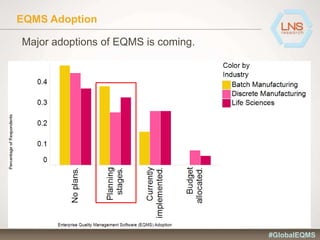
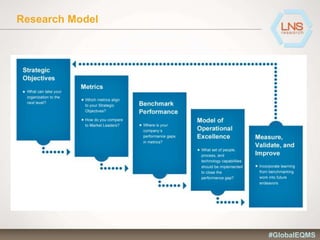
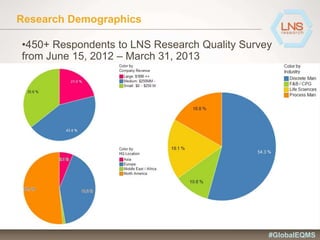
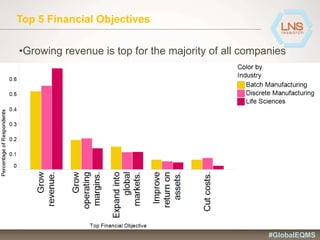
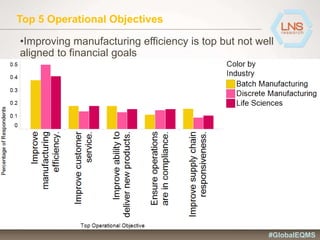
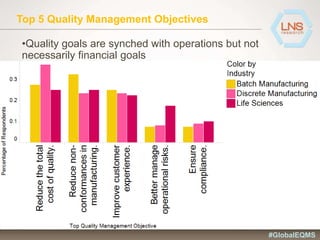
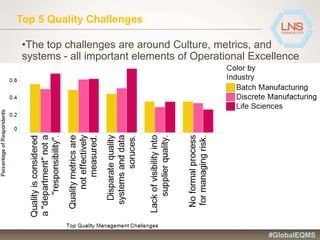
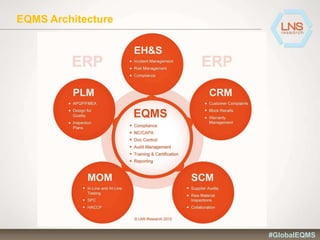
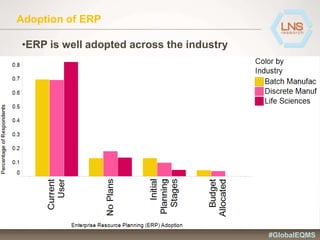
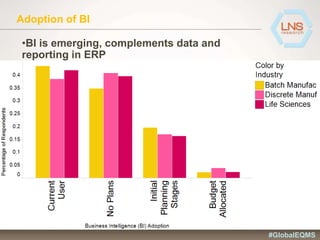
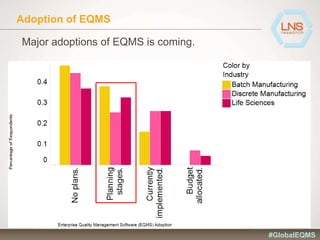
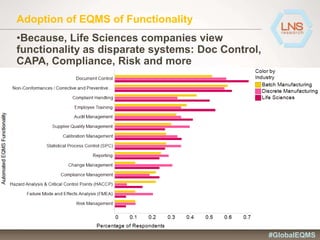
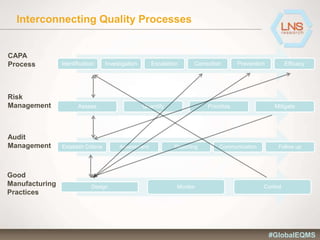
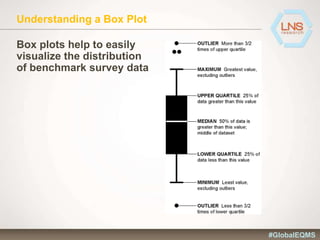
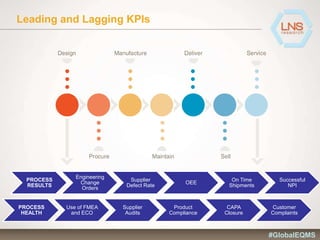
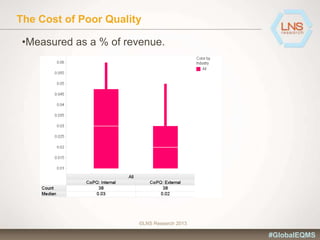
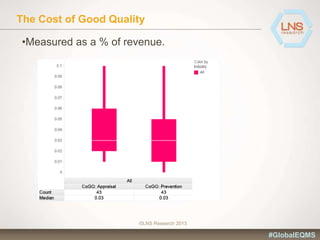
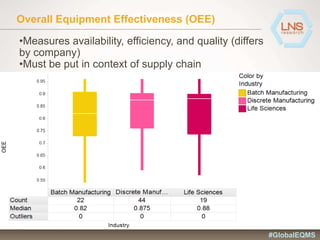
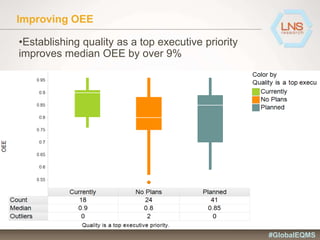
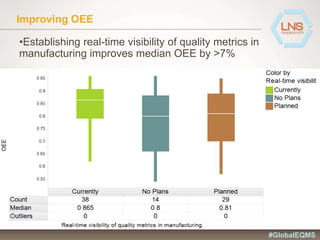
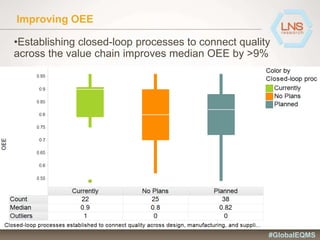
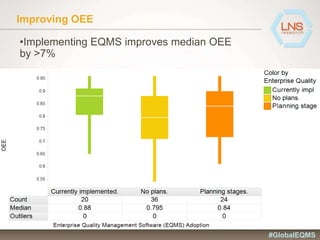
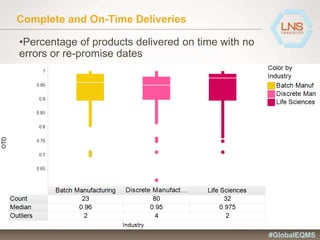
