Kurt Schalper, MD, PhD; and Edward B. Garon, MD, MS prepared useful Practice Aids pertaining to immunotherapy for this CME/MOC/CC/CNE activity titled, "New Frontiers in Precision Immuno-Oncology: Leveraging Biomarkers to Refine and Expand the Use of Cancer Immunotherapies and Combinations." For the full presentation, monograph, complete CME/MOC/CC/CNE information, and to apply for credit, please visit us at http://bit.ly/2UJuQBq. CME/MOC/CC/CNE credit will be available until April 25, 2020.New Frontiers in Precision Immuno-Oncology: Leveraging Biomarkers to Refine a...

New Frontiers in Precision Immuno-Oncology: Leveraging Biomarkers to Refine a...PVI, PeerView Institute for Medical Education
Michael Wang, MD, and Michael J. Keating, MB, BS, prepared useful Practice Aids pertaining to B-cell malignancies for this CME activity titled "Integrating BTK Inhibitors Into the Management of B-Cell Malignancies: How Is Evidence Driving Patient Care?" For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2zChqfa. CME credit will be available until October 11, 2019.Integrating BTK Inhibitors Into the Management of B-Cell Malignancies: How Is...

Integrating BTK Inhibitors Into the Management of B-Cell Malignancies: How Is...PVI, PeerView Institute for Medical Education
John P. Leonard, MD, John C. Byrd, MD, Professor Dr. med Georg Lenz, and David G. Maloney, MD, PhD, discuss non-Hodgkin lymphoma management in this CME activity titled, "The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal Guide to Integrating New Therapy Into Patient Care." For the full presentation, downloadable Practice Aids, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2M7vUqT. CME credit will be available until July 4, 2019.The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...

The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...PVI, PeerView Institute for Medical Education
More Related Content
What's hot
The Importance of Biomarkers in Hematology/Oncology Drug Development - Presentation by Steven Fruchtman, Former Chief Medical Officer, Syndax Pharmaceuticals, at the marcus evans Evolution Summit 2014 held in Palm Beach, FL May 7-9The Importance of Biomarkers in Hematology/Oncology Drug Development - Steven...

The Importance of Biomarkers in Hematology/Oncology Drug Development - Steven...Life Sciences Network marcus evans
LLA 2011 - L. Mazzucchelli - Principles of pathology and microscopic diagnosi...

LLA 2011 - L. Mazzucchelli - Principles of pathology and microscopic diagnosi...European School of Oncology
Justin F. Gainor, MD; Kurt Schalper, MD, PhD; and Edward B. Garon, MD, MS prepared useful Practice Aids pertaining to immunotherapy for this CME/MOC/CC/CNE activity titled, "New Frontiers in Precision Immuno-Oncology: Leveraging Biomarkers to Refine and Expand the Use of Cancer Immunotherapies and Combinations." For the full presentation, monograph, complete CME/MOC/CC/CNE information, and to apply for credit, please visit us at http://bit.ly/2UJuQBq. CME/MOC/CC/CNE credit will be available until April 25, 2020.New Frontiers in Precision Immuno-Oncology: Leveraging Biomarkers to Refine a...

New Frontiers in Precision Immuno-Oncology: Leveraging Biomarkers to Refine a...PVI, PeerView Institute for Medical Education
What's hot (19)
Principles of cancer chemotherapy: a deep insight by RxVichuZ!

Principles of cancer chemotherapy: a deep insight by RxVichuZ!
The Importance of Biomarkers in Hematology/Oncology Drug Development - Steven...

The Importance of Biomarkers in Hematology/Oncology Drug Development - Steven...
LLA 2011 - L. Mazzucchelli - Principles of pathology and microscopic diagnosi...

LLA 2011 - L. Mazzucchelli - Principles of pathology and microscopic diagnosi...
New Frontiers in Precision Immuno-Oncology: Leveraging Biomarkers to Refine a...

New Frontiers in Precision Immuno-Oncology: Leveraging Biomarkers to Refine a...
DOUBLE HIT AND OTHER MOLECULARLY DEFINED LARGE CELL LYMPHOMAS

DOUBLE HIT AND OTHER MOLECULARLY DEFINED LARGE CELL LYMPHOMAS
cancer genetics, tumor marker and targeted therapy in cancer

cancer genetics, tumor marker and targeted therapy in cancer
Similar to Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL
Michael Wang, MD, and Michael J. Keating, MB, BS, prepared useful Practice Aids pertaining to B-cell malignancies for this CME activity titled "Integrating BTK Inhibitors Into the Management of B-Cell Malignancies: How Is Evidence Driving Patient Care?" For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2zChqfa. CME credit will be available until October 11, 2019.Integrating BTK Inhibitors Into the Management of B-Cell Malignancies: How Is...

Integrating BTK Inhibitors Into the Management of B-Cell Malignancies: How Is...PVI, PeerView Institute for Medical Education
John P. Leonard, MD, John C. Byrd, MD, Professor Dr. med Georg Lenz, and David G. Maloney, MD, PhD, discuss non-Hodgkin lymphoma management in this CME activity titled, "The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal Guide to Integrating New Therapy Into Patient Care." For the full presentation, downloadable Practice Aids, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2M7vUqT. CME credit will be available until July 4, 2019.The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...

The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...PVI, PeerView Institute for Medical Education
Andre H. Goy, MD, Richard R. Furman, MD, Krish Patel, MD, and Deborah M. Stephens, DO, prepared useful practice aids pertaining to B-cell malignancies for this CME activity titled "How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, and Beyond—Therapeutic Selection, Sequencing, and Next Steps." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2P5FeQk. CME credit will be available until December 29, 2020.How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, ...

