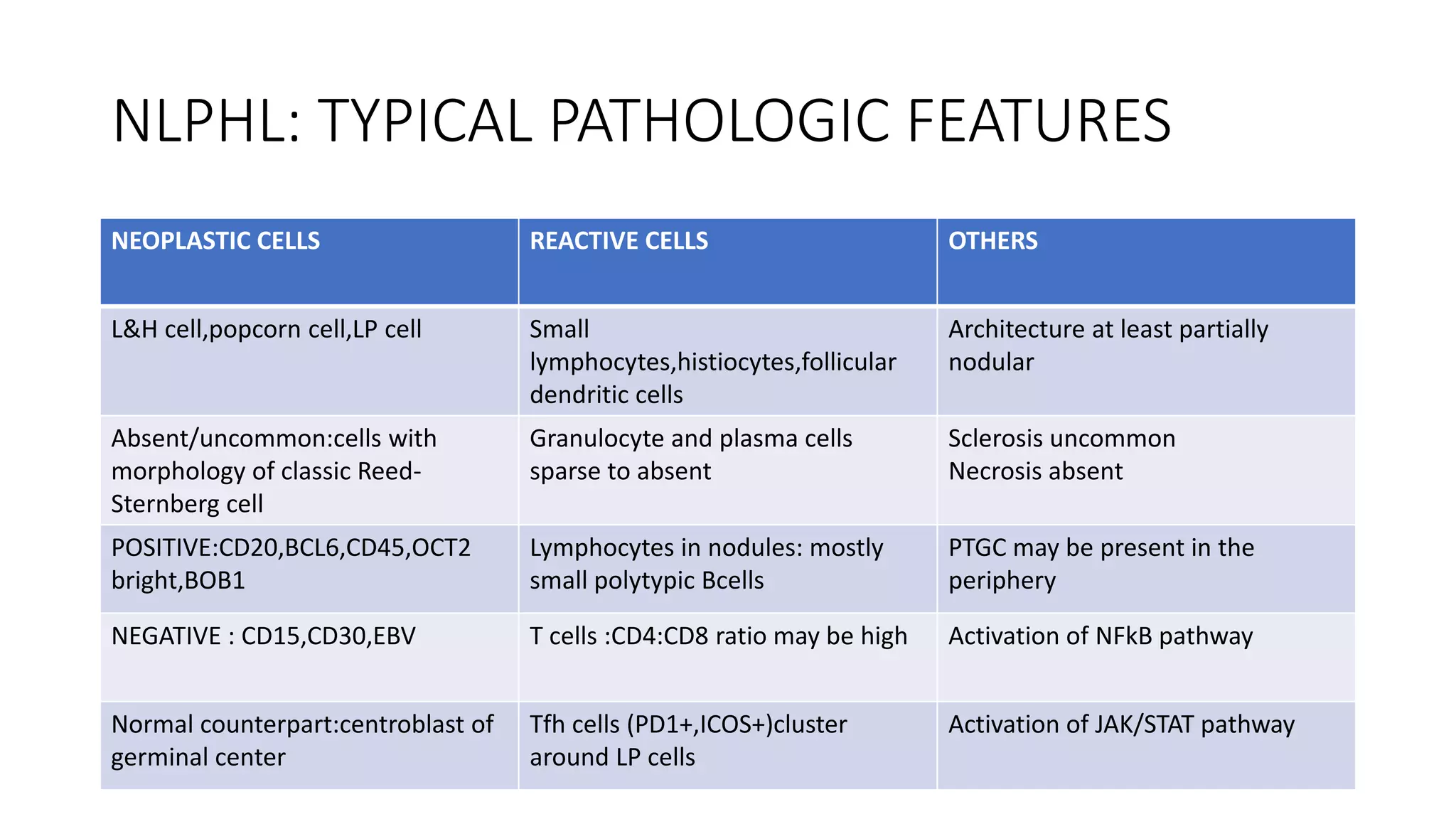
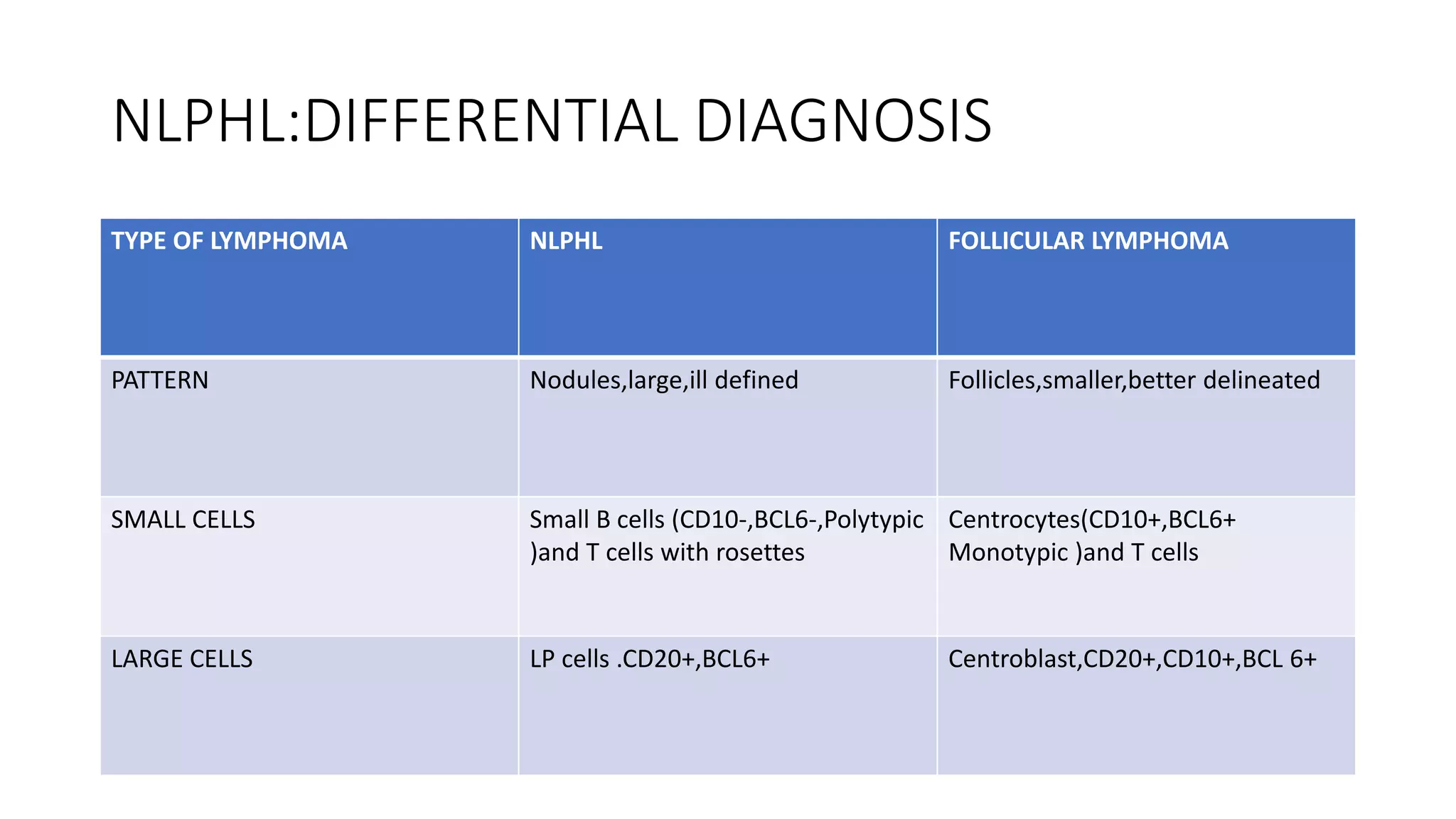
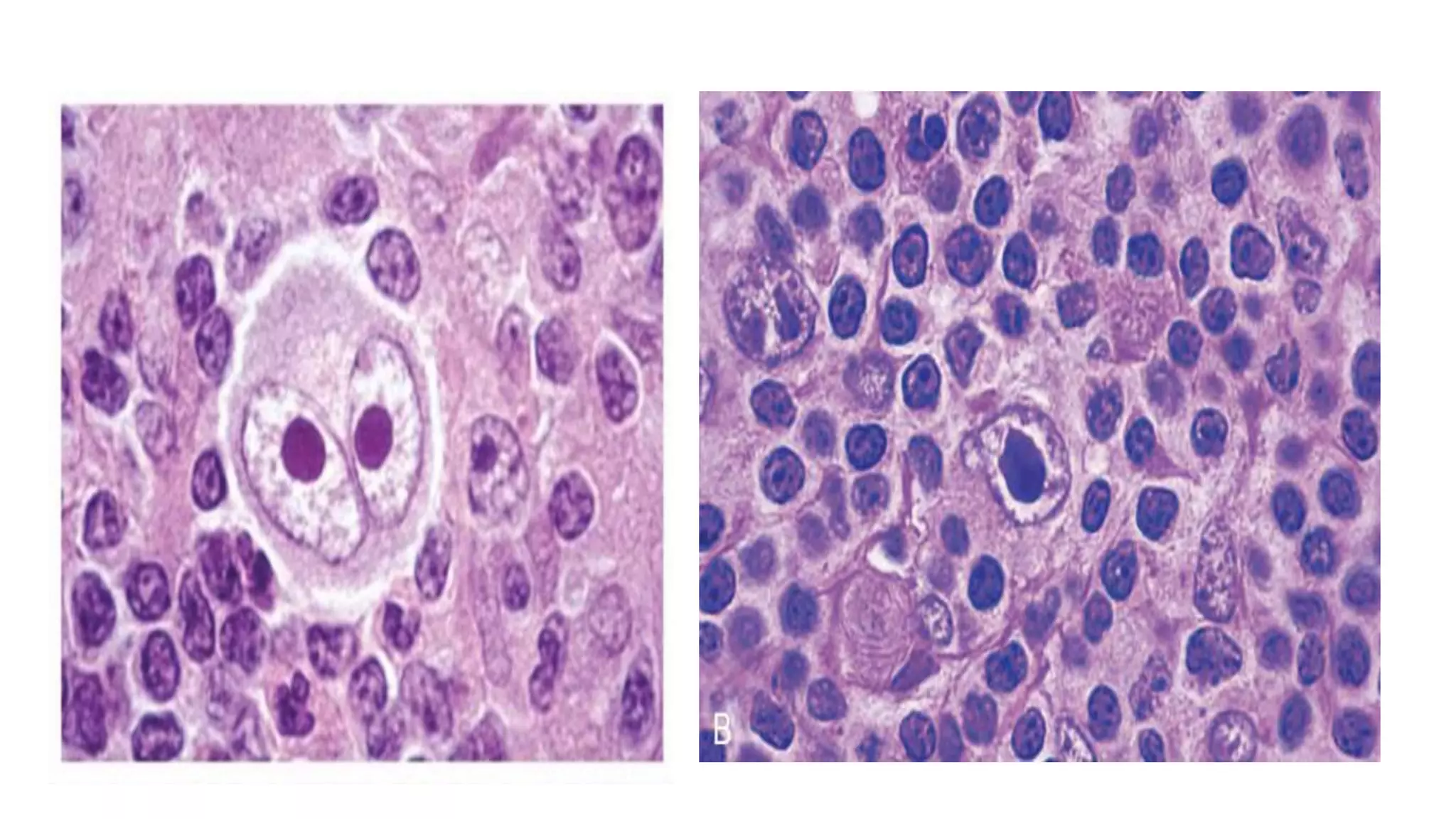
