Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on Molecular Biology, JAK Inhibitors, and Emerging Therapeutics
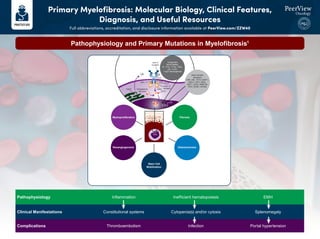
- 1. Pathophysiology and Primary Mutations in Myelofibrosis1 Pathophysiology Inflammation Inefficient hematopoiesis EMH Clinical Manifestations Constitutional systems Cytopenia(s) and/or cytosis Splenomegaly Complications Thromboembolism Infection Portal hypertension ! Myeloproliferation Neoangiogenesis Stem Cell Mobilization Osteosclerosis Fibrosis Cytogenetic Abnormalities +8, -7/7q-, i(17q), -5/5q-, 12p-, inv(3), 11q23 rearrangement ! ! ! #)%"3'(. CALR CALR Endoplasmic reticulum JAK2 515 exon 9 mutation MPL STAT ON exon 12 mutation or V617F HSC Other Genetic Mutations LNK, CBL, TET2, U2AF1, ASXL1, IDH1, IDH2, IKZF1, EZH2, DNMT3A, TP53, SF3B1, SFSR2 Primary Myelofibrosis: Molecular Biology, Clinical Features, Diagnosis, and Useful Resources Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZZW40
- 2. Clinical Manifestations in Patients With Myelofibrosis2 Splenomegaly • Abdominal pain • A feeling of early fullness • Bloating • High pressure in the liver vasculature Impaired Blood Cell Production • Thrombocytopenia • Anemia • Leukopenia Constitutional Systems • Fatigue, tiredness • Night sweats • Itching • Bone pain • Inactivity • Concentration problems • Fever • Weight loss Primary Myelofibrosis: Molecular Biology, Clinical Features, Diagnosis, and Useful Resources Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZZW40
- 3. WHO Criteria for Diagnosis of Primary Myelofibrosis3 Major Criteria Minor Criteria • Proliferation and atypia of megakaryocytes accompanied by either reticulin and/or collagen fibrosis grades 2 or 3 on a scale of 0 to 3 • Not meeting WHO criteria for ET, PV, BCR- ABL1+ CML, MDS, or other myeloid neoplasm • Presence of JAK2, CALR, or MPL mutation or in the absence of these mutations, presence of another clonal marker, or absence of reactive MF • Anemia not attributed to a comorbid condition • Leukocytosis ≥11 × 109 /L • Palpable splenomegaly • LDH increased to above upper normal limit of institutional reference range • Leukoerythroblastosis Diagnosis requires meeting all three major criteria, and at least two minor criteria confirmed in two consecutive determinations Primary Myelofibrosis: Molecular Biology, Clinical Features, Diagnosis, and Useful Resources Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZZW40
- 4. 1. Garmezy B. Blood Rev. 2020;100691. 2. Tefferi A. Am J Hematol. 2021;96:145-162. 3. Arber DA et al. Blood. 2016;127:2391-2324. Resources for Your Patients With Myelofibrosis The Leukemia Lymphoma Society https://www.lls.org/patient-support NCCN Guidelines for Patients®: Myeloproliferative Neoplasms https://www.nccn.org/patients/guidelines/content/PDF/mpn-patient.pdf MPN Education Foundation https://mpninfo.org/ MPN Cancer Connection https://mpncancerconnection.org/ National Cancer Institute https://www.cancer.gov/types/myeloproliferative MPN Research Foundation https://www.mpnresearchfoundation.org/ Clinical Resources for Prognosis and Risk Stratification DIPSS Plus Score for Prognosis in Myelofibrosis https://qxmd.com/calculate/calculator_315/dipss-plus-score-for-prognosis-in-myelofibrosis DIPSS (Dynamic International Prognostic Scoring System) for Myelofibrosis https://www.mdcalc.com/dipss-dynamic-international-prognostic-scoring-system-myelofibrosis MIPSS70 score http://www.mipss70score.it/ Estimating Prognosis in Myelofibrosis (MF) https://www.mpnconnect.com/estimate-myelofibrosis-prognosis-tool.aspx Primary Myelofibrosis: Molecular Biology, Clinical Features, Diagnosis, and Useful Resources Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZZW40
- 5. Ruxolitinib1,2,a FDA approval: November 16, 2011 Indication • Intermediate or high-risk myelofibrosis, including primary myelofibrosis, post-polycythemia vera myelofibrosis, and post-essential thrombocythemia myelofibrosis in adults Dosing Myelofibrosis • The starting dose is based on patient’s baseline platelet count: – 200 × 109 /L: 20 mg orally twice daily – 100 × 109 /L to 200 × 109 /L: 15 mg orally twice daily – 50 × 109 /L to 100 × 109 /L: 5 mg orally twice daily • Monitor complete blood counts every 2 to 4 weeks until doses are stabilized and then as clinically indicated. Modify or interrupt dosing for thrombocytopenia WARNING • Thrombocytopenia, anemia, and neutropenia • Risk of infection • Symptom exacerbation following interruption or discontinuation • Risk of non-melanoma skin cancer • Lipid elevations Adverse Events • Thrombocytopenia and anemia (incidence 20%) • Bruising, dizziness, headache, and diarrhea (incidence ≥10%) Fedratinib capsules3,4,b FDA approval: August 16, 2019 Indication • Intermediate-2 or high-risk primary or secondary (post-polycythemia vera or post-essential thrombocythemia) myelofibrosis in adults Dosing • Recommended dosage: 400 mg orally once daily with or without food for patients with a baseline platelet count of ≥50 × 109 /L • Reduce dose for patients taking strong CYP3A inhibitors or with severe renal impairment WARNING • Anemia and thrombocytopenia • Gastrointestinal toxicity • Hepatic toxicity • Amylase and lipase elevation Adverse Events • The most common adverse reactions (≥20%) are diarrhea, nausea, anemia, and vomiting Currently Approved JAK Inhibitors for Myelofibrosis BOXED WARNING: ENCEPHALOPATHY, INCLUDING WERNICKEb Serious and fatal encephalopathy, including Wernicke, has occurred in patients treated with fedratinib. Assess thiamine levels in all patients before starting fedratinib, periodically during treatment and as clinically indicated. Primary Myelofibrosis: Current and Emerging Treatment Options Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZZW40 a ONLY myelofibrosis-specific indications, doses, and adverse events are included here. See full prescribing information for complete list (https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/202192Orig1s019Rpllbl.pdf). b See full prescribing information for complete information, including assessment and management of warning/boxed warning (https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212327s000lbl.pdf).