How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, ...PVI, PeerView Institute for Medical Education
Chair & Presenter, Matthew S. Davids, MD, MMSc, Nitin Jain, MD, and Deborah M. Stephens, DO, prepared useful Practice Aids pertaining to chronic lymphocytic leukemia for this CME/MOC activity titled “Pushing Therapeutic Boundaries in CLL: The Convergence of Targeted Therapy, Novel Combinations, and Precision Medicine in Patient Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3ehHSPs. CME/MOC credit will be available until July 1, 2022.
Pushing Therapeutic Boundaries in CLL: The Convergence of Targeted Therapy, N...

Pushing Therapeutic Boundaries in CLL: The Convergence of Targeted Therapy, N...PVI, PeerView Institute for Medical Education
Andre H. Goy, MD, Owen A. O’Connor, MD, PhD, Stephen D. Smith, MD, and Michael Wang, MD, prepared useful practice aids pertaining to B-cell malignancies for this CME activity titled "Exploring the Present and Future of BTK Inhibition in B-Cell Malignancies: Expert Insights on Practical Implications for Patient Management." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2rvynmE. CME credit will be available until December 20, 2019.Exploring the Present and Future of BTK Inhibition in B-Cell Malignancies: Ex...

Exploring the Present and Future of BTK Inhibition in B-Cell Malignancies: Ex...PVI, PeerView Institute for Medical Education
Luciano J. Costa, MD, PhD, prepared useful Practice Aids pertaining to multiple myeloma for this CME activity titled "Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact of Novel Platforms and Agent Classes Across the Spectrum of Care." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2Dypn7b. CME credit will be available until March 12, 2020.Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...

Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...PVI, PeerView Institute for Medical Education
Kurt Schalper, MD, PhD, and Edward Garon, MD, MS, prepared useful Practice Aids pertaining to immuno-oncology biomarkers for this CME/MOC activity titled "Advances and Challenges in Refining the Use of Cancer Immunotherapies Through Biomarker Testing: Practical Guidance for Pathologists on the Front Lines of the Immuno-Oncology Revolution." For the full presentation, monograph, complete CME/MOC information, and to apply for credit, please visit us at http://bit.ly/2DE3X9J. CME/MOC credit will be available until December 2, 2019.Advances and Challenges in Refining the Use of Cancer Immunotherapies Through...

Advances and Challenges in Refining the Use of Cancer Immunotherapies Through...PVI, PeerView Institute for Medical Education
Chair, Anthony R. Mato, MD, MSCE, Kristen Battiato, MSN, RN, AGNP-C, Dipti Gupta, MD, MPH, and Amber C. King, PharmD, BCOP, prepared useful Practice Aids pertaining to B-cell cancers for this CME/MOC/NCPD activity titled “Interprofessional Perspectives on Safety Management With Targeted Therapy for B-Cell Malignancies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD information, and to apply for credit, please visit us at http://bit.ly/3cHX3zm. CME/MOC/NCPD credit will be available until October 3, 2023.Interprofessional Perspectives on Safety Management With Targeted Therapy for...

Interprofessional Perspectives on Safety Management With Targeted Therapy for...PVI, PeerView Institute for Medical Education
Naval Daver, MD, prepared useful practice aids pertaining to acute myeloid leukemia for this CME/CE activity titled "Groundbreaking Treatment Options for AML: How to Personalize Patient Care With New and Emerging Therapies." For the full presentation, monograph, complete CME/CE information, and to apply for credit, please visit us at http://bit.ly/2FCo09Y. CME/CE credit will be available until March 20, 2019.Groundbreaking Treatment Options for AML: How to Personalize Patient Care Wit...

Groundbreaking Treatment Options for AML: How to Personalize Patient Care Wit...PVI, PeerView Institute for Medical Education
Carlos E. Bueso-Ramos, MD, PhD, and Naval Daver, MD, prepared useful practice aids pertaining to AML for this CME/MOC/CC activity titled Pathology Insights on Innovation in AML: The Rapid Emergence of Precision Diagnostics & Novel Therapy Across the Spectrum of Care. For the full presentation, monograph, complete CME/MOC/CC information, and to apply for credit, please visit us at http://bit.ly/2mhXbz6. CME/MOC/CC credit will be available until October 27, 2020.Pathology Insights on Innovation in AML: The Rapid Emergence of Precision Dia...

Pathology Insights on Innovation in AML: The Rapid Emergence of Precision Dia...PVI, PeerView Institute for Medical Education
Similar to Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL (20)
Integrating BTK Inhibitors Into the Management of B-Cell Malignancies: How Is...

Integrating BTK Inhibitors Into the Management of B-Cell Malignancies: How Is...
The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...

The Arrival of Novel Agent Classes in B-Cell Non-Hodgkin Lymphoma: A Personal...
New Directions in the Management of Relapsed/Refractory Follicular Lymphoma

New Directions in the Management of Relapsed/Refractory Follicular Lymphoma
How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, ...

How I Think, How I Treat: BTK Inhibitors as a Clinical Strategy in CLL, MCL, ...
Pushing Therapeutic Boundaries in CLL: The Convergence of Targeted Therapy, N...

Pushing Therapeutic Boundaries in CLL: The Convergence of Targeted Therapy, N...
Exploring the Present and Future of BTK Inhibition in B-Cell Malignancies: Ex...

Exploring the Present and Future of BTK Inhibition in B-Cell Malignancies: Ex...
Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...

Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...
Advances and Challenges in Refining the Use of Cancer Immunotherapies Through...

Advances and Challenges in Refining the Use of Cancer Immunotherapies Through...
Stem Cell Transplantation in Hodgkin’s Lymphoma Past, Present and Future

Stem Cell Transplantation in Hodgkin’s Lymphoma Past, Present and Future
Interprofessional Perspectives on Safety Management With Targeted Therapy for...

Interprofessional Perspectives on Safety Management With Targeted Therapy for...
Groundbreaking Treatment Options for AML: How to Personalize Patient Care Wit...

Groundbreaking Treatment Options for AML: How to Personalize Patient Care Wit...
Tailoring Therapy for Follicular Lymphoma Based on the Latest Evidence

Tailoring Therapy for Follicular Lymphoma Based on the Latest Evidence
Pathology Insights on Innovation in AML: The Rapid Emergence of Precision Dia...

Pathology Insights on Innovation in AML: The Rapid Emergence of Precision Dia...
More from PVI, PeerView Institute for Medical Education
Chair Jamie Carroll, APRN, CNP, MSN, discusses breast cancer in this NCPD/ILNA/AAPA activity titled “Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy in TNBC and HR+, HER2- Breast Cancer: Best Practices for Adverse Event Management and Patient Education.” For the full presentation, downloadable Practice Aids, and complete NCPD/ILNA/AAPA information, and to apply for credit, please visit us at https://bit.ly/3SdnvWt. NCPD/ILNA/AAPA credit will be available until May 8, 2025.Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy...

Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy...PVI, PeerView Institute for Medical Education
Chair Jonathan A. Bernstein, MD, discusses chronic spontaneous urticaria in this CME activity titled “BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Treatment.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3P0cnvi. CME credit will be available until May 6, 2025.BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Tr...

BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Tr...PVI, PeerView Institute for Medical Education
Co-Chairs Milind Desai, MD, MBA, FACC, FAHA, FESC, and Andrew Willeford, PharmD, PhD, BCCP, prepared useful Practice Aids pertaining to hypertrophic cardiomyopathy for this CME/MOC/CPE/AAPA/IPCE activity titled “Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertrophic Cardiomyopathy.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4bb7eKc. CME/MOC/CPE/AAPA/IPCE credit will be available until May 16, 2025.Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...

Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...PVI, PeerView Institute for Medical Education
Co-Chairs Milind Desai, MD, MBA, FACC, FAHA, FESC, and Andrew Willeford, PharmD, PhD, BCCP, discuss hypertrophic cardiomyopathy in this CME/MOC/CPE/AAPA/IPCE activity titled “Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertrophic Cardiomyopathy.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4bb7eKc. CME/MOC/CPE/AAPA/IPCE credit will be available until May 16, 2025.Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...

Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...PVI, PeerView Institute for Medical Education
Chair A. Michael Lincoff, MD, discusses obesity in this CME activity titled “Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Weight Loss Pharmacotherapy.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3KAO98K. CME credit will be available until April 25, 2025.Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...PVI, PeerView Institute for Medical Education
Co-Chairs and Planners Saakshi Khattri, MBBS, MD, FAAD, FACR, Marla Dubinsky, MD, Emma Guttman-Yassky, MD, PhD, and Alexis Ogdie, MD, MSCE, discuss immune-mediated inflammatory diseases in this CME/AAPA activity titled “Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Diseases: Addressing Shared Pathophysiology With JAK Inhibitors.” For the full presentation, downloadable Practice Aids, and complete CME/AAPA information, and to apply for credit, please visit us at https://bit.ly/3JhsIZ7. CME/AAPA credit will be available until April 24, 2025.Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...PVI, PeerView Institute for Medical Education
Co-Chairs Alicia K. Morgans, MD, MPH, and Neal D. Shore, MD, FACS, discuss prostate cancer in this CME/MOC/NCPD/CPE/AAPA/IPCE activity titled “Treatment Advances and Individualized Therapeutic Strategies in Prostate Cancer: Expert Insights on Key Evidence, Practical Tips for Personalized Therapy, and Clinical Integration Approaches.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SQrJ6G. CME/MOC/NCPD/CPE/AAPA/IPCE credit will be available until April 24, 2025.Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...

Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...PVI, PeerView Institute for Medical Education
Co-Chairs Prof. Nicolas Girard, MD, PhD, and Aaron Lisberg, MD, discuss NSCLC in this CME/MOC/NCPD/AAPA/IPCE activity titled “Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Cancer: Unveiling Potential, Shaping Tomorrow.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3RmX3dU. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 24, 2025.Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...

Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...PVI, PeerView Institute for Medical Education
Co-Chairs, Joseph K. Han, MD, and Seth J. Isaacs, MD, prepared useful Practice Aids pertaining to chronic rhinosinusitis with nasal polyps for this CME/MOC/CC/AAPA/IPCE activity titled “Biologics in CRSwNP: Putting a Paradigm Shift Into Practice.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3Tq6n1G. CME/MOC/CC/AAPA/IPCE credit will be available until May 6, 2025.Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into PracticePVI, PeerView Institute for Medical Education
Co-Chairs, Joseph K. Han, MD, and Seth J. Isaacs, MD, discuss chronic rhinosinusitis with nasal polyps in this CME/MOC/CC/AAPA/IPCE activity titled “Biologics in CRSwNP: Putting a Paradigm Shift Into Practice.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3Tq6n1G. CME/MOC/CC/AAPA/IPCE credit will be available until May 6, 2025.Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into PracticePVI, PeerView Institute for Medical Education
Co-Chairs, R. Donald Harvey, PharmD, BCOP, FCCP, FHOPA, FASCO, Zahra Mahmoudjafari, PharmD, MBA, BCOP, FHOPA, and James Davis, PharmD, BCOP, discuss multiple myeloma in this CME/CPE/IPCE activity titled “Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering Effective Therapy With Antibody Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/CPE/IPCE information, and to apply for credit, please visit us at https://bit.ly/4aa0iMX. CME/CPE/IPCE credit will be available until May 2, 2025.Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...

Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...PVI, PeerView Institute for Medical Education
Co-Chairs, Carlos G. Romo, MD, and Aimee Sato, MD, discuss Neurofibromatosis in this CME/MOC activity titled “Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & Multimodal Care for NF1 pNF and Other Tumors.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3SZRz8p. CME/MOC credit will be available until May 2, 2025.Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...

Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...PVI, PeerView Institute for Medical Education
Chair and Presenters Kathleen N. Moore, MD, MS, Floor J. Backes, MD, and Bhavana Pothuri, MD, MS, prepared useful Practice Aids pertaining to endometrial cancer for this CME/MOC/NCPD/AAPA/IPCE activity titled “Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Potential of Immunotherapy, ADCs, PARP Inhibitors, and Other Emerging Treatment Strategies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SjJyuH. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 17, 2025.Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...PVI, PeerView Institute for Medical Education
Chair and Presenters Kathleen N. Moore, MD, MS, Floor J. Backes, MD, and Bhavana Pothuri, MD, MS, discuss endometrial cancer in this CME/MOC/NCPD/AAPA/IPCE activity titled “Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Potential of Immunotherapy, ADCs, PARP Inhibitors, and Other Emerging Treatment Strategies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SjJyuH. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 17, 2025.Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...PVI, PeerView Institute for Medical Education
Chair and Presenters Bradley J. Monk, MD, FACS, FACOG, Kathleen N. Moore, MD, MS, and Ana Oaknin, MD, PhD, discuss gynecologic cancers in this CME/MOC/NCPD/AAPA/IPCE activity titled “Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Implementation Strategies, and Patient Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4a56tly. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 16, 2025.Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...

Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...PVI, PeerView Institute for Medical Education
Chair Lecia V. Sequist, MD, MPH, and Patrick Nana-Sinkam, MD, FCCP, prepared useful Practice Aids pertaining to lung cancer for this CME/MOC/AAPA/IPCE activity titled “Screening and Early Intervention as the Keys to Success in Lung Cancer: A Practical Approach to Implementing Lung Cancer Screening for High-Risk Individuals.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/46VvwoP. CME/MOC/AAPA/IPCE credit will be available until April 16, 2025.Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...PVI, PeerView Institute for Medical Education
Chair Lecia V. Sequist, MD, MPH, and Patrick Nana-Sinkam, MD, FCCP, discuss lung cancer screening in this CME/MOC/AAPA/IPCE activity titled “Screening and Early Intervention as the Keys to Success in Lung Cancer: A Practical Approach to Implementing Lung Cancer Screening for High-Risk Individuals.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/46VvwoP. CME/MOC/AAPA/IPCE credit will be available until April 16, 2025.Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...PVI, PeerView Institute for Medical Education
Chair and Presenter, Olalekan Oluwole, MBBS, MPH, Veronika Bachanova, MD, PhD, and David L. Porter, MD, prepared useful Practice Aids pertaining to CAR-T therapy for this CME/NCPD activity titled “Democratizing the CAR-T Experience: The Principles and Practice of Outpatient Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/NCPD information, and to apply for credit, please visit us at https://bit.ly/3TfIABM. CME/NCPD credit will be available until April 15, 2025.Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...PVI, PeerView Institute for Medical Education
Chair and Presenter, Olalekan Oluwole, MBBS, MPH, Veronika Bachanova, MD, PhD, and David L. Porter, MD, discuss CAR-T therapy in this CME/NCPD activity titled “Democratizing the CAR-T Experience: The Principles and Practice of Outpatient Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/NCPD information, and to apply for credit, please visit us at https://bit.ly/3TfIABM. CME/NCPD credit will be available until April 15, 2025.Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...PVI, PeerView Institute for Medical Education
Co-Chairs Lipika Goyal, MD, MPhil, and Riad Salem, MD, MBA, discuss HCC in this CME activity titled “The Convergence of Interventional Radiologists and Oncologists in HCC: Shared Decision-Making and Care Coordination at the Center of Personalized Care Across the Disease Continuum.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/48BAasz. CME credit will be available until April 26, 2025.The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...

The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...PVI, PeerView Institute for Medical Education
More from PVI, PeerView Institute for Medical Education (20)
Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy...

Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy...
BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Tr...

BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Tr...
Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...

Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...
Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...

Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...
Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...
Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...
Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...

Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...
Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...

Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...
Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into Practice
Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into Practice
Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...

Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...
Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...

Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...
Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...
Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...
Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...

Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...
Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...
Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...
Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...
Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...
The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...

