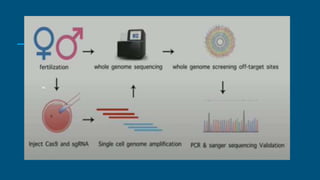
1. In November 2018, He Jiankui revealed that he used CRISPR gene-editing to genetically modify twin babies, Lulu and Nana, born in late 2018.
2. He claims he disabled the CCR5 gene to make the babies resistant to HIV, as their father was HIV positive. However, He failed to receive proper ethics approval and conducted unsafe and unethical human experimentation.
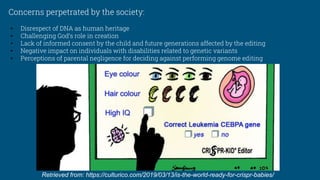
3. He's actions faced significant backlash from the scientific community due to violations of ethical standards and risks to human research subjects.