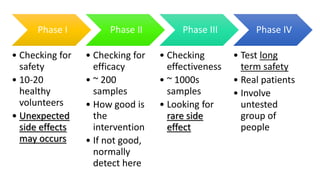
The document provides an overview of good clinical practices (GCP) and the historical standards that contributed to its development. It discusses key milestones like the Nuremberg Code, Declaration of Helsinki, and Belmont Report that established ethical and quality standards for clinical research involving human subjects. The document also outlines the four phases of clinical trials and principles of GCP like prior approval, informed consent, and quality assurance. It notes that GCP provides an international quality standard to ensure the rights, safety, and well-being of clinical trial participants.