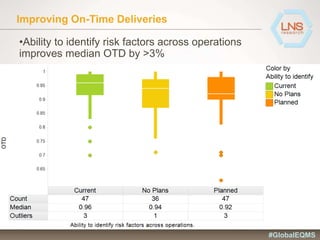
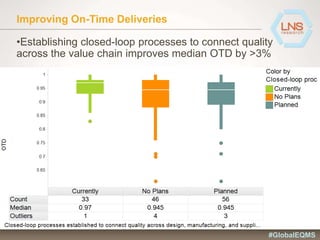
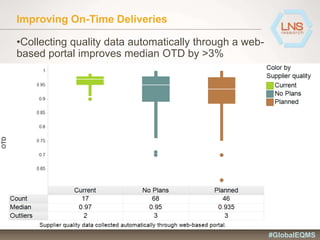
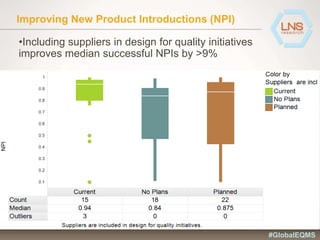
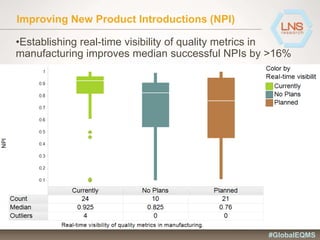
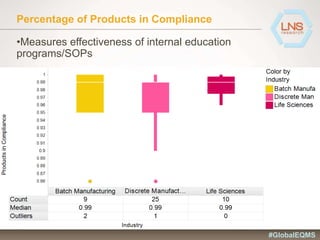
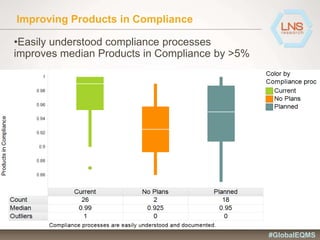
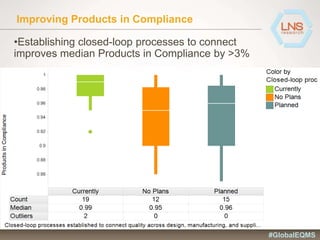
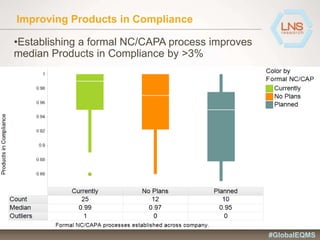
The document discusses the importance of Enterprise Quality Management Software (EQMS) in improving operational excellence, addressing key challenges in quality management, and aligning quality objectives with financial goals. It highlights insights from the LNS research and a global quality advisory council, focusing on metrics such as Overall Equipment Effectiveness (OEE) and the cost of quality. Recommendations for success include defining the role of quality in strategic objectives and creating closed-loop quality management processes.