Interprofessional Perspectives on Safety Management With Targeted Therapy for B-Cell Malignancies
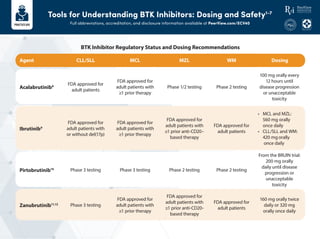
- 1. Tools for Understanding BTK Inhibitors: Dosing and Safety1-7 Full abbreviations, accreditation, and disclosure information available at PeerView.com/ECV40 CLL/SLL Agent Acalabrutinib8 Ibrutinib9 Pirtobrutinib10 Zanubrutinib11,12 MCL MZL WM Dosing FDA approved for adult patients FDA approved for adult patients with ≥1 prior therapy Phase 1/2 testing Phase 2 testing 100 mg orally every 12 hours until disease progression or unacceptable toxicity FDA approved for adult patients with or without del(17p) FDA approved for adult patients with ≥1 prior therapy FDA approved for adult patients with ≥1 prior anti-CD20– based therapy FDA approved for adult patients • MCL and MZL: 560 mg orally once daily • CLL/SLL and WM: 420 mgorally once daily Phase 3 testing FDA approved for adult patients with ≥1 prior therapy FDA approved for adult patients with ≥1 prior anti-CD20- based therapy FDA approved for adult patients 160 mg orally twice daily or 320 mg orally once daily Phase 3 testing Phase 3 testing Phase 2 testing Phase 2 testing From the BRUIN trial: 200 mg orally daily until disease progression or unacceptable toxicity BTK Inhibitor Regulatory Status and Dosing Recommendations
- 2. • Don’t give BTKi concomitantly with warfarin • For new-onset AF, consider nonwarfarin anticoagulation and monitor • Hypertension: manage with antihypertensives • Monitor for and manage cardiac arrhythmia/AF and treat appropriately • Monitor patients for signs of bleeding • Arthralgia: rule out other causes, monitor, and use supportive care for lower-grade events – Dose reduction once symptoms affect ADLs, dose holds for higher-grade arthralgia • Monitor for infections and secondary malignancies • Headaches commonly occur early in therapy with acalabrutinib and typically resolve in 1-2 months; manage with acetaminophen and caffeine • Monitor for neutropenia (particularly with zanubrutinib) In real-world settings, BTKi toxicity appears to be the most common reason for treatment discontinuation in the frontline and relapsed/refractory settings. More selective covalent BTK inhibitors appear to have fewer off-target effects leading to AEs versus ibrutinib.13 Understanding the spectrum of treatment-emergent AEs with BTK inhibitors is an important first step toward effective toxicity management Management approaches for BTKi AEs are available for all currently approved agents used in B-cell cancer settings Selected Clinical Notes Additional Important Toxicities Common Toxicities Dermatologic changes Atrial fibrillation Arthralgia Infection Ventricular arrhythmia Fatigue Diarrhea Hypertension Bleeding Cytopenias Tools for Understanding BTK Inhibitors: Dosing and Safety1-7 Full abbreviations, accreditation, and disclosure information available at PeerView.com/ECV40 1. NCCN Clinical Practice Guidelines in Oncology: Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma. Version 1.2022. https://www.nccn.org/professionals/physician_gls/pdf/cll.pdf. 2. Lipsky A, Lamanna N. Hematology Am Soc Hematol Educ Program. 2020;1:336-345. 3. Byrd JC et al. ASCO 2021. Abstract 7500. 4. Hillmen P et al. EHA 2021. Abstract LB1900. 5. Mato AR et al. Lancet. 2021;397:892-901. 6. Mato AR et al. Blood. 2016.28:2199-2205. 7. Mato AR et al. Haematologica. 2018;103:874-879. 8. Calquence (acalabrutinib) Prescribing Information. https://www.accessdata.fda. gov/drugsatfda_docs/label/2017/210259s000lbl.pdf. 9. Imbruvica (ibrutinib) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/205552s002lbl.pdf. 10. https://clinicaltrials.gov/ct2/show/NCT04666038. 11. https://www.cancernetwork.com/view/sequoia-trial- meets-primary-pfs-end-point-improvement-with-frontline-zanubrutinib-for-cll. 12. Brukinsa (zanubrutinib) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/213217s000lbl.pdf. 13. Kaptein A et al. ASH 2018. Abstract 1871.