The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...
Recently uploaded
PEMESANAN OBAT ASLI : +6287776558899
Cara Menggugurkan Kandungan usia 1 , 2 , bulan - obat penggugur janin - cara aborsi kandungan - obat penggugur kandungan 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 bulan - bagaimana cara menggugurkan kandungan - tips Cara aborsi kandungan - trik Cara menggugurkan janin - Cara aman bagi ibu menyusui menggugurkan kandungan - klinik apotek jual obat penggugur kandungan - jamu PENGGUGUR KANDUNGAN - WAJIB TAU CARA ABORSI JANIN - GUGURKAN KANDUNGAN AMAN TANPA KURET - CARA Menggugurkan Kandungan tanpa efek samping - rekomendasi dokter obat herbal penggugur kandungan - ABORSI JANIN - aborsi kandungan - jamu herbal Penggugur kandungan - cara Menggugurkan Kandungan yang cacat - tata cara Menggugurkan Kandungan - obat penggugur kandungan di apotik kimia Farma - obat telat datang bulan - obat penggugur kandungan tuntas - obat penggugur kandungan alami - klinik aborsi janin gugurkan kandungan - ©Cytotec ™misoprostol BPOM - OBAT PENGGUGUR KANDUNGAN ®CYTOTEC - aborsi janin dengan pil ©Cytotec - ®Cytotec misoprostol® BPOM 100% - penjual obat penggugur kandungan asli - klinik jual obat aborsi janin - obat penggugur kandungan di klinik k-24 || obat penggugur ™Cytotec di apotek umum || ®CYTOTEC ASLI || obat ©Cytotec yang asli 200mcg || obat penggugur ASLI || pil Cytotec© tablet || cara gugurin kandungan || jual ®Cytotec 200mcg || dokter gugurkan kandungan || cara menggugurkan kandungan dengan cepat selesai dalam 24 jam secara alami buah buahan || usia kandungan 1_2 3_4 5_6 7_8 bulan masih bisa di gugurkan || obat penggugur kandungan ®cytotec dan gastrul || cara gugurkan pembuahan janin secara alami dan cepat || gugurkan kandungan || gugurin janin || cara Menggugurkan janin di luar nikah || contoh aborsi janin yang benar || contoh obat penggugur kandungan asli || contoh cara Menggugurkan Kandungan yang benar || telat haid || obat telat haid || Cara Alami gugurkan kehamilan || obat telat menstruasi || cara Menggugurkan janin anak haram || cara aborsi menggugurkan janin yang tidak berkembang || gugurkan kandungan dengan obat ©Cytotec || obat penggugur kandungan ™Cytotec 100% original || HARGA obat penggugur kandungan || obat telat haid 1 bulan || obat telat menstruasi 1-2 3-4 5-6 7-8 BULAN || obat telat datang bulan || cara Menggugurkan janin 1 bulan || cara Menggugurkan Kandungan yang masih 2 bulan || cara Menggugurkan Kandungan yang masih hitungan Minggu || cara Menggugurkan Kandungan yang masih usia 3 bulan || cara Menggugurkan usia kandungan 4 bulan || cara Menggugurkan janin usia 5 bulan || cara Menggugurkan kehamilan 6 Bulan
________&&&_________&&&_____________&&&_________&&&&____________
Cara Menggugurkan Kandungan Usia Janin 1 | 7 | 8 Bulan Dengan Cepat Dalam Hitungan Jam Secara Alami, Kami Siap Meneriman Pesanan Ke Seluruh Indonesia, Melputi: Ambon, Banda Aceh, Bandung, Banjarbaru, Batam, Bau-Bau, Bengkulu, Binjai, Blitar, Bontang, Cilegon, Cirebon, Depok, Gorontalo, Jakarta, Jayapura, Kendari, Kota Mobagu, Kupang, LhokseumaweCara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...

Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...Cara Menggugurkan Kandungan 087776558899
❤️ Chandigarh Call Girls☎️98151-579OO☎️ Call Girl service in Chandigarh ☎️ Chandigarh Call Girls Service ☎️ Call Girls In Chandigarh BEST CALL GIRL ESCORTS SERVICE IN CHANDIGARH CALL WATTSAPP 98151-579OO THE MOST BEAUTIFUL INDEPENDENT ESCORT CALL GIRL SERVICE In Chandigarh WE ARE PROVIDING GENUINE CALL GIRL SERVICE
I AM A a NATURAL BRUNETTES, SLIM BODY, NATURAL LONG HAIR AND ALL TYPE OF HAIR IS A NATURAL BRUNETTE IN THE MOST BEAUTIFUL MODELS INDEPENDENT ESCORT GIRL I AM A NATURAL BRUNETTE WITH ROOM AND HOTEL AND A NATURAL BRUNETTE WITH A BODY MADE FOR SIN AND ALL TYPE OF ME ALL THE TIME
I SEND YOU A HAIR, VERY SOCIABLE AND FUNNY, READY TO ENTERTAIN TO ENTERTAIN U AND MAKE FORGET ABOUT TO AGET ENTERTAINMENT YOU AND MAKE FORGET ABOUT ALL THE PROBLEMS. LET'S HAVE A WONDERFUL TIME TOGETHER AND FORGET ABOUT EVERYTHING ALL TYPE SERVICE ENJOYMENT SAFE AND SECURE IN CALL OUT CALL HOME AND HOTEL ANYTIME AVAILABLE
AND ALL TYPE SERVICE ENJOYMENTPANCHKULA INDEPENDENT BEST CALL GIRL ESCORTS SERVICE IN PANCHKULA INDEPENDENT CALL GIRL Chandigarh Call Girls In Chandigarh BEST Call Girls in CHANDIGARH Escort Service provide Cute Nice sweet and Sexy Models in beautiful CHANDIGARH city cash in hand to hand call girl in CHANDIGARH and CHANDIGARH escorts. HOT & SEXY MODELS // COLLEGE GIRLS IN CHANDIGARH AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME ★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE ★ 100% SATISFACTION,UNLIMITED ENJOYMENT. ★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE Profiles Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & 24x7 :: 3 * 5 *7 *Star Hotel Service .In Call & Out call
Services :
★ A-Level (5 star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without a Condom)-❤️ Chandigarh Call Girls Service☎️98151-579OO☎️ Call Girl service in Chandigarh ☎️ Chandigarh Call Girls Service ☎️ Call Girls In Chandigarh❤️ Chandigarh Call Girls☎️98151-579OO☎️ Call Girl service in Chandigarh ☎️ Ch...