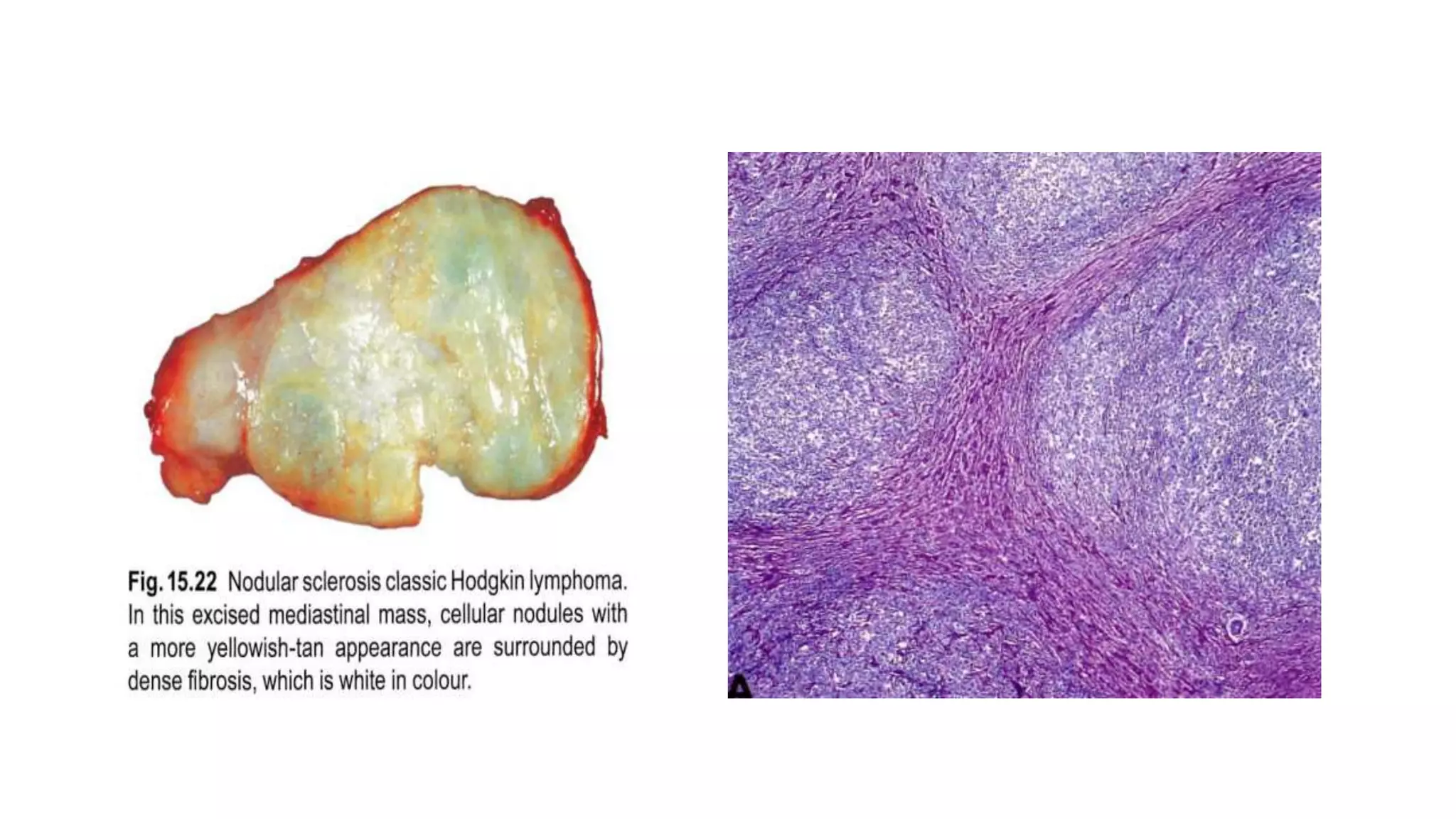
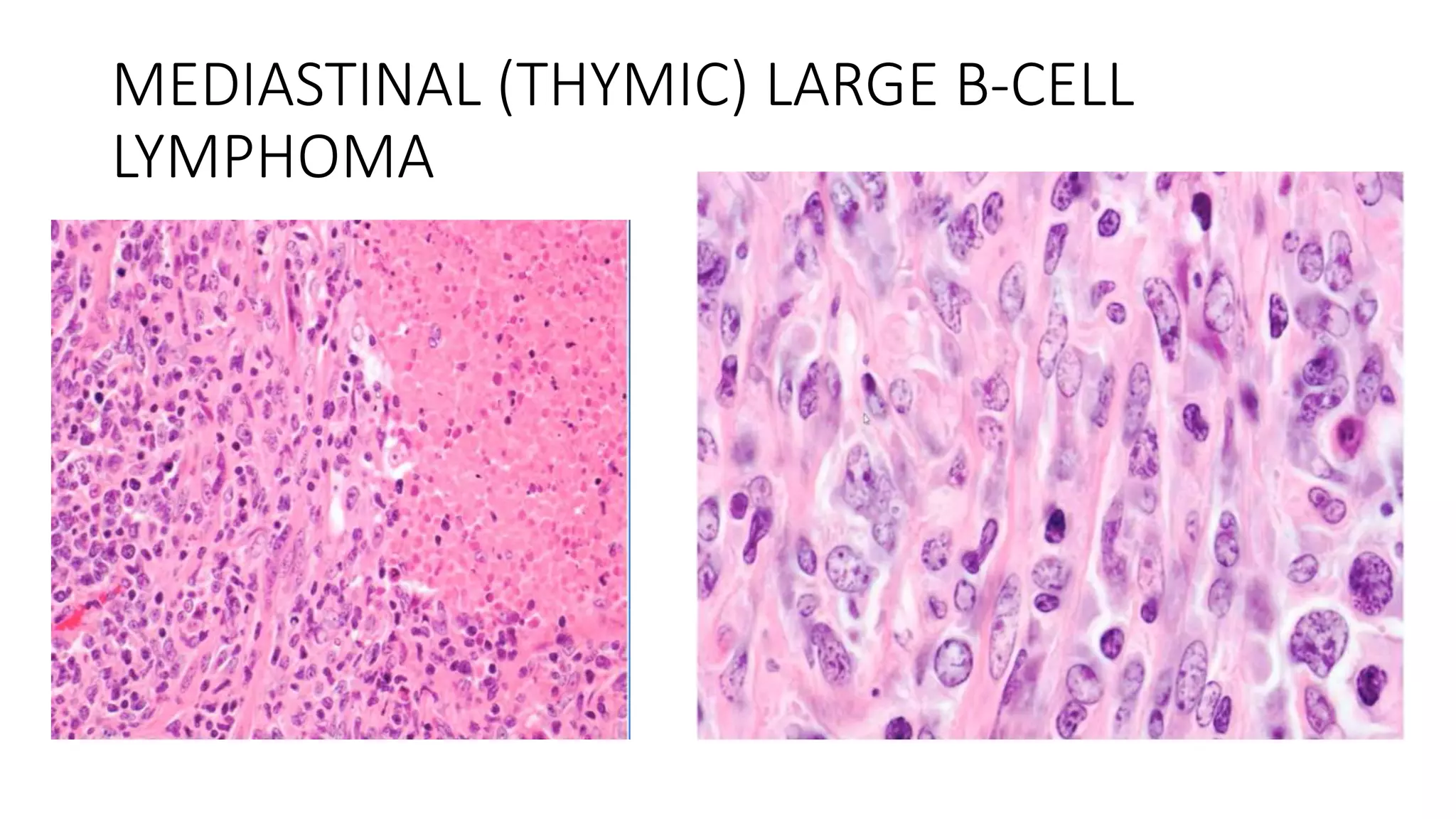
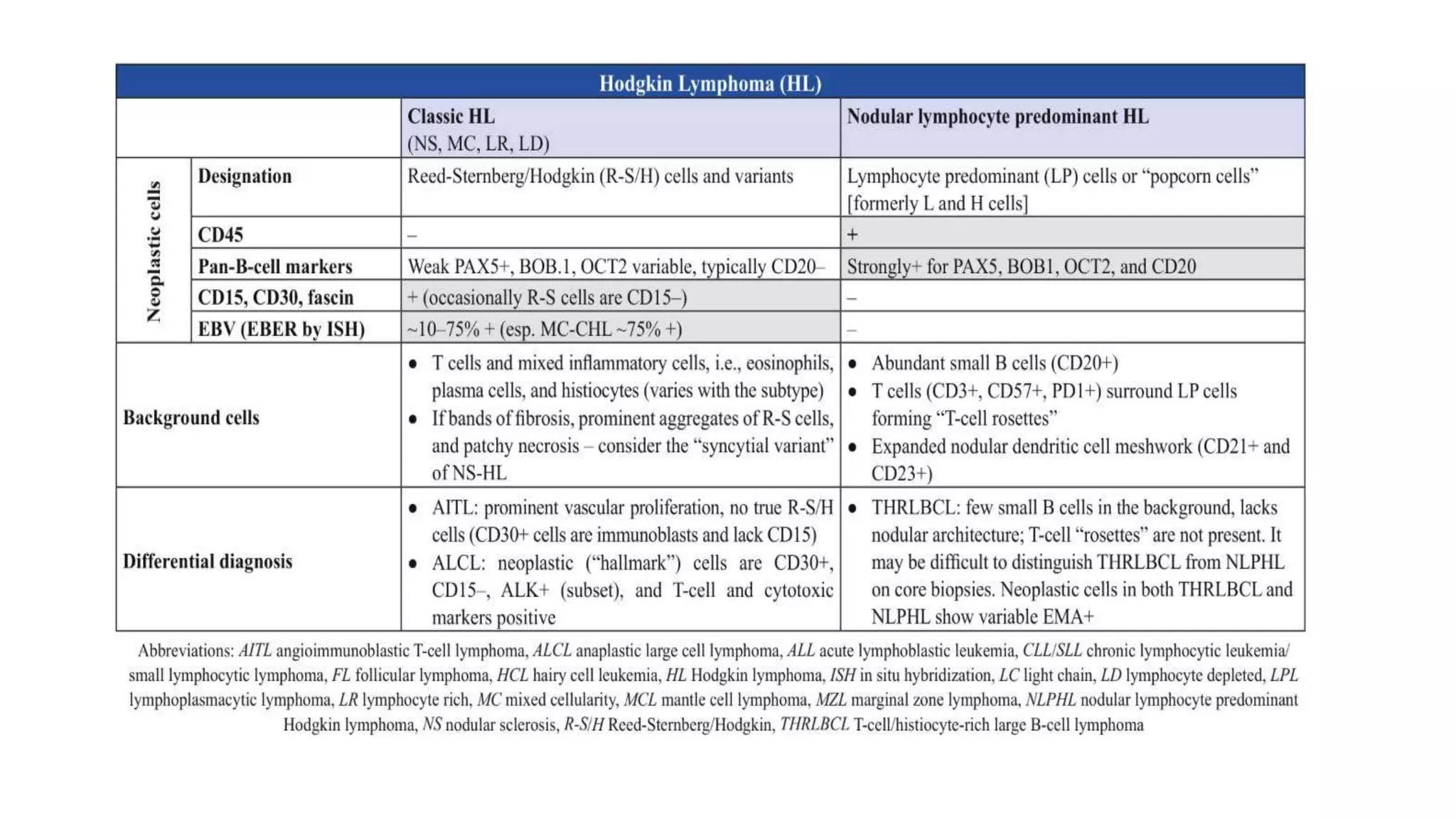
This document provides an overview of Hodgkin lymphoma, including its normal structure and histology, WHO classification, and subtypes. It discusses the pathogenesis and epidemiology of Hodgkin lymphoma, describing the four main subtypes of classical Hodgkin lymphoma and nodular lymphocyte predominant Hodgkin lymphoma. Key features of each subtype are summarized, along with diagnostic criteria and differential diagnoses. Prognosis and management are also briefly covered.