- 3. Managing Ibrutinib-Associated AF1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/ECV40 1. Ganatra S et al. JACC Clin Electrophysiol. 2018;4:1491-1500. 12 Use beta blocker • Avoid non-DHP CCB (CYP450 3A4 inhibitor, increases ibrutinib level) • Avoid digoxin (P-gp substrate, ibrutinib increased digoxin level) • Add CCB with ibrutinib dose reduction and close monitoring or temporary withholding of ibrutinib after discussion with a hematologist OR • Add digoxin at lower dose 6 hours prior to or after ibrutinib dose with close monitoring of plasma levels and development of toxicity Persistent rapid ventricular rate, severe symptoms, persistent reduced LVEF, or refractory HF Continue beta blocker Ibrutinib-associated AF Assess suitability for anticoagulation • High CHADS2VASc score/low HAS-BLED score • No prior bleeding events • Normal platelets, Hb, and INR Consider anticoagulation • Choose an agent based on individual patient profile and preference – LMWH if normal Cr – Caution with Factor Xa inhibitors – Avoid direct thrombin inhibitors Rhythm control strategy (cardioversion and anti-arrhythmic medications) Assess echocardiogram, Cr, electrolytes, CBC, INR, and TSH Rate control strategy preferred due to: • High likelihood of recurrence on ibrutinib • Need for uninterrupted anticoagulation after cardioversion Hemodynamically unstable Hemodynamically stable Yes No Adequate rate control achieved
- 4. Tools for Understanding BCL-2 Inhibitor Dosing and Safety Full abbreviations, accreditation, and disclosure information available at PeerView.com/ECV40 1. Venclexta (venetoclax) Prescribing information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/208573s009lbl.pdf. Venetoclax Dosing Schedule for Ramp-Up Phase in Patients With CLL/SLL 400 mg 400 mg Week 5 20 mg Week 1 50 mg Week 2 100 mg Week 3 200 mg Week 4 Myelosuppression TLS GI events Infections • Manage with dose interruption/reduction • For grade ≥3, consider G-CSF and/or antibiotics • Assess risk in all patients • Pretreatment CT scan to assess internal lymphadenopathy • Premedicate with antihyperuricemics; ensure adequate hydration • Diarrhea: rule out infectious causes; treat with antidiarrheals and PO hydration • Nausea: adjust dose timing and use antiemetics • Grade 3/4: withhold until resolution and resume at same or reduced dose Venetoclax is FDA approved for the treatment of adult patients with CLL/SLL, with or without del(17p), who have received at ≥1 prior therapy1
- 5. CLL Society Resources Full abbreviations, accreditation, and disclosure information available at PeerView.com/ECV40 Ask the Experts CLL Support Groups Email Alerts for CLL Breaking News cllsociety.org/ask-the-expert cllsociety.org/cll-specific- patient-support-groups cllsociety.org/cll_alerts The mission of the CLL Society is to be an inclusive, patient-centric, physician-curated nonprofit organization that addresses the unmet needs of the CLL community through patient education, advocacy, support, and research. Here you will find highlights of helpful resources from cllsociety.org to better inform your practice. Test Before Treat™ Resources and Handouts cllsociety.org/cll-101/test-before-treat Current NCCN and iwCLL guidelines indicate the importance of obtaining biomarker testing before the first treatment and again before every subsequent therapy Patient Education Toolkit Binder cllsociety.org/patient-education-toolkit Free resource for healthcare providers and CLL/SLL patients TM