❤️ Chandigarh Call Girls☎️98151-579OO☎️ Call Girl service in Chandigarh ☎️ Ch...Rashmi Entertainment
Recently uploaded (20)
Lucknow Call Girls Service { 9984666624 } ❤️VVIP ROCKY Call Girl in Lucknow U...

Lucknow Call Girls Service { 9984666624 } ❤️VVIP ROCKY Call Girl in Lucknow U...
Lucknow Call Girls Just Call 👉👉8630512678 Top Class Call Girl Service Available

Lucknow Call Girls Just Call 👉👉8630512678 Top Class Call Girl Service Available
Call Girls Mussoorie Just Call 8854095900 Top Class Call Girl Service Available

Call Girls Mussoorie Just Call 8854095900 Top Class Call Girl Service Available
Call girls Service Phullen / 9332606886 Genuine Call girls with real Photos a...

Call girls Service Phullen / 9332606886 Genuine Call girls with real Photos a...
7 steps How to prevent Thalassemia : Dr Sharda Jain & Vandana Gupta

7 steps How to prevent Thalassemia : Dr Sharda Jain & Vandana Gupta
Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...

Cara Menggugurkan Kandungan Dengan Cepat Selesai Dalam 24 Jam Secara Alami Bu...
❤️ Chandigarh Call Girls☎️98151-579OO☎️ Call Girl service in Chandigarh ☎️ Ch...

❤️ Chandigarh Call Girls☎️98151-579OO☎️ Call Girl service in Chandigarh ☎️ Ch...
💰Call Girl In Bangalore☎️63788-78445💰 Call Girl service in Bangalore☎️Bangalo...

💰Call Girl In Bangalore☎️63788-78445💰 Call Girl service in Bangalore☎️Bangalo...
Race Course Road } Book Call Girls in Bangalore | Whatsapp No 6378878445 VIP ...

Race Course Road } Book Call Girls in Bangalore | Whatsapp No 6378878445 VIP ...
Call Girl in Chennai | Whatsapp No 📞 7427069034 📞 VIP Escorts Service Availab...

Call Girl in Chennai | Whatsapp No 📞 7427069034 📞 VIP Escorts Service Availab...
Call Girls in Lucknow Just Call 👉👉 8875999948 Top Class Call Girl Service Ava...

Call Girls in Lucknow Just Call 👉👉 8875999948 Top Class Call Girl Service Ava...
Call Girls Bangalore - 450+ Call Girl Cash Payment 💯Call Us 🔝 6378878445 🔝 💃 ...

Call Girls Bangalore - 450+ Call Girl Cash Payment 💯Call Us 🔝 6378878445 🔝 💃 ...
💞 Safe And Secure Call Girls Coimbatore🧿 6378878445 🧿 High Class Coimbatore C...

💞 Safe And Secure Call Girls Coimbatore🧿 6378878445 🧿 High Class Coimbatore C...
Cardiac Output, Venous Return, and Their Regulation

Cardiac Output, Venous Return, and Their Regulation
Call 8250092165 Patna Call Girls ₹4.5k Cash Payment With Room Delivery

Call 8250092165 Patna Call Girls ₹4.5k Cash Payment With Room Delivery
Call Girls Kathua Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Kathua Just Call 8250077686 Top Class Call Girl Service Available
ANATOMY AND PHYSIOLOGY OF REPRODUCTIVE SYSTEM.pptx

ANATOMY AND PHYSIOLOGY OF REPRODUCTIVE SYSTEM.pptx
👉 Chennai Sexy Aunty’s WhatsApp Number 👉📞 7427069034 👉📞 Just📲 Call Ruhi Colle...

👉 Chennai Sexy Aunty’s WhatsApp Number 👉📞 7427069034 👉📞 Just📲 Call Ruhi Colle...
Call Girls Rishikesh Just Call 9667172968 Top Class Call Girl Service Available

Call Girls Rishikesh Just Call 9667172968 Top Class Call Girl Service Available
Bhawanipatna Call Girls 📞9332606886 Call Girls in Bhawanipatna Escorts servic...

