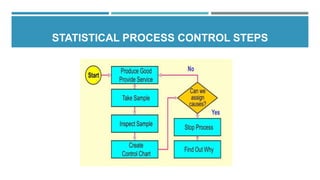
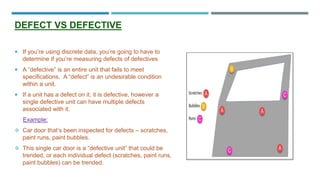
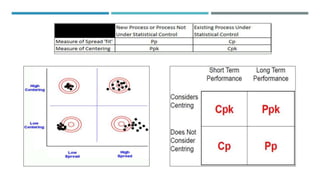
This document provides an overview of statistical process control (SPC). It discusses the key concepts of SPC including the 5M's (man, machine, material, method, milieu), control chart basics, process variability, common SPC tools like control charts, histograms, Pareto charts, and their purposes. Control charts are described as the most important SPC tool for distinguishing common from special cause variation to monitor if a process is in control. The document also covers variable and attribute control charts and considerations for chart selection based on data type.