- 6. Selected Clinical Trials for Emerging JAK-Based Combinations for Myelofibrosis5 Primary Myelofibrosis: Current and Emerging Treatment Options Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZZW40 NCT Number Phase Status Study Title Agent (Therapeutic Target) NCT02158858 Phase 1 Phase 2 Recruiting Recruititng Recruiting Recruiting A Phase 2 Study of CPI-0610 With and Without Ruxolitinib in Patients With Myelofibrosis CPI-0610 (small-molecule inhibitor of BET protein) Ruxolitinib (JAKi) NCT04603495 Phase 3 Study of CPI-0610 in Myelofibrosis (MF) (MANIFEST-2) CPI-0610 (small-molecule inhibitor of BET protein) NCT03136185 Phase 2 Active, not recruiting IMG-7289 in Patients With Myelofibrosis IMG-7289 (LSD1 inhibitor) NCT04576156 Phase 3 A Study Comparing Imetelstat Versus Best Available Therapy for the Treatment of Intermediate-2 or High-risk Myelofibrosis (MF) Who Have Not Responded to Janus Kinase (JAK)-Inhibitor Treatment Imetelstat (telomerase inhibitor ) NCT03069326 Phase 2 A Clinical Study to Test the Effects of Ruxolitinib and Thalidomide Combination for Patients With Myelofibrosis Ruxolitinib (JAKi) Thalidomide (IMiD)
- 7. Primary Myelofibrosis: Current and Emerging Treatment Options Full abbreviations, accreditation, and disclosure information available at PeerView.com/ZZW40 Selected Clinical Trials for Emerging JAK-Based Combinations for Myelofibrosis5 (Cont’d) 1. Jakafi (ruxolitinib) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/202192Orig1s019Rpllbl.pdf. 2. https://www.drugs.com/newdrugs/fda-approves-jakafi-myelofibrosis-2949.html. 3. Inrebic (fedratinib) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/212327s000lbl.pdf. 4. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-fedratinib-myelofibrosis#:~:text=On%20August%2016%2C%202019%2C% 20 the,thrombocythemia)%20myelofibrosis%20(MF). 5. https://clinicaltrials.gov/. NCT Number Phase Status Study Title Agent (Therapeutic Target) NCT04097821 Phase 1 Phase 2 Recruiting Recruititng Recruiting Recruiting Platform Study of Novel Ruxolitinib Combinations in Myelofibrosis Patients Ruxolitinib (JAKi) Siremadlin (p53-MDM2 inhibitor) Crizanlizumab (anti–P-selectin antibody) Sabatolimab (anti–TIM-3 antibody) LTT462 (ERK1/2 inhibitor) NIS793 (anti–TGF-β antibody) NCT03878199 Phase 1 Phase 2 Testing the Effect of Taking Ruxolitinib and CPX-351 in Combination for the Treatment of Advanced Phase Myeloproliferative Neoplasms Ruxolitinib (JAKi) CPX-351 (cytarabine and daunorubicin) NCT04468984 Phase 3 Recruiting Study of Oral Navitoclax Tablet in Combination With Oral Ruxolitinib Tablet to Assess Change in Spleen Volume in Adult Participants With Relapsed/Refractory Myelofibrosis Navitoclax (Bcl-2 inhibitor) Ruxolitinib (JAKi) NCT03222609 Phase 2 A Study Evaluating Tolerability and Efficacy of Navitoclax Alone or in Combination With Ruxolitinib in Participants With Myelofibrosis Navitoclax (Bcl-2 inhibitor) Ruxolitinib (JAKi) NCT04472598 Phase 3 Study of Oral Navitoclax Tablet in Combination With Oral Ruxolitinib Tablet When Compared With Oral Ruxolitinib Tablet to Assess Change in Spleen Volume in Adult Participants With Myelofibrosis Navitoclax (Bcl-2 inhibitor) Ruxolitinib (JAKi)