Bhawanipatna Call Girls 📞9332606886 Call Girls in Bhawanipatna Escorts servic...
Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL
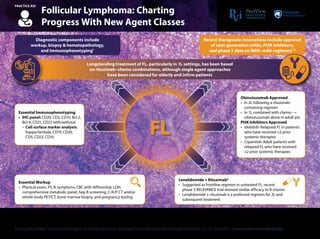
- 1. Bcl-2: B-cell lymphoma 2; C/A/P: chest/abdominal/pelvic; CBC: complete blood count; CD: cluster of differentiation; chemo: chemotherapy; CT: computed tomography; FL: follicular lymphoma; hep: hepatitis; IHC: immunohistochemistry; LDH: lactate dehydrogenase; PI3K: phosphatidylinositol-3 kinase; PET: positron emission tomography; PS: performance status; R: rituximab. 1. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology. B-Cell Lymphomas. Version 4.2018. 2. Aliqopa (copanlisib) Prescribing Information. http://labeling.bayerhealthcare.com/html/products/pi/Aliqopa_PI.pdf. Accessed August 14, 2018. 3. Zydelig (idelalisib) Prescribing Information. http://www.gilead.com/~/media/Files/pdfs/medicines/oncology/zydelig/zydelig_pi.pdf. Accessed August 14, 2018. 4. Fowler N et al. 2018 American Society of Clinical Oncology Annual Meeting (ASCO 2018). Abstract 7500. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Access the activity,“Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL,”at www.peerview.com/ZAJ40. Follicular Lymphoma: Charting Progress With New Agent Classes PRACTICE AID Diagnostic components include workup, biopsy & hematopathology, and immunophenotyping1 Essential Immunophenotyping • IHC panel: CD20, CD3, CD10, Bcl-2, Bcl-6, CD21, CD23 with/without: - Cell surface marker analysis: Kappa/lambda, CD19, CD20, CD5, CD23, CD10 Essential Workup • Physical exam, PS, B symptoms, CBC with differential, LDH, comprehensive metabolic panel, hep B screening, C/A/P CT and/or whole-body PET/CT, bone marrow biopsy, and pregnancy testing Lenalidomide + Rituximab4 • Suggested as frontline regimen in untreated FL; recent phase 3 RELEVANCE trial showed similar efficacy to R-chemo • Lenalidomide ± rituximab is a preferred regimen for 2L and subsequent treatment Obinutuzumab Approved • In 2L following a rituximab- containing regimen • In 1L combined with chemo → obinutuzumab alone in adult pts PI3K Inhibitors Approved • Idelalisib: Relapsed FL in patients who have received ≥2 prior systemic therapies • Copanlisib: Adult patients with relapsed FL who have received ≥2 prior systemic therapies Longstanding treatment of FL, particularly in 1L settings, has been based on rituximab–chemo combinations, although single agent approaches have been considered for elderly and infirm patients Recent therapeutic innovations include approval of next-generation mAbs, PI3K inhibitors, and phase 3 data on IMiD–mAb regimens1-3 FL
- 2. Bcl-2: B-cell lymphoma 2; BTK: Bruton tyrosine kinase; CBC: complete blood count; CD: cluster of differentiation; CLL: chronic lymphocytic leukemia; IHC: immunohistochemistry; PS: performance status. 1. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology. Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma. Version 1.2019. 2. Imbruvica (ibrutinib) Prescribing Information. https://www.imbruvica.com/docs/librariesprovider7/ default-document-library/prescribing-information.pdf. Accessed August 14, 2018. 3. Gazyva (obinutuzumab) Prescribing Information. https://www.gene.com/download/pdf/gazyva_prescribing.pdf. Accessed August 14, 2018. 4. Venclexta (venetoclax) Prescribing Information. https://www.rxabbvie.com/pdf/venclexta.pdf. Accessed August 14, 2018. 5. https://clinicaltrials.gov/ct2/show/NCT02048813. Accessed August 21, 2018. 6. https://clinicaltrials.gov/ct2/show/NCT02477696. Accessed August 21, 2018. https://clinicaltrials.gov/ct2/show/ NCT02475681. Accessed August 21, 2018. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Chronic Lymphocytic Leukemia: Charting Progress With New Agent Classes PRACTICE AID Access the activity,“Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL,”at www.peerview.com/ZAJ40. Diagnostic components include workup, biopsy & hematopathology, and immunophenotyping1 Essential Immunophenotyping • IHC panel: CD3, CD5, CD10, CD20, CD23, cyclin D1 with/without: - Cell surface marker analysis: Kappa/lambda, CD19, CD20, CD5, CD23, CD10 Essential Workup • Physical exam including measurement of liver and spleen size, PS, B symptoms, CBC with differential, and comprehensive metabolic panel Bcl-2 Inhibitor Approved • Venetoclax: for patients with 17p deletion, as detected by an FDA-approved test, who have received ≥1 prior therapy Obinutuzumab Approved • With chlorambucil for previously untreated CLL Ibrutinib Approved • Adult patients with CLL ± 17p deletion • Phase 3 study testing ibrutinib combinations in 1L CLL (E1912)5 Acalabrutinib6,7 • In phase 3 testing (Elevate CLL trials in 1L and 2L settings) • Recommended (NCCN 2018) for relapsed/refractory CLL in frail older pts (≥65 years) and younger patients (<65 years) Historically, treatment of symptomatic CLL was chemotherapy or immunochemotherapy based; in the past several years, novel agent classes have emerged and established roles as part of CLL care Recent therapeutic innovations include approval of next-generation mAbs, BTK inhibitors, and Bcl-2 inhibitors1-4 CLL
- 3. BR: bendamustine and rituximab; BTK: Bruton tyrosine kinase; C/A/P: chest/abdominal/pelvic; CD: cluster of differentiation; chemo: chemotherapy; CT: computed tomography; ECG: electrocardiogram; IHC: immunohistochemistry; IMiD: immunomodulatory drug; LDH: lactose dehydrogenase; MCL: mantle cell lymphoma; MUGA: multiple-gated acquisition; PET: positron emission tomography; PS: performance status; RT: radiation therapy. 1. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology. B-Cell Lymphomas. Version 4.2018. 2. Calquence (acalabrutinib) Prescribing Information. https://www.azpicentral.com/calquence/calquence.pdf#page=1. Accessed August 14, 2018. 3. Revlimid (lenalidomide) Prescribing Information. http://media.celgene.com/content/uploads/revlimid-pi.pdf. Accessed August 14, 2018. 4. https://clinicaltrials.gov/ct2/show/NCT03112174. Accessed August 21, 2018. 5. https://clinicaltrials.gov/ct2/show/NCT02972840. Accessed August 21, 2018. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Mantle Cell Lymphoma: Charting Progress With New Agent Classes PRACTICE AID Access the activity,“Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL,”at www.peerview.com/ZAJ40. Diagnostic components include workup, biopsy & hematopathology, and immunophenotyping1 Essential Immunophenotyping • IHC panel: CD20, CD3, CD5, cyclin D1, CD10, CD21, CD23, BCL2, BCL6, Ki-67 with/without: - Cell surface marker analysis: Kappa/lambda, CD19, CD20, CD5, CD23, CD10 Essential Workup • Physical exam, PS, B symptoms, CBC with differential, comprehensive metabolic panel, LDH, bone marrow biopsy ± aspirate, C/A/P CT and/or whole-body PET/CT, hep B screening (if rituximab contemplated), ECG or MUGA scan (if anthracycline or anthracenedione-based regimen is indicated) and pregnancy testing (if chemo or RT planned) IMiD Approved • Lenalidomide: Relapsed MCL in patients who have received ≥2 prior systemic therapies, one of which included bortezomib BTK Inhibitors Approved • Ibrutinib: Adult patients with relapsed MCL who have received ≥2 prior systemic therapies • Ibrutinib–venetoclax combination is being studied in the phase 3 SIMPATICO trial4 • Acalabrutinib: Adult patients with relapsed MCL who have received ≥1 prior therapy • Phase 3 study of 1L acalabrutinib + BR is underway5 Immunochemotherapy, often aggressive in nature, is a mainstay of 1L therapy in MCL; however, relapsed MCL has historically been characterized by poor response rates and overall survival; newer therapies have emerged in the 2L setting that are more active than traditional tx Recent therapeutic innovations include approval of BTK inhibitors and IMiDs1-3 MCL
- 4. Bcl-2: B-cell lymphoma 2; BID: twice a day; BTK: Bruton tyrosine kinase; C: cycle; CD: cluster of differentiation; cGVHD: chronic graft-versus-host disease; CLL: chronic lymphocytic leukemia; FL: follicular lymphoma; IMiD: immunomodulatory drug; MCL: mantle cell lymphoma; MZL: marginal zone lymphoma; PI3K: phosphatidylinositol-3 kinase; SLL: small lymphocytic lymphoma; TLS: tumor lysis syndrome; WM: Waldenstrom macroglobulinemia. 1. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology. B-Cell Lymphomas. Version 4.2018. 2. Imbruvica (ibrutinib) Prescribing Information. https://www.imbruvica.com/docs/librariesprovider7/default-document-library/prescribing- information.pdf. Accessed August 14, 2018. 3. Calquence (acalabrutinib) Prescribing Information. https://www.azpicentral.com/calquence/calquence.pdf#page=1. Accessed August 14, 2018. 4. Zydelig (idelalisib) Prescribing Information. http://www.gilead.com/~/media/Files/ pdfs/medicines/oncology/zydelig/zydelig_pi.pdf. Accessed August 14, 2018. 5. Aliqopa (copanlisib) Prescribing Information. http://labeling.bayerhealthcare.com/html/products/pi/Aliqopa_PI.pdf. Accessed August 14, 2018. 6. Revlimid (lenalidomide) Prescribing Information. http://media.celgene.com/content/uploads/revlimid-pi.pdf. Accessed August 14, 2018. 7. Gazyva (obinutuzumab) Prescribing Information. https://www.gene.com/download/pdf/gazyva_prescribing.pdf. Accessed August 14, 2018. 8. Venclexta (venetoclax) Prescribing Information. https://www.rxabbvie.com/pdf/venclexta.pdf. Accessed August 14, 2018. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Dosing Guide for Novel Therapies in FL, CLL, and MCL PRACTICE AID Access the activity,“Innovative Therapy in B-Cell Malignancies: An Expert Tumor Board on Novel Agent Classes in CLL, FL, and MCL,”at www.peerview.com/ZAJ40. Bcl-2 INHIBITOR Idelasib1,4 • CLL, FL: 150 mg PO BID Ibrutinib1,2 • CLL/SLL, WM, cGVHD: 420 mg PO daily continuously until progression of disease • MCL, MZL: 560 mg PO once daily Copanlisib1,5 • FL: 60 mg administered as a 1-h IV infusion on d 1, 8, and 15 of a 28-d treatment cycle on an intermittent schedule (3 wk on, 1 wk off) Lenalidomide1,6 • MCL: 25 mg once daily orally on d 1-21 of repeated 28-d cycles • Renal impairment: Adjust starting dose based on clinical and laboratory findings • CLL: 100 mg on d 1 + 900 mg on d 2 of C 1 + 1,000 mg on d 8 + 15 of C 1 + 1,000 mg on d 1 of C 2-6 • FL: 1,000 mg on d 1, 8, 15 of C 1 + 1,000 mg on d 1 of C 2-6 or C 2-8, and then 1,000 mg every 2 mo for up to 2 y Obinutuzumab1,7 Venetoclax1,8 • CLL: 400 mg PO daily until disease progression or unacceptable toxicity • Initiate at 20 mg for 1 wk; gradually escalate to target dose over 5 wk to reduce the risk of TLS • Consider re-initiating at a lower dose, then continue with dose escalation in pts who have tx interruption for >1 wk during escalation Acalabrutinib1,3 • MCL: 100 mg PO approximately every 12 hours; manage toxicities using treatment interruption, dose reduction, or discontinuation NEXT-GEN mAbIMiDPI3K INHIBITORSBTK INHIBITORS
